CEFEPIME-AFT POWDER FOR INJECTION 1G/VIAL [SIN14658P]
Active ingredients: CEFEPIME-AFT POWDER FOR INJECTION 1G/VIAL
Product Info
CEFEPIME-AFT POWDER FOR INJECTION 1G/VIAL
[SIN14658P]
Product information
Active Ingredient and Strength | CEFEPIME HYDROCHLORIDE 1,189 MG EQV CEFEPIME BASE - 1,000 MG |
Dosage Form | INJECTION, POWDER, LYOPHILIZED, FOR SOLUTION |
Manufacturer and Country | QILU PHARMACEUTICAL CO., LTD - CHINA |
Registration Number | SIN14658P |
Licence Holder | APEX PHARMA MARKETING PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J01DE01 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
INDICATIONS
Adults:
Cefepime-AFT is indicated in the treatment of the infections listed below when caused by susceptible bacteria.
Lower respiratory tract infections, including pneumonia and bronchitis.
Urinary tract infections, both complicated, including pyelonephritis, and uncomplicated infections.
Skin and skin structure infections.
Intra-abdominal infections, including peritonitis and biliary tract infections.
Gynaecological infections.
Septicaemia
Empiric treatment in febrile neutropenic patients (See PRECAUTIONS – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information)
Cefepime-AFT is also indicated for surgical prophylaxis in patients undergoing intra-abdominal surgery. In this indication it is essential that metronidazole also be administered.
Paediatrics:
Cefepime-AFT is indicated in paediatric patients over 2 months of age for the treatment of the infections listed below when caused by susceptible bacteria:
Pneumonia
Urinary tract infections, both complicated, including pyelonephritis, and uncomplicated infections
Skin and skin structure infections
Septicaemia
Empiric treatment in febrile neutropenic patients (See PRECAUTIONS – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information)
Culture and susceptibility studies should be performed when appropriate to determine susceptibility of the causative organism(s) to cefepime. Empiric therapy with Cefepime-AFT may be instituted before results of susceptibility studies are known; however, once these results become available, the antibiotic treatment should be adjusted accordingly.
Because of its broad spectrum of bactericidal activity against gram-positive and gram-negative bacteria, Cefepime-AFT can be used appropriately as monotherapy prior to identification of the causative organisms(s). In patients who are at risk of mixed aerobic-anaerobic infection, particularly if bacteria not susceptible to cefepime may be present, concurrent initial therapy with anti-anaerobic agent is recommended before the causative organism(s) is(are) known.
Dosing
DOSAGE AND ADMINISTRATION
Cefepime-AFT can be administered either intravenously or intramuscularly every 12 hours. The dosage and route vary according to the susceptibility of the causative organisms, the severity of the infection, renal function and overall condition of the patient.
Adults
Guidelines for dosage of Cefepime-AFT for adults are provided in Table 5.
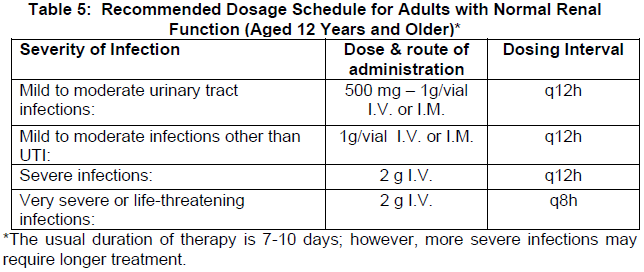
Surgical Prophylaxis
The dose recommendation for prophylaxis to prevent infection in adults undergoing intra-abdominal surgery is as follows:
A single 2 g I.V. dose of cefepime (as a 30-minute infusion, see DOSAGE AND ADMINISTRATION) starting 60 minutes before initial surgical incision. A single 500 mg I.V. dose of metronidazole should be administered immediately following completion of the cefepime infusion. The metronidazole dose should be prepared and administered in accordance with official product labelling. Due to incompatibility, cefepime and metronidazole should not be mixed together in the same container (see COMPATIBILITY AND STABILITY – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information); flushing of the intravenous line with a compatible fluid before infusion of the metronidazole is recommended.
If the surgical procedure lasts longer than 12 hours from the initial prophylactic dose, a second dose of cefepime followed by metronidazole should be administered 12 hours following the initial prophylactic dose.
