ACTILYSE TREATMENT-SET 50 MG/VIAL [SIN02340P]
Active ingredients: ACTILYSE TREATMENT-SET 50 MG/VIAL
Product Info
ACTILYSE TREATMENT-SET 50 MG/VIAL
[SIN02340P]
Product information
Active Ingredient and Strength | RECOMBINANT HUMAN TISSUE-TYPE PLASMINOGEN ACTIVATOR - 50 MG/VIAL |
Dosage Form | INJECTION, POWDER, FOR SOLUTION |
Manufacturer and Country | BOEHRINGER INGELHEIM PHARMA GMBH & CO KG - GERMANY |
Registration Number | SIN02340P |
Licence Holder | BOEHRINGER INGELHEIM SINGAPORE PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | B01AD02 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
1. Thrombolytic treatment in acute myocardial infarction (AMI)
90 minutes (accelerated) dose regimen (see section 4.2 Posology and administration):
for patients in whom treatment can be started within 6 hours of symptom onset;3 hours dose regimen (see section 4.2 Posology and administration):
for patients in whom treatment can be started between 6 – 12 hours after symptom onset.
ACTILYSE has proven to reduce 30-day-mortality in patients with acute myocardial infarction.
2. Thrombolytic treatment in acute massive pulmonary embolism with haemodynamic instability (PE)
The diagnosis should be confirmed whenever possible by objective means such as pulmonary angiography or non-invasive procedures such as lung scanning.
There are no clinical trials on mortality and late morbidity related to pulmonary embolism.
3. Thrombolytic treatment of acute ischaemic stroke (AIS)
Treatment must be started as early as possible within 4.5 hours after onset of stroke symptoms and after exclusion of intracranial haemorrhage by appropriate imaging techniques (e.g. cranial computerised tomography or other diagnostic imaging method sensitive for the presence of haemorrhage). The treatment effect is time-dependent; therefore earlier treatment increases the probability of a favourable outcome.
Dosing
4.2 Posology and method of administration
ACTILYSE should be given as early as possible after symptom onset.
Posology
1. Acute myocardial infarction
a) 90 minutes (accelerated) dose regimen
for patients with acute myocardial infarction, in whom treatment can be started within 6 hours after symptom onset.
In patients with a body weight ≥ 65 kg:
15 mg as an intravenous bolus, immediately followed by
50 mg as an intravenous infusion over the first 30 minutes, immediately followed by an intravenous infusion of
35 mg over 60 minutes, until the maximum total dose of 100 mg.
In patients with a body weight < 65 kg the total dose should be weight adjusted with
15 mg as an intravenous bolus, immediately followed by
0.75 mg/kg body weight as an intravenous infusion over the first 30 minutes (maximum 50 mg), immediately followed by an intravenous infusion of
0.5 mg/kg over 60 minutes (up to a maximum of 35 mg).
b) 3 hours dose regimen
for patients with acute myocardial infarction, in whom treatment can be started between 6 and 12 hours after symptom onset:
In patients with a body weight ≥ 65 kg:
10 mg as an intravenous bolus, immediately followed by
50 mg as an intravenous infusion over the first hour, immediately followed by an intravenous infusion of
40 mg over two hours, until the maximum total dose of 100 mg.
In patients with a body weight < 65 kg:
10 mg as an intravenous bolus, immediately followed by
an intravenous infusion over three hours up to a maximum total dose of 1.5 mg/kg body weight.
Adjunctive therapy:
Antithrombotic adjunctive therapy is recommended according to the current international guidelines for the management of patients with ST-elevation myocardial infarction.
2. Acute massive pulmonary embolism
In patients with a body weight ≥ 65 kg:
A total dose of 100 mg should be administered in 2 hours. The most experience available is with the following dose regimen:
10 mg as an intravenous bolus over 1 – 2 minutes, immediately followed by
90 mg as an intravenous infusion over two hours until the total dose of 100mg
In patients with a body weight < 65 kg:
10 mg as an intravenous bolus over 1 – 2 minutes, immediately followed by
an intravenous infusion up to a maximum total dose of 1.5 mg/kg.
