ICUNES® CONCENTRATE FOR SOLUTION FOR INFUSION 100MCG/ML [SIN16592P]
Active ingredients: ICUNES® CONCENTRATE FOR SOLUTION FOR INFUSION 100MCG/ML
Product Info
ICUNES® CONCENTRATE FOR SOLUTION FOR INFUSION 100MCG/ML
[SIN16592P]
Product information
Active Ingredient and Strength | DEXMEDETOMIDINE HCL EQV DEXMEDETOMIDINE - 100 MCG/ML |
Dosage Form | INFUSION, SOLUTION CONCENTRATE |
Manufacturer and Country | PT. NOVELL PHARMACEUTICAL LABORATORIES - INDONESIA REP OF |
Registration Number | SIN16592P |
Licence Holder | NOVEM HEALTHCARE PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | N05CM18 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
1 INDICATIONS AND USAGE
1.1 Intensive Care Unit Sedation
Dexmedetomidine HCl is indicated for sedation of initially intubated and mechanically ventilated patients during treatment in an intensive care setting. Dexmedetomidine HCl should be administered by continuous infusion not to exceed 24 hours.
Dexmedetomidine HCl has been continuously infused in mechanically ventilated patients prior to extubation, during extubation, and post-extubation. It is not necessary to discontinue Dexmedetomidine HCl prior to extubation.
1.2 Procedural Sedation
Dexmedetomidine HCl is indicated for sedation of non-intubated patients prior to and/or during surgical and other procedures.
Dosing
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Guidelines
Dexmedetomidine HCl dosing should be individualized and titrated to desired clinical response.
Dexmedetomidine HCl is not indicated for infusions lasting longer than 24 hours.
Dexmedetomidine HCl should be administered using a controlled infusion device.
2.2 Dosage Information
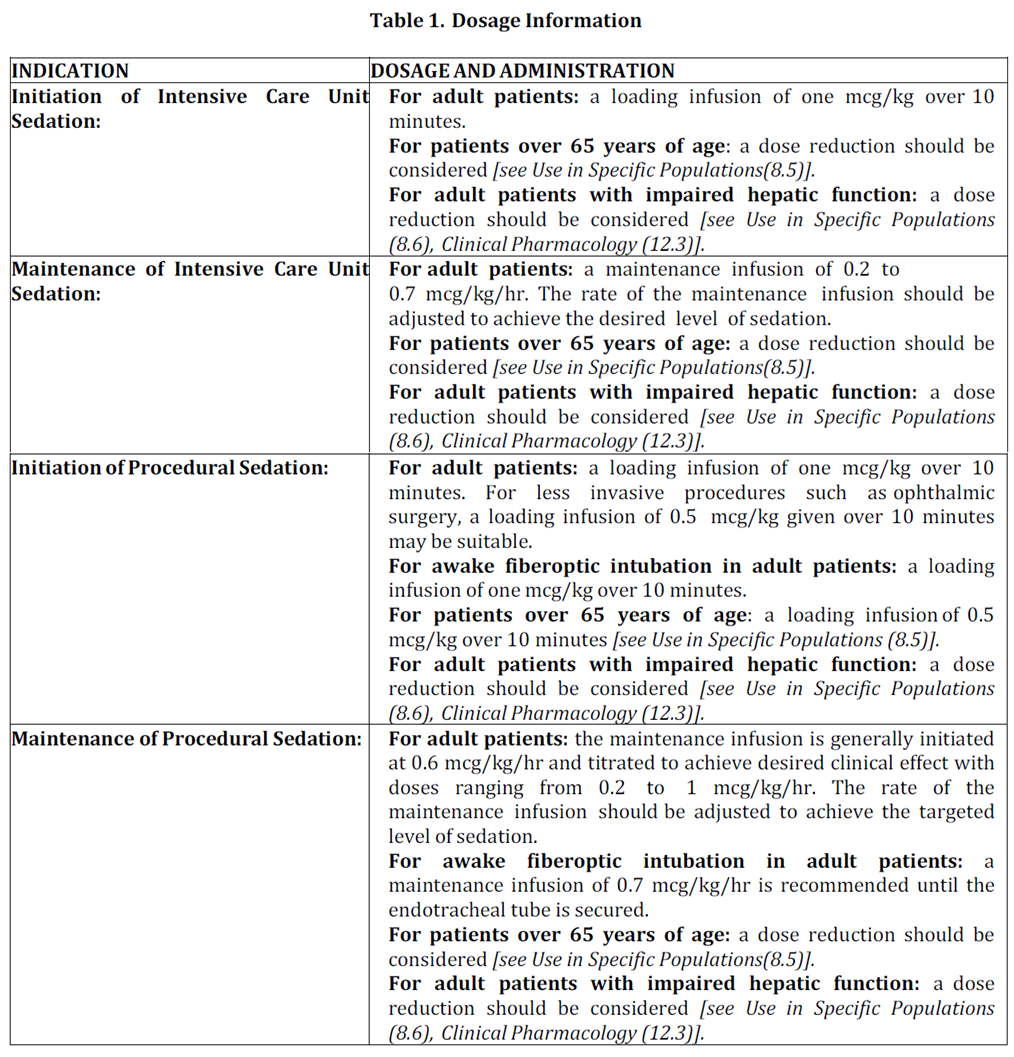
2.3 Dosage Adjustment
Due to possible pharmacodynamic interactions, a reduction in dosage of Dexmedetomidine HCl or other concomitant anesthetics, sedatives, hypnotics or opioids may be required when co- administered [see Drug Interactions (7.1) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
Dosage reductions may need to be considered for adult patients with hepatic impairment, and geriatric patients [see Warnings and Precautions (5.7), Use in Specific Populations (8.6), Clinical Pharmacology (12.3) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
2.4 Preparation of Solution
Dexmedetomidine HCl must be diluted in 0.9% sodium chloride solution to achieve required concentration (4 mcg/mL) prior to administration. Preparation of solutions is the same, whether for the loading dose or maintenance infusion.
