RYBREVANT CONCENTRATE FOR SOLUTION FOR INFUSION 350MG/7ML [SIN16548P]
Active ingredients: RYBREVANT CONCENTRATE FOR SOLUTION FOR INFUSION 350MG/7ML
Product Info
RYBREVANT CONCENTRATE FOR SOLUTION FOR INFUSION 350MG/7ML
[SIN16548P]
Product information
Active Ingredient and Strength | AMIVANTAMAB - 350 MG/7 ML |
Dosage Form | INFUSION, SOLUTION CONCENTRATE |
Manufacturer and Country | CILAG AG - SWITZERLAND |
Registration Number | SIN16548P |
Licence Holder | JOHNSON & JOHNSON INTERNATIONAL (SINGAPORE) PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01FX18 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Indications
RYBREVANT® is indicated:
in combination with lazertinib for the first-line treatment of adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) exon 19 deletions or exon 21 L858R substitution mutations.
in combination with carboplatin and pemetrexed for the first-line treatment of adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) with activating epidermal-growth factor receptor (EGFR) exon 20 insertion mutations.
in combination with carboplatin and pemetrexed for the treatment of adult patients with locally advanced or metastatic NSCLC with EGFR exon 19 deletions or exon 21 L858R substitution mutations, whose disease has progressed on or after treatment with an EGFR tyrosine kinase inhibitor.
as monotherapy for the treatment of adult patients with locally advanced or metastatic NSCLC with activating EGFR exon 20 insertion mutations whose disease has progressed on or after platinum-based chemotherapy.
Dosing
Dosage and Administration
RYBREVANT® should be administered by a healthcare professional with appropriate medical support to manage infusion-related reactions (IRRs) if they occur (see Warnings and Precautions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Administer pre-infusion medications (see Dosage and Administration – Pre-infusion medications).
Administer diluted RYBREVANT® intravenously according to the infusion rates in Tables 5 and 6, with the initial dose as a split infusion on Week 1 on Day 1 and Day 2.
When considering the use of RYBREVANT®, EGFR exon 19 deletion, exon 21 L858R substitution, or exon 20 insertion mutation presence should be established using a validated test (see Pharmacodynamic Effects – Clinical studies – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
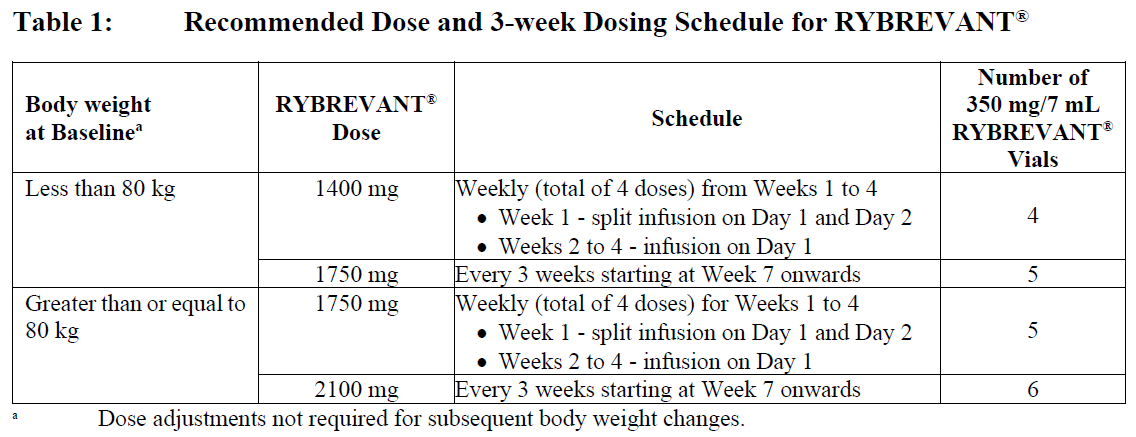
Dosage – adults (≥18 years)
Every 3 Weeks
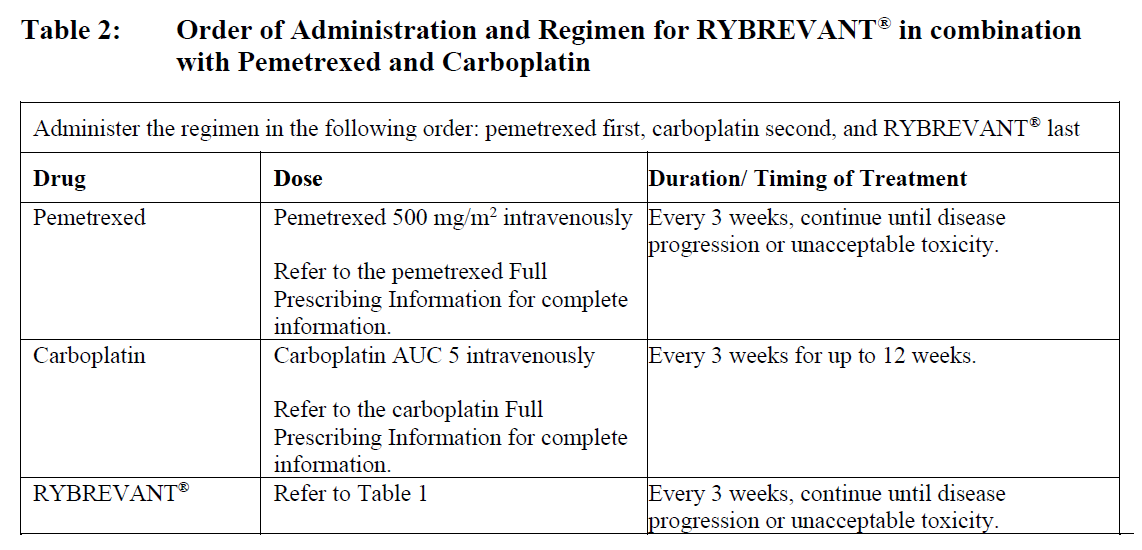
The recommended dosage of RYBREVANT®, when used in combination with carboplatin and pemetrexed, is provided in Table 1 (see Infusion Rates – Table 5).
