PEGASET 75 CAPSULES 75MG [SIN16402P]
Active ingredients: PEGASET 75 CAPSULES 75MG
Product Info
PEGASET 75 CAPSULES 75MG
[SIN16402P]
Product information
Active Ingredient and Strength | PREGABALIN - 75 MG |
Dosage Form | CAPSULE |
Manufacturer and Country | AUROBINDO PHARMA LIMITED - UNIT VII - INDIA |
Registration Number | SIN16402P |
Licence Holder | APOTHECA MARKETING PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | N03AX16 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Therapeutic indications
Neuropathic pain
Pregabalin Capsules are indicated for the treatment of neuropathic pain, which includes diabetic peripheral neuropathy and post-herpetic neuralgia, in adults.
Epilepsy
Pregabalin Capsules are indicated as adjunctive therapy in adults with partial seizures with or without secondary generalization.
Generalised Anxiety Disorder
Pregabalin Capsules are indicated for the treatment of Generalised Anxiety Disorder (GAD) in adults.
Dosing
Posology and method of administration
Posology
The dose range is 150 to 600 mg per day given in either two or three divided doses.
Neuropathic pain
Pregabalin treatment can be started at a dose of 150 mg per day given as two or three divided doses. Based on individual patient response and tolerability, the dose may be increased to 300 mg per day after an interval of 3 to 7 days, and if needed, to a maximum dose of 600 mg per day after an additional 7-day interval.
Epilepsy
Pregabalin treatment can be started with a dose of 150 mg per day given as two or three divided doses. Based on individual patient response and tolerability, the dose may be increased to 300 mg per day after 1 week. The maximum dose of 600 mg per day may be achieved after an additional week.
Generalised Anxiety Disorder
The dose range is 150 to 600 mg per day given as two or three divided doses. The need for treatment should be reassessed regularly.
Pregabalin treatment can be started with a dose of 150 mg per day. Based on individual patient response and tolerability, the dose may be increased to 300 mg per day after 1 week. Following an additional week the dose may be increased to 450 mg per day. The maximum dose of 600 mg per day may be achieved after an additional week.
Discontinuation of Pregabalin
In accordance with current clinical practice, if Pregabalin has to be discontinued it is recommended this should be done gradually over a minimum of 1 week independent of the indication.
Patients with renal impairment
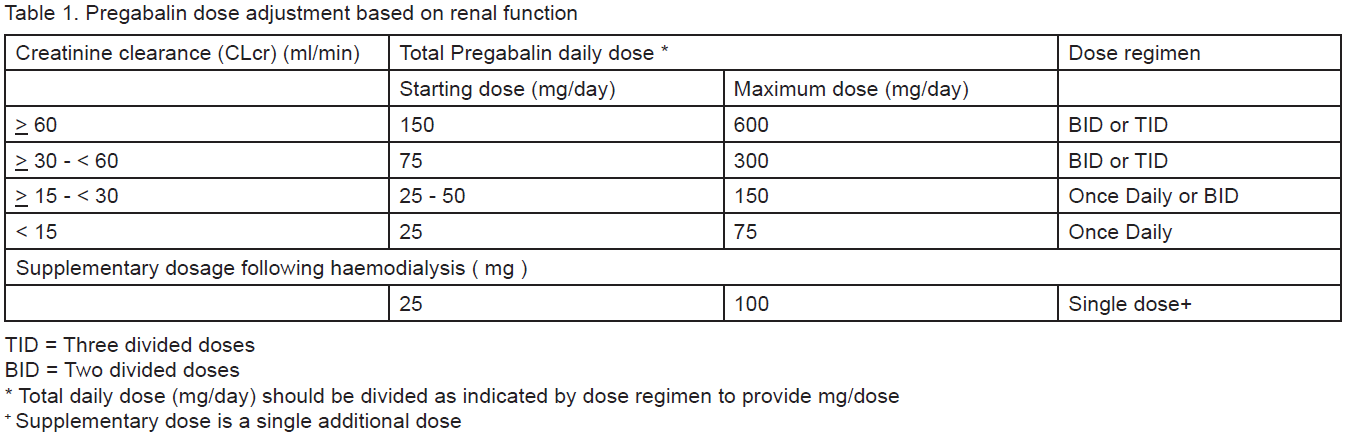
Pregabalin is eliminated from the systemic circulation primarily by renal excretion as unchanged drug. As Pregabalin clearance is directly proportional to creatinine clearance, dose reduction in patients with compromised renal function must be individualised according to creatinine clearance (CLcr), as indicated in Table 1 determined using the following formula:
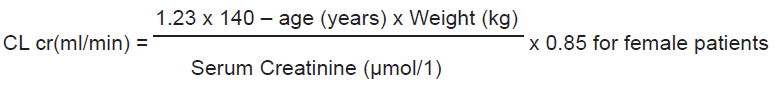
Pregabalin is removed effectively from plasma by haemodialysis (50% of drug in 4 hours). For patients receiving haemodialysis, the Pregabalin daily dose should be adjusted based on renal function. In addition to the daily dose, a supplementary dose should be given immediately following every 4-hour haemodialysis treatment (see Table 1).
Patients with hepatic impairment
No dose adjustment is required for patients with hepatic impairment.
Paediatric population
The safety and efficacy of Pregabalin in children below the age of 12 years and in adolescents (12–17 years of age) have not been established. No data is available. The use in children and adolescents is not recommended.
Elderly (over 65 years of age) population
Elderly patients may require a dose reduction of Pregabalin due to a decreased renal function (see patients with renal impairment).
Method of administration
Pregabalin may be taken with or without food.
Pregabalin is for oral use only.
Contraindications
Contraindications
Pregabalin Capsules are contraindicated in patients with hypersensitivity to Pregabalin or to any of the Excipients.
