POLARINE SYRUP [SIN03182P]
Active ingredients: POLARINE SYRUP
Product Info
POLARINE SYRUP
[SIN03182P]
Product information
Active Ingredient and Strength | DEXCHLORPHENIRAMINE MALEATE - 2 MG/5 ML |
Dosage Form | SYRUP |
Manufacturer and Country | SUNWARD PHARMACEUTICAL PRIVATE LIMITED - SINGAPORE |
Registration Number | SIN03182P |
Licence Holder | SUNWARD PHARMACEUTICAL PRIVATE LIMITED |
Forensic Classification | PHARMACY ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | R06AB52 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Indications:
It is indicated in nasal and respiratory congestion, common cold, acute sinusitis, allergic rhinitis.
Dosing
Dosage:
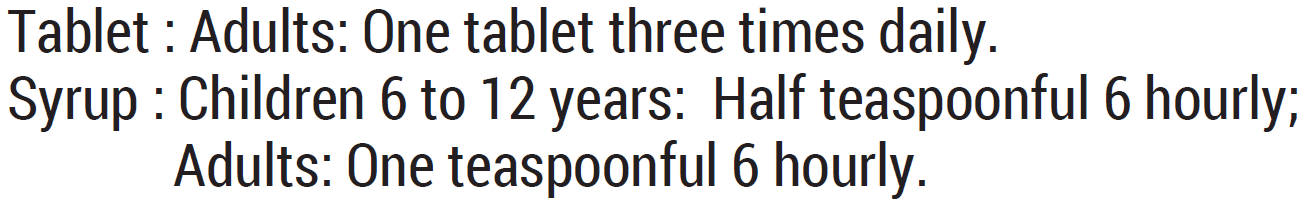
Contraindications
Contraindications:
This formula is contraindicated in patients exhibiting hypersensitivity to any of the components. Antihistamine are contraindicated in patients receiving monoamine oxidase inhibitors since these agents prolong and intensify the anticholinergic effects of anti-histamines. Antihistamines should not be used to treat lower respiratory tract symptoms. Sympathomimetic preparations are contraindicated in patients with severe hypertension, severe coronary artery disease and patients receiving MAO inhibitor therapy due to potentiation of the pressor effects of pseudoephedrine.