When used in combination with carboplatin and pemetrexed, RYBREVANT® should be administered after carboplatin and pemetrexed in the following order: pemetrexed, carboplatin and then RYBREVANT®. See Pharmacodynamic Effects – Clinical studies – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information and the manufacturer’s prescribing information for dosing instructions for carboplatin and pemetrexed.
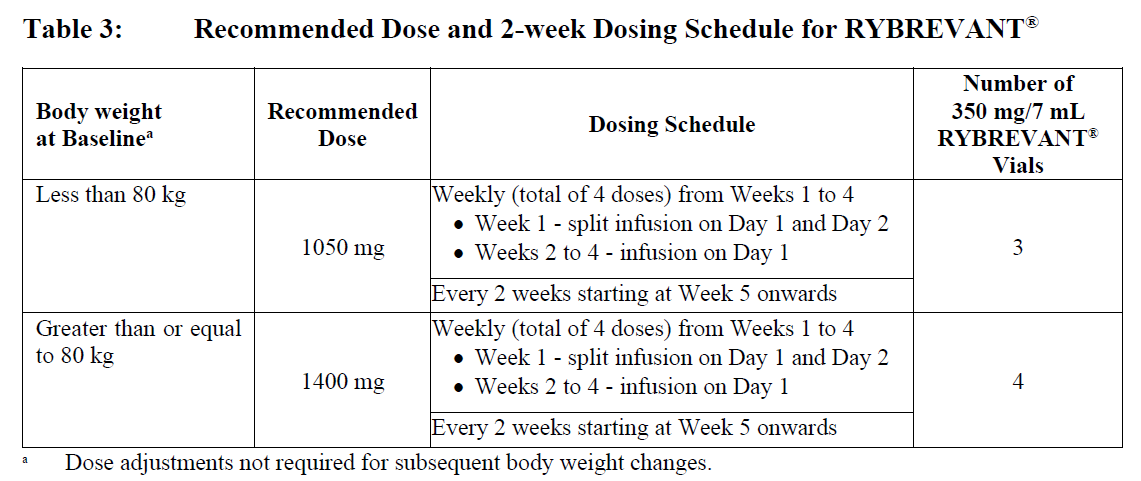
Every 2 Weeks
The recommended dosage of RYBREVANT® monotherapy or in combination with lazertinib is provided in Table 3 (see Infusion Rates – Table 6).
When used in combination with lazertinib, RYBREVANT® should be administered anytime after lazertinib when given on the same day. See Pharmacodynamic Effects – Clinical studies – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information and the manufacturer’s prescribing information for dosing instructions for lazertinib.
Duration of treatment
It is recommended that patients are treated with RYBREVANT® until unacceptable toxicity or disease progression.
Pre-infusion medications
Two days before the first infusion:
Oral dexamethasone
During the two days prior to the initial RYBREVANT® infusion, patients should receive 8 mg dexamethasone orally, twice daily. On the day of the initial infusion (Week 1, Day 1), patients should receive 8 mg dexamethasone orally, one hour prior to infusion in addition to intravenous dexamethasone to further reduce the risk of IRR.
Day of infusion:
Prior to initial infusion of RYBREVANT® (Week 1, Days 1 and 2), administer antihistamines, antipyretics, and glucocorticoids to reduce the risk of IRRs. For subsequent doses, administer antihistamines and antipyretics. Administer antiemetics as needed.