Adjunctive therapy:
After treatment with ACTILYSE heparin therapy should be initiated (or resumed) when aPTT values are less than twice the upper limit of normal. The infusion should be adjusted to maintain aPTT between 50 – 70 seconds (1.5 to 2.5 fold of the reference value).
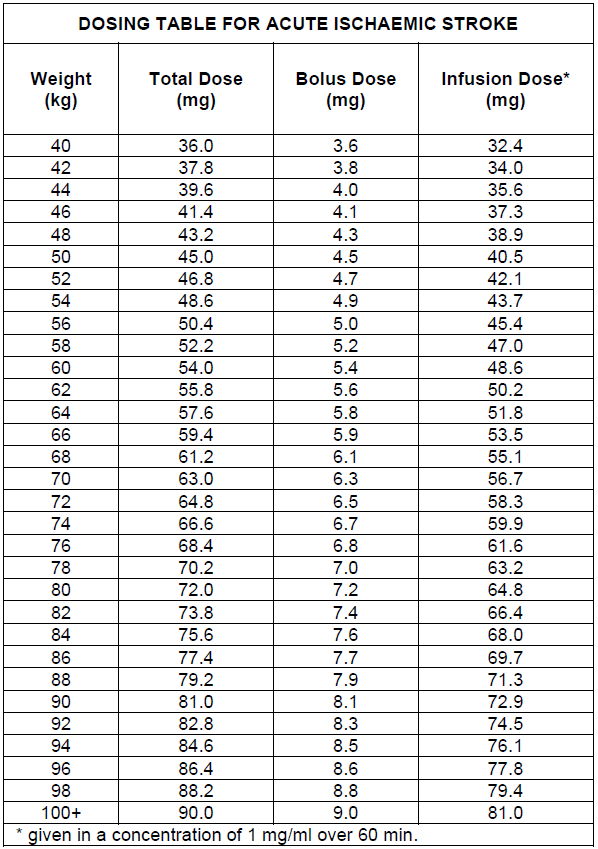
3. Acute ischaemic stroke
The recommended total dose is 0.9 mg/kg body weight (maximum of 90 mg) infused starting with 10% of the total dose as an initial intravenous bolus, immediately followed by the remainder of the total dose infused intravenously over 60 minutes.
Treatment should be initiated for adults and children ≥ 16 years of age (see sections 4.3 Contraindications and 4.4 Special warnings and precautions) as early as possible and no later than 4.5 hours after last known well and after exclusion of intracranial haemorrhage by appropriate imaging techniques. The treatment effect is time-dependent; therefore, earlier treatment increases the probability of a favourable outcome. Beyond 4.5 hours after onset of stroke symptoms there is a negative benefit risk ratio associated with Actilyse administration and so it should not be administered (see 5.1 Pharmacological properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Adjunctive therapy:
Drugs affecting coagulation/platelet function
The safety and efficacy of this regimen with concomitant administration of heparin or platelet aggregation inhibitors such as acetylsalicylic acid during the first 24 hours after treatment with Actilyse has not been investigated sufficiently. Therefore, administration of intravenous heparin or platelet aggregation inhibitors such as acetylsalicylic acid should be avoided in the first 24 hours after treatment with ACTILYSE due to an increased haemorrhagic risk.
If heparin is required for other indications (e.g. prevention of deep vein thrombosis) the dose should not exceed 10,000 international units per day, administered subcutaneously.
Method of administration
The reconstituted solution should be administered intravenously and is for immediate use.
Handling Instructions
Under aseptic conditions the contents of an injection vial of ACTILYSE (50 mg) dry substance is dissolved with sterilised water for injection according to the following table to obtain a final concentration of 1 mg alteplase per ml.

For this purpose a transfer cannula is included with the pack-sizes of 50 mg.
