GAMUNEX®-C SOLUTION FOR INFUSION 10% [SIN14928P]
Active ingredients: GAMUNEX®-C SOLUTION FOR INFUSION 10%
Product Info
GAMUNEX®-C SOLUTION FOR INFUSION 10%
[SIN14928P]
Product information
Active Ingredient and Strength | HUMAN NORMAL IMMUNOGLOBULIN - 0.1 G/ML |
Dosage Form | INJECTION, SOLUTION |
Manufacturer and Country | GRIFOLS THERAPEUTICS LLC - UNITED STATES |
Registration Number | SIN14928P |
Licence Holder | GRIFOLS ASIA PACIFIC PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J06BA02 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Replacement therapy in adults, children and adolescents (0–18 years) in:
Primary immunodeficiency syndromes (PID) with impaired antibody production
Secondary immunodeficiencies (SID) in patients who suffer from severe or recurrent infections, ineffective antimicrobial treatment and either proven specific antibody failure (PSAF)* or serum IgG level of <4 g/l
*PSAF = failure to mount at least a 2-fold rise in IgG antibody titer to pneumococcal polysaccharide and polypeptide antigen vaccines
Measles pre-/post exposure prophylaxis for susceptible adults, children and adolescents (0–18 years) in whom active immunization is contraindicated or not advised.
Consideration should also be given to official recommendations on intravenous human immunoglobulin use in measles pre-/post exposure prophylaxis and active immunization.
Immunomodulation in adults, children and adolescents (0–18 years) in:
Primary immune thrombocytopenia (ITP), in patients at high risk of bleeding or prior to surgery to correct the platelet count
Guillain Barré syndrome
Kawasaki disease (in conjunction with acetylsalicylic acid; see 4.2)
Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP)
Multifocal motor neuropathy (MMN)
Immunomodulation in adults aged ≥18 years in:
Severe acute exacerbations of myasthenia gravis
Dosing
4.2 Posology and method of administration
IVIg therapy should be initiated and monitored under the supervision of a physician experienced in the treatment of immune system disorders.
Posology
The dose and dose regimen are dependent on the indication.
The dose may need to be individualized for each patient dependent on the clinical response. Dose based on body weight may require adjustment in underweight or overweight patients.
The following dose regimens are given as a guidance.
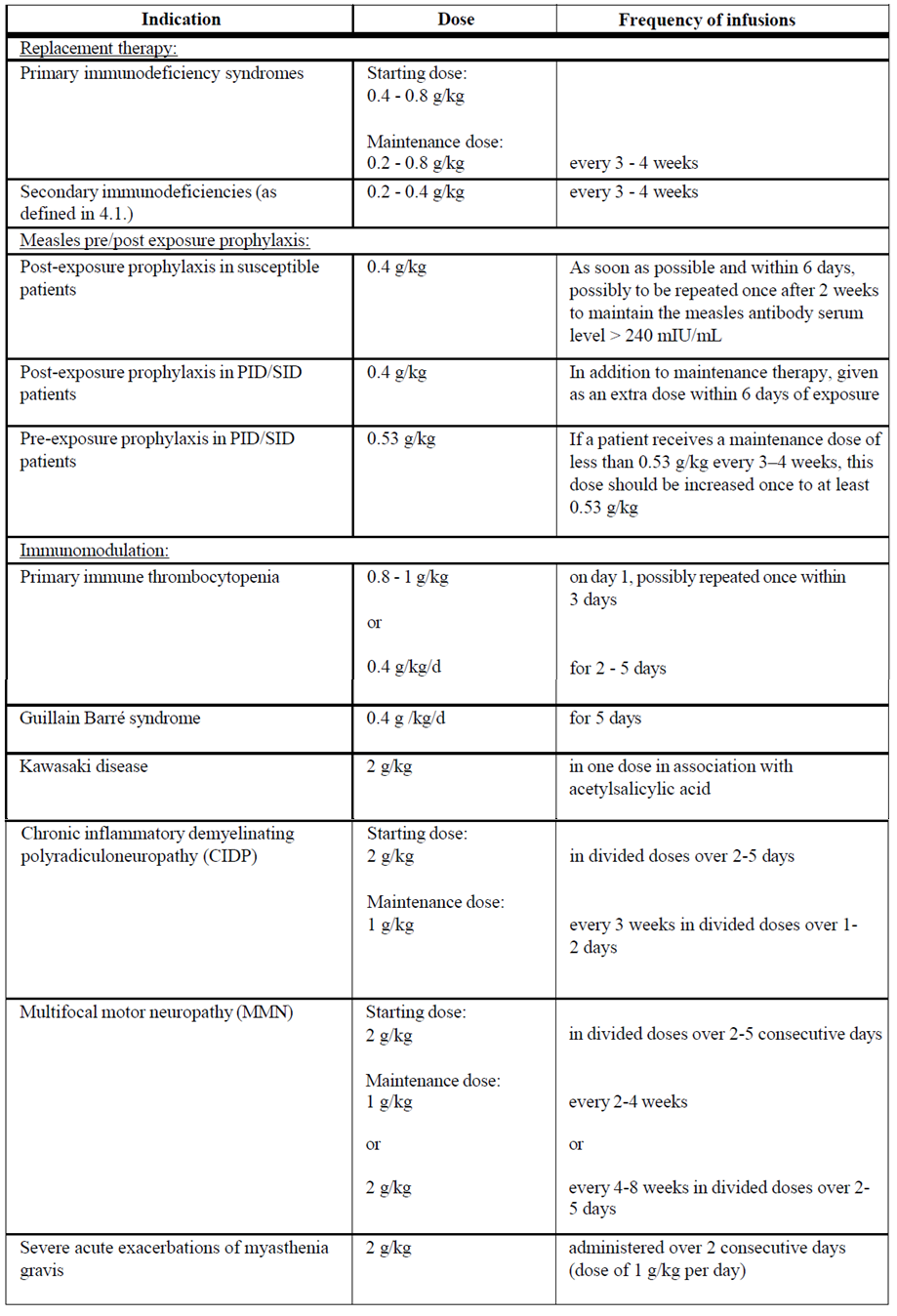
Replacement therapy in primary immunodeficiency syndromes
The dose regimen should achieve a trough level of IgG (measured before the next infusion) of at least 6 g/L or within the normal reference range for the population age. 3–6 months are required after the initiation of therapy for equilibration (steady-state IgG levels) to occur. The recommended starting dose is 0.4–0.8 g/kg given once followed by at least 0.2 g/kg given every 3–4 weeks.
