TRANDATE INJECTION 100 MG/20 ML [SIN02281P]
Active ingredients: TRANDATE INJECTION 100 MG/20 ML
Product Info
TRANDATE INJECTION 100 MG/20 ML
[SIN02281P]
Product information
Active Ingredient and Strength | LABETALOL HCL - 100 MG/20 ML |
Dosage Form | INJECTION |
Manufacturer and Country | UBI PHARMA INC - TAIWAN |
Registration Number | SIN02281P |
Licence Holder | DCH AURIGA SINGAPORE |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | C07AG01 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Indications
TRANDATE injection is indicated for:
▪ severe hypertension, including severe hypertension of pregnancy, when rapid control of blood pressure is essential
▪ May be used to achieve control hypotension during anaesthesia
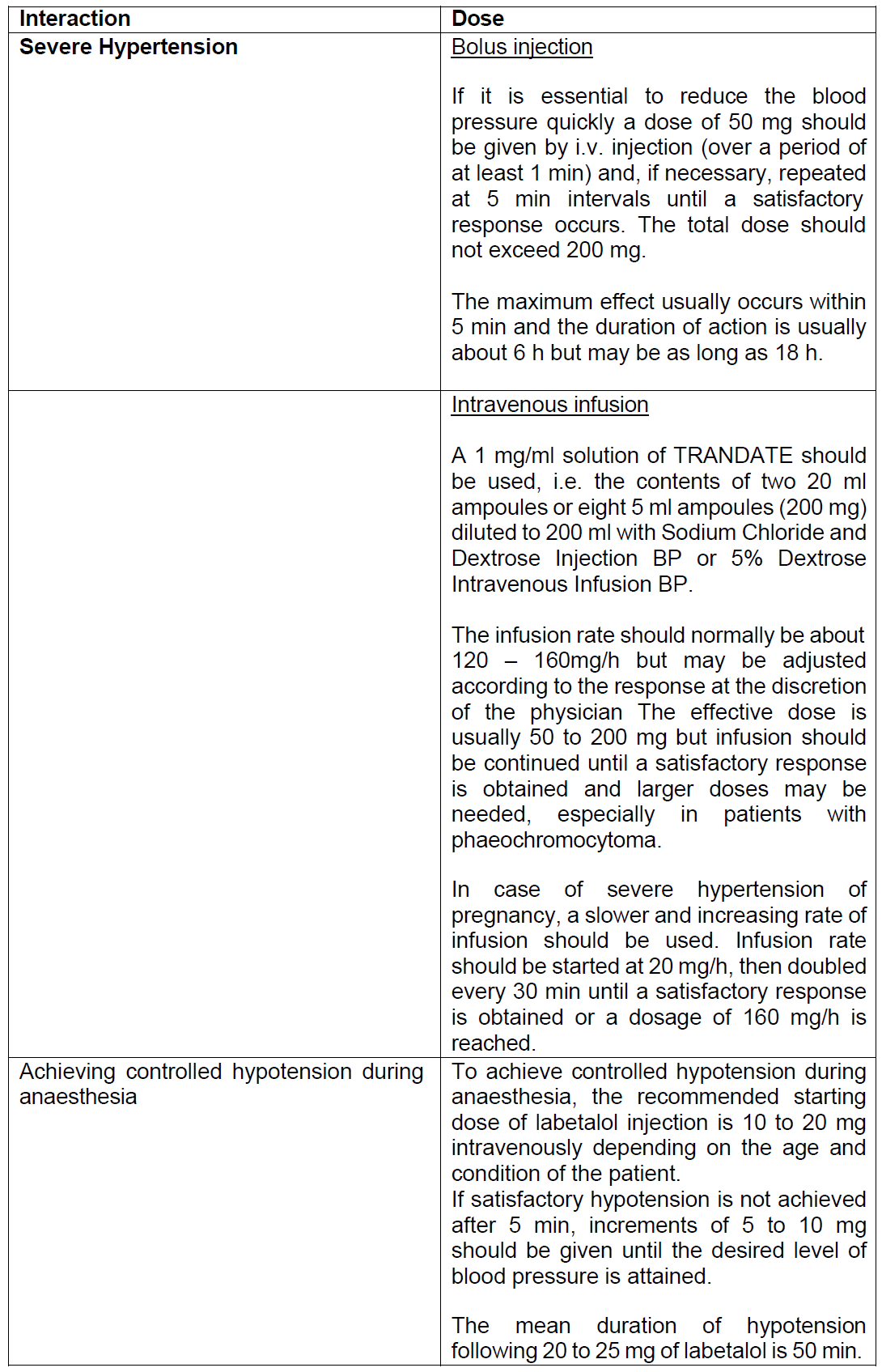
Dosing
Dosage and Administration
TRANDATE injection is intended for i.v. use in hospitalised patients. Patients should always receive the drug whilst in the supine or left lateral position. Raising the patient into the upright position within 3 h of i.v. TRANDATE administration should be avoided since excessive postural hypotension may occur. It is desirable to monitor the blood pressure and heart rate after injection and during infusion. In most patients, there is a small decrease in the heart rate; severe bradycardia is unusual but may be controlled by injecting atropine 1 to 2 mg intravenously. Respiratory function should be observed particularly in patients with any known impairment.
TRANDATE injection has been administered to patients with uncontrolled hypertension already receiving other hypotensive agents, including beta-blocking drugs, without adverse effects.
Populations
• Adults
• Paediatric population
Safety and efficacy in paediatric patients aged 0 to 18 years has not been established. No data are available.
Contraindications
Contraindications
TRANDATE injection is contraindicated in second or third degree heart block (unless pacemaker is in situ), cardiogenic shock and other conditions associated with severe and prolonged hypotension or severe bradycardia.
Non-selective beta-blockers should not be used in patients with asthma or a history of obstructive airway disease.
Uncompensated heart failure
Unstable/uncontrolled heart insufficiency
Sick sinus syndrome (including sinus atrial block) unless pacemaker in situ
Sinus node dysfunction
Prinzmetal angina
TRANDATE injection is contraindicated for patients known to have hypersensitivity to the active substance or to any of the excipients listed.