Instructions for reconstituting Actilyse
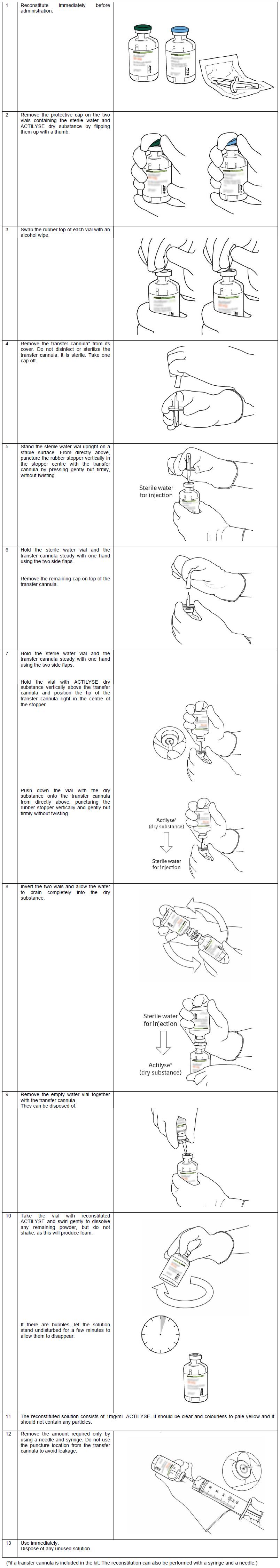
The 1mg/mL reconstituted solution may be diluted further with sterile sodium chloride 9 mg/mL (0.9%) solution for Injection up to a minimal concentration of 0.2 mg/mL since the occurrence of turbidity of the reconstituted solution cannot be excluded.
A further dilution of the 1mg/mL reconstituted solution with sterilised water for injections or in general, the use of carbohydrate infusion solutions, e.g. dextrose is not recommended due to increasing formation of turbidity of the reconstituted solution.
ACTILYSE should not be mixed with other drugs, neither in the same infusion-vial nor the same venous line (not even with heparin).
Traceability
In order to improve traceability of biological medicinal products, the trade name and the batch number of the administered product should be clearly recorded in the patient file.
Contraindications
4.3 Contraindications
ACTILYSE is contraindicated in
patients with known hypersensitivity to the active substance alteplase or to any of the excipients
situations associated with a risk of bleeding such as:
significant bleeding disorder at present or within the past 6 months, known haemorrhagic diathesis
any history of central nervous system damage (i.e. neoplasm, aneurysm, intracranial or spinal surgery)
severe uncontrolled arterial hypertension (see section 4.4 Special warnings and precautions for use – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information)
major surgery or significant trauma in the past 10 days (this includes any trauma associated with the current acute myocardial infarction), recent trauma to head or cranium
obstetrical delivery, within the past 10 days, recent puncture of a non-compressible blood-vessel (e.g. subclavian or jugular vein puncture)
severe hepatic dysfunction, including hepatic failure, cirrhosis, portal hypertension (oesophageal varices) and active hepatitis
bacterial endocarditis, pericarditis
acute pancreatitis
active ulcerative gastro-intestinal disease
known arterial aneurysm, and/or arterial/venous malformation
neoplasm with increased bleeding risk
In the indication of acute myocardial infarction and acute massive pulmonary embolism the following contraindications apply in addition:
Situations associated with a risk of bleeding such as:
haemorrhagic stroke or stroke of unknown origin at any time
patients receiving effective oral anticoagulant treatment (e.g. vitamin K antagonists with INR > 1.3) (please see section 4.4 Special warnings and precautions for use, subsection Bleeding – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information)
In the indication acute ischaemic stroke the following contraindications apply in addition:
Situations associated with a risk of bleeding such as:
symptoms of acute ischaemic stroke that were either rapidly improving or only minor before start of infusion
severe stroke as assessed clinically (e.g. NIHSS > 25) and/or by appropriate imaging techniques
patients receiving effective oral anticoagulation (e.g. vitamin K antagonists with INR > 1.7) (please see section 4.4 Special warnings and precautions for use, subsection Bleeding – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information)
administration of heparin within 48 hours preceding the onset of stroke with an elevated activated partial thromboplastin time (aPTT) at presentation
platelet count of less than 100,000 / mm3
history or evidence or suspicion of intracranial haemorrhage including sub-arachnoid haemorrhage
history of serious head-trauma within three months
children under 16 years of age (for children ≥ 16 years of age see section Special warnings and precautions for use – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information)
