CEFTAZIDIME MEVON POWDER FOR SOLUTION FOR INJECTION OR INFUSION 1G/ VIAL [SIN14738P]
Active ingredients: CEFTAZIDIME MEVON POWDER FOR SOLUTION FOR INJECTION OR INFUSION 1G/ VIAL
Product Info
CEFTAZIDIME MEVON POWDER FOR SOLUTION FOR INJECTION OR INFUSION 1G/ VIAL
[SIN14738P]
Product information
Active Ingredient and Strength | CEFTAZIDIME PENTAHYDRATE 1.164 G EQUIVALENT TO CEFTAZIDIME - 1 G/VIAL |
Dosage Form | INJECTION, POWDER, FOR SOLUTION |
Manufacturer and Country | SINOPHARM ZHIJUN (SHENZHEN) PHARMACEUTICAL CO., LTD - CHINA |
Registration Number | SIN14738P |
Licence Holder | NOVEM PHARMA PRIVATE LIMITED |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J01DD02 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Treatment of single or multiple infections caused by susceptible organisms.
May be used alone as first choice drug before the results of sensitivity tests are available.
May be used in combination with an aminoglycoside or most other beta-lactam antibiotics.
May be used with an antibiotic against anaerobes when the presence of Bacteroides fragilis is suspected.
Indications include:
Table caption
– severe infections e.g. | – septicaemia, bacteraemia, peritonitis, meningitis. |
– respiratory tract infections including lung infections in cystic fibrosis
– ear, nose and throat infections
– urinary tract infections
– skin and soft tissue infections
– gastrointestinal, biliary and abdominal infections
– bone and joint infections
– infections associated with haemo- and peritoneal dialysis and with continuous ambulatory peritoneal dialysis (CAPD)
– prophylaxis: prostatic surgery (transurethral resection).
Dosing
4.2 Posology and method of administration
Dosage depends upon the severity, sensitivity, site and type of infection and upon the age and renal function of the patient.
Use CEFTAZIDIME MEVON injection by intravenous (i.v.) or by deep intramuscular (i.m.) injection. Recommended i.m. injection sites are the upper outer quadrant of the gluteus maximus or lateral part of the thigh.
CEFTAZIDIME MEVON solutions may be given directly into the vein or introduced into the tubing of a giving set if the patient is receiving parenteral fluids.
• Adults
1 to 6 g/day in two or three divided doses by i.v. or i.m. injection.
Urinary tract and less severe infections: – 500 mg or 1 g every 12 hours.
Most infections: – 1 g every 8 hours or 2 g every 12 hours.
Very severe infections particularly in immunocompromised patients including those with neutropenia: – 2 g every eight or 12 hours.
Fibrocystic adults with pseudomonal lung infections: - 100 to 150 mg/kg/day in three divided doses.
In adults with normal renal function 9 g/day has been used without ill effect.
When used as a prophylactic agent in prostatic surgery, 1 g should be given at the induction of anaesthesia. A second dose should be considered at the time of catheter removal.
• Infants and children (greater than 2 months)
30 to 100 mg/kg/day in two or three divided doses.
Doses up to 150 mg/kg/day (maximum 6 g/day) in three divided doses may be given to infected immunocompromised or fibrocystic children or children with meningitis.
• Neonates (0 to 2 months)
25 to 60 mg/kg/day in two divided doses.
In neonates, the serum half-life of ceftazidime can be three to four times that in adults.
• Elderly
In view of the reduced clearance of ceftazidime in acutely ill elderly patients, the daily dosage should not normally exceed 3 g, especially in those over 80 years of age.
• Renal Impairment
Ceftazidime is excreted unchanged by the kidneys. Therefore, in patients with impaired renal function, the dosage should be reduced.
An initial loading dose of 1 g should be given. Maintenance doses should be based on creatinine clearance as shown in Table 1:
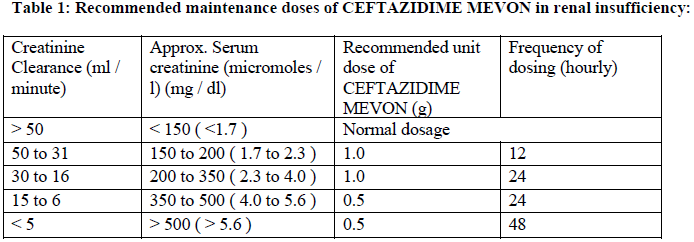
In patients with severe infections the unit dose should be increased by 50% or the dosing frequency increased. In such patients the ceftazidime serum levels should be monitored and trough levels should not exceed 40 mg/l.
In children the creatinine clearance should be adjusted for body surface area or lean body mass.
Haemodialysis
The serum half-life during haemodialysis ranges from 3 to 5 hours.
Following each haemodialysis period, the maintenance dose of CEFTAZIDIME MEVON recommended in the above table should be repeated.
Peritoneal dialysis
CEFTAZIDIME MEVON may be used in peritoneal dialysis and continuous ambulatory peritoneal dialysis (CAPD).
In addition to i.v. use, CEFTAZIDIME MEVON can be incorporated into the dialysis fluid (usually 125 to 250 mg for 2 litres of dialysis solution).
For patients in renal failure on continuous arteriovenous haemodialysis or high-flux haemofiltration in intensive therapy units; 1 g daily either as a single dose or in divided doses. For low-flux haemofiltration, follow the dosage recommended under impaired renal function.
Contraindications
4.3 Contraindications
Hypersensitivity to the active substance, any other cephalosporin or to any of the excipients, listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
History of severe hypersensitivity (e.g. anaphylactic reaction) to any other type of beta-lactam antibacterial agent (penicillins, monobactams and carbapenems).