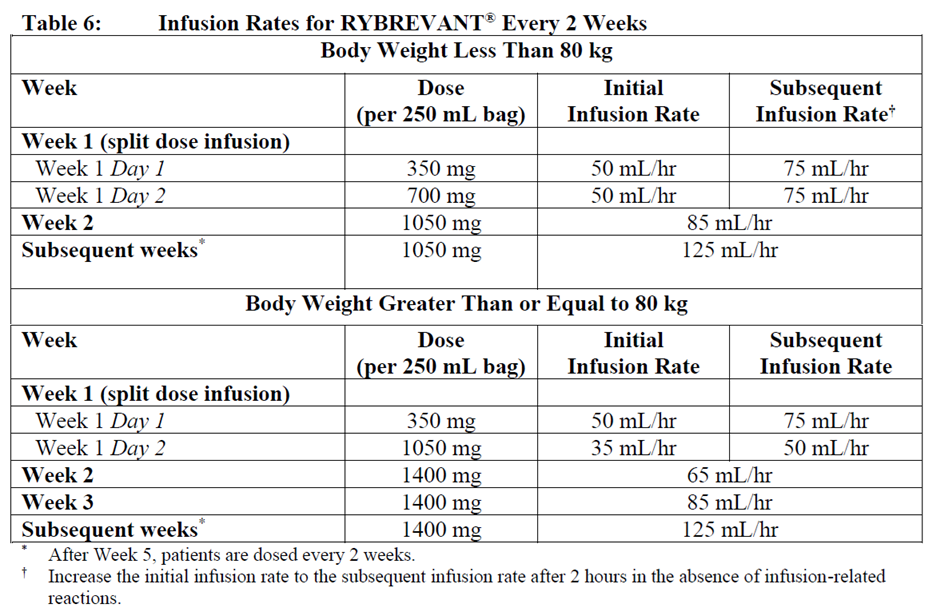
Infusion Rates
Administer RYBREVANT® infusion every 3 weeks intravenously according to the infusion rates in Table 5, and administer RYBREVANT® infusion every 2 weeks intravenously according to the infusion rates in Table 6.
Due to the frequency of IRRs at the first dose, infusion via a peripheral vein at Week 1 and Week 2 should be considered to minimize drug exposure in the event of an IRR; infusion via central line may be administered for subsequent weeks. It is recommended for the first dose to be diluted as close to administration as possible to allow for maximal flexibility in IRR management.

Missed dose(s)
If a planned dose of RYBREVANT® is missed, the dose should be administered as soon as possible and the dosing schedule should be adjusted accordingly, maintaining the treatment interval.
Dose modifications
The recommended dose reductions for adverse reactions (see Table 8) are listed in Table 7.

The recommended dosage modifications for adverse reactions are provided in Table 8.

Special populations
Pediatrics (17 years of age and younger)
The safety and efficacy of RYBREVANT® have not been established in pediatric patients.
Elderly (65 years of age and older)
Of the 1082 patients treated with RYBREVANT® in EDI1001, NSC3001, NSC3002, and NSC3003, 41.6% were 65 years of age or older, and 10.5% were 75 years of age or older. No overall differences in safety or effectiveness were observed between patients ≥ 65 years of age and patients < 65 years of age. No dosage adjustment is necessary (see Pharmacokinetic Properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal impairment
No formal studies of amivantamab in patients with renal impairment have been conducted. Based on population pharmacokinetic (PK) analyses, no dosage adjustment is necessary for patients with mild or moderate renal impairment. No data are available in patients with severe renal impairment (see Pharmacokinetic Properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Hepatic impairment
No formal studies of amivantamab in patients with hepatic impairment have been conducted. Based on population PK analyses, no dosage adjustment is necessary for patients with mild hepatic impairment. No data are available in patients with moderate or severe hepatic impairment (see Pharmacokinetic Properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Administration
Preparation for administration
RYBREVANT® solution must be diluted and prepared for intravenous infusion by a healthcare professional using aseptic technique.
Determine the dose required and number of RYBREVANT® vials needed based on patient’s baseline weight (see Dosage). Each vial of RYBREVANT® contains 350 mg of amivantamab.
Check that the RYBREVANT® solution is colorless to pale yellow. Do not use if discoloration or visible particles are present.
Withdraw and then discard a volume of either 5% dextrose [glucose] solution or 0.9% sodium chloride solution from the 250 mL infusion bag equal to the volume of RYBREVANT® to be added (i.e., discard 7 mL diluent from the infusion bag for each RYBREVANT® vial). Infusion bags must be made of polyvinylchloride (PVC), polypropylene (PP), polyethylene (PE), or polyolefin blend (PP+PE).
Withdraw 7 mL of RYBREVANT® from each vial and add it to the infusion bag. The final volume in the infusion bag should be 250 mL. Each vial contains a 0.5 mL overfill to ensure sufficient extractable volume. Discard any unused portion left in the vial.
Gently invert the bag to mix the solution. Do not shake.
Visually inspect the diluted solution before administration. Do not use if discoloration or visible particles are observed.
Diluted solutions should be administered within 10 hours (including infusion time) at room temperature (15°C to 25°C) and in room light.
Administration
Prior to administration, prime the infusion set with the diluent (either 5% dextrose solution or 0.9% sodium chloride solution).
Administer the diluted solution by intravenous infusion using an infusion set fitted with a flow regulator and with an in-line, sterile, non-pyrogenic, low protein-binding polyethersulfone (PES) filter (pore size 0.2 micrometer). Administration sets must be made of either polyurethane (PU), polybutadiene (PBD), PVC, PP, or PE.
Do not infuse RYBREVANT® concomitantly in the same intravenous line with other agents.
This medicinal product is for single use only. Any unused medicinal product should be disposed of in accordance with local requirements.
Contraindications
Contraindications
Hypersensitivity to the active substance(s) or to any of the excipients listed in List of Excipients – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