Paediatrics (aged 2 months up to 12 years with normal renal function)
Usual recommended dosages:
Pneumonia, urinary tract infections, and skin and skin structure infections: Patients > 2 months of age with body weight ≤ 40 kg: 50 mg/kg q12h for 10 days. For more severe infections, a dosage schedule of q8h can be used.
Septicemia and Empiric treatment of febrile neutropenia: Patients > 2 months of age with body weight ≤ 40 kg: 50 mg/kg q8h for 7–10 days.
Experience with the use of cefepime in pediatric patients < 2 months of age is limited. While this experience has been attained using the 50 mg/kg dose, modeling of pharmacokinetic data obtained in patients > 2 months of age suggests that a dosage of 30 mg/kg q12h or q8h may be considered for patients aged 1 month up to 2 months. Both the 50 mg/kg dose for patients > 2 months of age and the 30mg/kg dose for patients between 1 and 2 months of age are comparable to a 2 g adult dose. Administration of cefepime in these patients should be carefully monitored.
For pediatric patients with body weights > 40kg, adult dosing recommendations apply (see Table 5). For patients older than 12 years who are ≤ 40 kg, the dosage recommendations for younger patients ≤ 40 kg should be used. Dosage in pediatric patients should not exceed the maximum recommended dosage in adults (2 g q8h). Experience with intramuscular administration in pediatric patients is limited.
Patients with Impaired Renal Function
In patients with impaired renal function, the dose of cefepime should be adjusted to compensate for the slower rate of renal elimination.
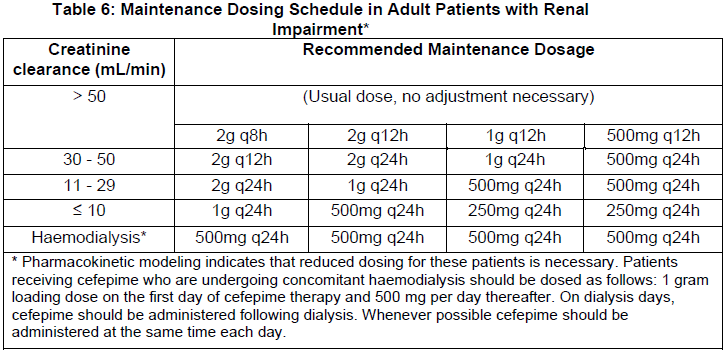
The recommended initial dose of cefepime in patients with mild to moderate renal impairment should be the same as in patients with normal renal function. The recommended maintenance dosages of cefepime in adult patients with renal insufficiency are presented in Table 6.
When only a serum creatinine measurement is available, the following formula (Cockcroft and Gault equation) may be used to estimate creatinine clearance. The serum creatinine should represent a steady state of renal function:
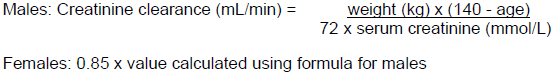
Dialysis Patients
In patients undergoing haemodialysis, approximately 68% of the total amount of cefepime present in the body at the start of dialysis will be removed during a 3 hour dialysis period. In patients undergoing continuous ambulatory peritoneal dialysis, cefepime may be administered at normally recommended doses, i.e.: 500 mg, 1 g or 2 g, depending on infection severity, but at a dosage interval of every 48 hours.
Children with Impaired Renal Function
Since urinary excretion is the primary route of elimination of cefepime in paediatric patients (see PHARMACOLOGY – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information), an adjustment of the dosage of cefepime should also be considered in patients < 12 years of age with renal impairment.
A dose of 50 mg/kg in patients aged 2 months up to 12 years, and a dose of 30 mg/kg in patients aged 1 month up to 2 months, are comparable to a dose of 2g in an adult. As recommended in Table 6 above, the same increase in interval between doses and/or reduction in dose should be used. When only serum creatinine is available, creatinine clearance may be estimated using either of the following methods:
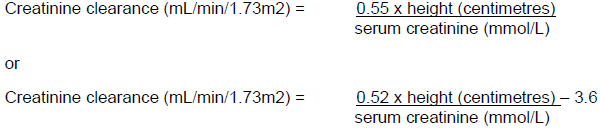
Hepatic Impairment
No dosage adjustment is necessary for patients with impaired hepatic function
Contraindications
CONTRAINDICATIONS
Cefepime is contraindicated in patients who have shown immediate hypersensitivity reactions to any component of the formulation (including L-arginine), the cephalosporin class of antibiotics, penicillins or other beta-lactam antibiotics.
