TAZPEN FOR INJECTION 4.5G/VIAL [SIN13758P]
Active ingredients: TAZPEN FOR INJECTION 4.5G/VIAL
Product Info
TAZPEN FOR INJECTION 4.5G/VIAL
[SIN13758P]
Product information
Active Ingredient and Strength | PIPERACILLIN SODIUM EQUIVALENT TO PIPERACILLIN - 4 G |
Dosage Form | INJECTION, POWDER, FOR SOLUTION |
Manufacturer and Country | SHANDONG ANXIN PHARMACEUTICAL COMPANY LTD. (DRUG PRODUCT INTERMEDIATE- STERILE MIXTURE) - CHINA |
Registration Number | SIN13758P |
Licence Holder | VIATRIS PRIVATE LIMITED |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J01CR05 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
INDICATIONS:
Tazpen is indicated for the treatment of systemic and/or local bacterial infections caused by susceptible organisms (detected/suspected) in the conditions listed below:
Lower respiratory tract infections
Urinary tract infections (complicated and uncomplicated)
Intra-abdominal infections
Skin and skin structure infections
Bacterial septicaemia
Polymicrobic infections including those where aerobic and anaerobic organisms are suspected (intra-abdominal, skin and skin structure, lower respiratory tract)
Bacterial infections in neutropenic adults or children (in combination with an aminoglycoside).
Antimicrobial therapy should be adjusted, if appropriate, once the results of culture(s) and antimicrobial susceptibility testing are known.
Dosing
DOSAGE:
Dosage:
Neutropenic patients with signs of infection (e.g. fever) should receive immediate empirical antibiotic therapy before laboratory results are available.
Adults and children 12 years and older with Normal Renal Function: Usual dose: 4.5g given every eight hours. The total daily dose depends on the severity and localization of the infection and can vary from 2.25g to 4.5g administered every six or eight hours.
Neutropenia: Recommended dose: 4.5g given every six hours in combination with an aminoglycoside.
Children under the age of 12 years:
Recommended only for the treatment of children with neutropenia. Tazpen should not be used in children who do not have neutropenia.
Children weighing more than 50 kg: Follow adult dosing guidance, including the aminoglycoside.
Children with Normal Renal Function and weighing less than 50 kg: Adjust dose to 90 mg/kg (80mg piperacillin/10mg tazobactam) given every 6 hours in combination with an aminoglycoside.
Hospitalized children with intra-abdominal infection: For children aged 2 to 12 years, weighing up to 40kg, and with normal renal function, the recommended dosage is 112.5mg/kg (100mg piperacillin/12.5mg tazobactam) every 8 hours. For children aged 2 to 12 years, weighing over 40kg, and normal renal function, follow the adult dose guidance, i.e. 4.5g (4g piperacillin/0.5g tazobactam) every 8 hours. The duration of therapy should be guided by the severity of the infection and the patient’s clinical and bacteriological progress.
Elderly: Recommended dose the same as adults except in cases of renal impairment.
Renal insufficiency:
Adults and Children weighing more than 50 kg: The intravenous dose should be adjusted to the degree of actual renal function impairment. The suggested daily doses are as follows in Table 1:
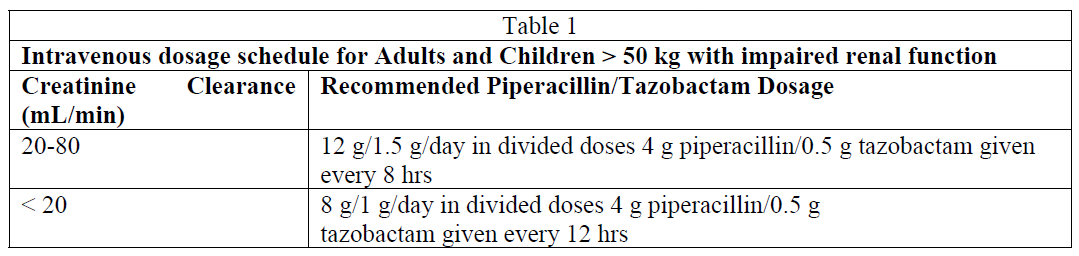
For patients on haemodialysis, the maximum daily dose is 8 g/1 g/day Piperacillin and Tazobactam. In addition, because haemodialysis removes 30%–50% of piperacillin in 4 hours, one additional dose of 2 g piperacillin/0.25 g tazobactam should be administered following each dialysis period. For patients with renal failure and hepatic insufficiency, measurement of serum levels of Tazpen will provide additional guidance for adjusting dosage.
Adults and Children weighing less than 50 kg: The intravenous dosage should be adjusted to the degree of actual renal impairment as in Table 2:

Children weighing less than 50 kg on haemodialysis: Recommended dose is 45mg/kg every 8 hours.
The pharmacokinetics of piperacillin and tazobactam have not been studied in paediatric patients with renal impairment. Each patient must be monitored closely for signs of drug toxicity. Drug dose and interval dose should be adjusted accordingly. In patients with renal insufficiency or hemodialysis patients, intravenous dosage and administration intervals should be adjusted to the degree of renal function impairment.
Duration of therapy: Therapy is recommended to be a minimum of 5 days and maximum of 14 days, considering that dose administration should continue at least 48 hours after the resolution of clinical signs and symptoms or fever.
Administration: Tazpen may be given by slow intravenous injection (3–5 minutes) or by infusion (20–30 minutes).
Directions for Reconstitution and Dilution: Diluents for reconstitution:
0.9% (9 mg/ml) sodium chloride solution for injection
Sterile water for injections
Dextrose 5%
IV injection: Reconstitute each vial with the volume of diluent shown in the table below, using one of the above diluents. Shake until dissolved.

IV infusion: Reconstitute each vial with at least 20 mL using one of the reconstitution diluents above. Shake until dissolved. The reconstituted solution may be further diluted (recommended volume per dose of 50–150 mL) with a compatible IV solution listed as follows: 0.9% Sodium Chloride for Injection; Dextrose 5%.
Administer by infusion over a period of at least 30 min. During the infusion, it is desirable to discontinue the primary infusion solution.
Contraindications
CONTRAINDICATIONS:
The use of Tazpen is contraindicated in patients with a history of allergic reactions to any of the penicillins and/or cephalosporins or β-lactamase inhibitors.