The dose required to achieve a trough level of IgG of 6 g/L is of the order of 0.2–0.8 g/kg/month. The dosage interval when steady state has been reached varies from 3–4 weeks.
IgG trough levels should be measured and assessed in conjunction with the incidence of infection. To reduce the rate of bacterial infections, it may be necessary to increase the dosage and aim for higher trough levels.
Replacement therapy in secondary immunodeficiencies (as defined in 4.1.)
The recommended dose is 0.2–0.4 g/kg every 3–4 weeks.
IgG trough levels should be measured and assessed in conjunction with the incidence of infection. Dose should be adjusted as necessary to achieve optimal protection against infections, an increase may be necessary in patients with persisting infection; a dose decrease can be considered when the patient remains infection free.
Measles pre-/post exposure prophylaxis
Post-exposure prophylaxis
If a susceptible patient has been exposed to measles, a dose of 0.4 g/kg given as soon as possible and within 6 days of exposure should provide a serum level > 240 milli-international units/mL of measles antibodies for at least 2 weeks. Serum levels should be checked after 2 weeks and documented. A further dose of 0.4 g/kg possibly to be repeated once after 2 weeks may be necessary to maintain the serum level > 240 milli-international units/mL.
If a PID/SID patient has been exposed to measles and regularly receives IVIg infusions, it should be considered to administer an extra dose of IVIg as soon as possible and within 6 days of exposure. A dose of 0.4 g/kg should provide a serum level > 240 milli-international units/mL of measles antibodies for at least 2 weeks.
Pre-exposure prophylaxis
If a PID/SID patient is at risk of future measles exposure and receives an IVIg maintenance dose of less than 0.53 g/kg every 3–4 weeks, this dose should be increased once to 0.53 g/kg. This should provide a serum level of > 240 milli-international units/mL of measles antibodies for at least 22 days after infusion.
Immunomodulation in:
Primary immune thrombocytopenia
There are two alternative treatment schedules:
0.8–1 g/kg given on day 1; this dose may be repeated once within 3 days
0.4 g/kg given daily for 2–5 days. The treatment can be repeated, if relapse occurs.
Guillain Barré syndrome
0.4 g/kg/day over 5 days (possible repeat of dosing in case of relapse).
Kawasaki disease
2.0 g/kg should be administered as a single dose. Patients should receive concomitant treatment with acetylsalicylic acid.
Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP)
Starting dose: 2 g/kg divided over 2–5 consecutive days
Maintenance doses:
1 g/kg divided over 1–2 consecutive days every 3 weeks.
The treatment effect should be evaluated after each cycle; if no treatment effect is seen after 6 months, the treatment should be discontinued.
If the treatment is effective, long term treatment should be subject to the physician’s discretion based upon the patient response and maintenance response. The dosing and intervals may have to be adapted according to the individual course of the disease.
Multifocal motor neuropathy (MMN)
Starting dose: 2 g/kg divided over 2–5 consecutive days.
Maintenance dose: 1 g/kg every 2–4 weeks or 2 g/kg every 4–8 weeks.
The treatment effect should be evaluated after each cycle; if no treatment effect is seen after 6 months, the treatment should be discontinued.
If the treatment is effective, long term treatment should be subject to the physician’s discretion based upon the patient response and maintenance response. The dosing and intervals may have to be adapted according to the individual course of the disease.
Severe acute exacerbations of myasthenia gravis
2 g/kg divided over 2 consecutive days (dose of 1 g/kg per day).
If the desired clinical response is not observed, consideration should be given to alternative modes of therapy at the doctor’s discretion in consultation with physicians with expertise in the management of neurological disorders/severe acute exacerbations of myasthenia gravis.
Clinical studies of Gamunex-C did not include sufficient numbers of subjects aged 65 and over to determine a precise treatment effect.
The dosage recommendations are summarized in the following table:
Pediatric population
The posology in children and adolescents (0–18 years) is not different to that of adults as the posology for each indication is given by body weight and must be adjusted to the clinical outcome of the above-mentioned conditions.
Hepatic impairment
No evidence is available to require a dose adjustment.
Renal impairment
No dose adjustment unless clinically warranted, see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Elderly
No dose adjustment unless clinically warranted, see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Method of administration
For intravenous use.
Human normal immunoglobulin should be infused intravenously at an initial rate of 0.6 – 1.2 mL/kg/hr for 0.5 hr. See section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information. In case of adverse reaction, either the rate of administration must be reduced or the infusion stopped. If well tolerated, the rate of administration may gradually be increased to a maximum of 4.8 – 8.4 mL/kg/hr.
Contraindications
4.3 Contraindications
Hypersensitivity to the active substance (human immunoglobulins) or to any of the excipients (see sections 4.4 and 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Patients with selective IgA deficiency who developed antibodies to IgA, as administering an IgA-containing product can result in anaphylaxis.
