PENTAGLOBIN INJECTION 50 MG/ML [SIN11205P]
Active ingredients: PENTAGLOBIN INJECTION 50 MG/ML
Product Info
PENTAGLOBIN INJECTION 50 MG/ML
[SIN11205P]
Product information
Active Ingredient and Strength | IMMUNOGLOBULIN (HUMAN) - 50 MG/ML |
Dosage Form | INJECTION |
Manufacturer and Country | BIOTEST AG - GERMANY |
Registration Number | SIN11205P |
Licence Holder | GRIFOLS ASIA PACIFIC PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J06BA02 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Indications
Adjuvant therapy of severe bacterial infections additional to antibiotic therapy.
Product is only intended for use in high risk patients with proven bacterial sepsis and who are unlikely to have underlying IgA deficiency.
Immunoglobulin substitution in immunocompromised patients.
Dosing
Dosage instructions and method of administration
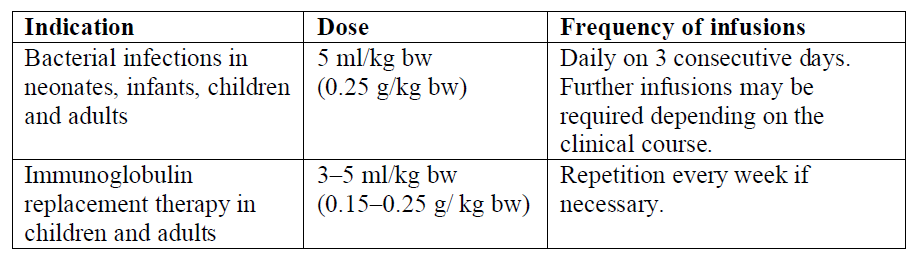
The dosage is dependent on the patient's immune status and on the severity of the disease. Dose based on body weight may require adjustment in underweight or overweight patients. The following dosage suggestions may be used as reference:
Bacterial infections
Neonates, infants, children and adults:
The recommended dose is 5 ml (0.25 g)/kg body weight (bw) daily on 3 consecutive days.
Further infusions may be required depending on the clinical course.
Immunoglobulin replacement therapy
Children and adults:
The recommended dose is 3 – 5 ml (0.15 – 0.25 g)/kg body weight (bw). Repetition at weekly intervals, if necessary.
The dosage recommendations are summarized in the following:
Hepatic impairment
No evidence is available to require a dose adjustment.
Renal impairment
No dose adjustment unless clinically warranted, see section ‘Special warnings and precautions for use’ – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Elderly
No dose adjustment unless clinically warranted, see section ‘Special warnings and precautions for use’ – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
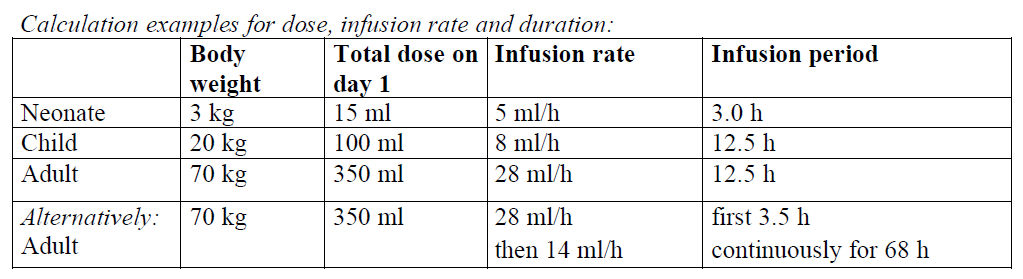
Method of administration
Intravenous use.
Pentaglobin should only be mixed with normal saline.
Pentaglobin should be brought to room or body temperature before use and should be infused intravenously at the following infusion rates:

In case of adverse reaction, either the rate of administration must be reduced or the infusion stopped.
Contraindications
Contraindications
Hypersensitivity to the active substance (human immunoglobulins) or to any of the excipients listed above – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Patients with selective IgA deficiency who developed antibodies to IgA, as administering an IgA-containing product can result in anaphylaxis
