VALTREX TABLET 500 MG [SIN08229P]
Active ingredients: VALTREX TABLET 500 MG
Product Info
VALTREX TABLET 500 MG
[SIN08229P]
Product information
Active Ingredient and Strength | VALACICLOVIR HCL EQV VALACICLOVIR - 500 MG |
Dosage Form | TABLET, FILM COATED |
Manufacturer and Country | GLAXO WELLCOME SA - SPAIN |
Registration Number | SIN08229P |
Licence Holder | GLAXOSMITHKLINE PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J05AB11 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Indications
VALTREX is indicated for the treatment of herpes zoster (shingles). VALTREX accelerates the resolution of pain: it reduces the duration of and the proportion of patients with zoster-associated pain, which includes acute and post-herpetic neuralgia.
VALTREX is indicated for the treatment of herpes simplex infections of the skin and mucous membranes, including initial and recurrent genital herpes.
VALTREX is indicated for the prevention (suppression) of recurrent herpes simplex infections of the skin and mucous membranes, including genital herpes.
VALTREX is indicated for reduction of transmission of genital herpes in patients suffering from recurrent genital herpes. In addition to therapy with VALTREX, it is recommended that patients use safer sex practices.
Dosing
Dosage and Administration
Pharmaceutical form: Film-coated tablets
• Adults
Treatment of herpes zoster (shingles)
The dosage is 1000 mg of VALTREX to be taken three times daily for seven days.
Treatment of herpes simplex infections
The dosage is 500 mg of VALTREX to be taken twice daily.
For recurrent episodes, treatment should be for five days. For initial episodes, which can be more severe, treatment may have to be extended from five days to ten days. Dosing should begin as early as possible. For recurrent episodes of herpes simplex, this should ideally be during the prodromal period or immediately when the first signs or symptoms appear. VALTREX can prevent lesion development when taken at the first signs and symptoms of an HSV recurrence.
Prevention (suppression) of recurrences of herpes simplex infections
Immunocompetent adults
In immunocompetent patients, 500 mg of VALTREX to be taken once daily.
Some patients with very frequent recurrences (e.g. 10 or more per year) may gain additional benefit from the daily dose of 500 mg being taken as a divided dose (250 mg twice daily).
Immunocompromised adults
For immunocompromised patients, the dose is 500 mg twice daily.
Reduction of transmission of genital herpes
In immunocompetent heterosexual adults with 9 or fewer recurrences per year and with the susceptible partner discordant for HSV-2 antibodies, 500 mg of VALTREX to be taken once daily by the infected partner.
The efficacy of reducing transmission beyond 8 months in discordant couples has not been established.
There are no data on the reduction of transmission in other patient populations.
• Children
There are no data available on the use of VALTREX in children.
• Elderly
The possibility of renal impairment in the elderly must be considered and the dosage should be adjusted accordingly (see Renal impairment below).
Adequate hydration should be maintained.
• Renal impairment
Caution is advised when administering valaciclovir to patients with impaired renal function. Adequate hydration should be maintained.
The dosage of VALTREX should be reduced in patients with impaired renal function as shown in the table below:
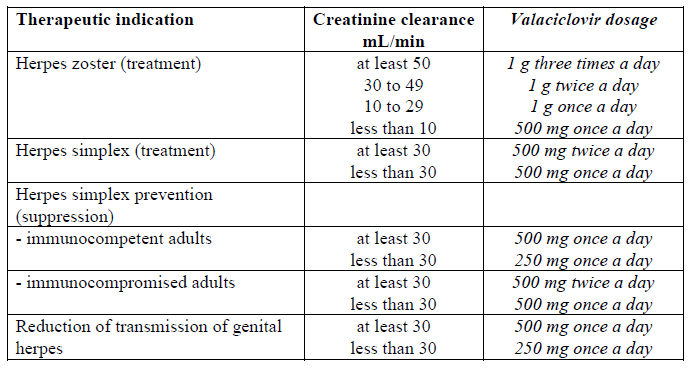
In patients on intermittent haemodialysis, the VALTREX dosage should be administered after the haemodialysis has been performed.
The creatinine clearance should be monitored frequently, especially during periods when renal function is changing rapidly, the VALTREX dosage should be adjusted accordingly.
• Hepatic impairment
Studies with a 1 g unit dose of VALTREX show that dose modification is not required in patients with mild or moderate cirrhosis (hepatic synthetic function maintained). Pharmacokinetic data in patients with advanced cirrhosis (impaired hepatic synthetic function and evidence of portal-systemic shunting), do not indicate the need for dosage adjustment; however, clinical experience is limited. For higher doses (4 g or more/day), see Warnings and Precautions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Contraindications
Contraindications
VALTREX is contraindicated in patients known to be hypersensitive to valaciclovir, aciclovir or any components of formulations of VALTREX.