Strict aseptic technique must always be maintained during handling of Dexmedetomidine HCl.
To prepare the infusion, withdraw 2 mL of Dexmedetomidine HCl and add to 48 mL of 0.9% sodium chloride injection to a total of 50 mL. Shake gently to mix well.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
2.5 Administration with Other Fluids
Dexmedetomidine HCl infusion should not be co-administered through the same intravenous catheter with blood or plasma because physical compatibility has not been established.
Dexmedetomidine HCl has been shown to be incompatible when administered with the following drugs: amphotericin B, diazepam.
Dexmedetomidine HCl has been shown to be compatible when administered with the following intravenous fluids and drugs:
0.9% sodium chloride in water, 5% dextrose in water, 20% mannitol, alfentanil hydrochloride, amikacin sulfate, aminophylline, amiodarone hydrochloride, ampicillin sodium, ampicillin sodium-sulbactam sodium, atracurium besylate, atropine sulfate, azithromycin, aztreonam, bretylium tosylate, bumetanide, butorphanol tartrate, calcium gluconate, cefazolin sodium, cefepime hydrochloride, cefoperazone sodium, cefotaxime sodium, cefotetan sodium, cefoxitin sodium, ceftazidime, ceftizoxime sodium, ceftriaxone sodium, cefuroxime sodium, chlorpromazine hydrochloride, cimetidine hydrochloride, ciprofloxacin, cisatracurium besylate, clindamycin phosphate, dexamethasone sodium phosphate, digoxin, diltiazem hydrochloride, diphenhydramine hydrochloride, dobutamine hydrochloride, dolasetron mesylate, dopamine hydrochloride, doxycycline hyclate, droperidol, enalaprilat, ephedrine hydrochloride, epinephrine hydrochloride, erythromycin lactobionate, esmolol, etomidate, famotidine, fenoldopam mesylate, fentanyl citrate, fluconazole, furosemide, gatifloxacin, gentamicin sulfate, glycopyrrolate bromide, granisetron hydrochloride, haloperidol lactate, heparin sodium, hydrocortisone sodium succinate, hydromorphone hydrochloride, hydroxyzine hydrochloride, inamrinone lactate, isoproterenol hydrochloride, ketorolac tromethamine, labetalol, lactated Ringer’s solution, levofloxacin, lidocaine hydrochloride, linezolid, lorazepam, magnesium sulfate, meperidine hydrochloride, methylprednisolone sodium succinate, metoclopramide hydrochloride, metronidazole, midazolam, milrinone lactate, mivacurium chloride, morphine sulfate, nalbuphine hydrochloride, nitroglycerin, norepinephrine bitartrate, ofloxacin, ondansetron hydrochloride, pancuronium bromide, phenylephrine hydrochloride, piperacillin sodium, piperacillin sodium-tazobactam sodium, potassium chloride, procainamide hydrochloride, prochlorperazine edisylate, promethazine hydrochloride, propofol, ranitidine hydrochloride, rapacuronium bromide, remifentanil hydrochloride, rocuronium bromide, sodium bicarbonate, sodium nitroprusside, succinylcholine, sufentanil citrate, sulfamethoxazole-trimethoprim, theophylline, thiopental sodium, ticarcillin disodium, ticarcillin disodium-clavulanate potassium, tobramycin sulfate, vancomycin hydrochloride, vecuronium bromide, verapamil hydrochloride, and a plasma-substitute.
2.6 Compatibility with Natural Rubber
Compatibility studies have demonstrated the potential for absorption of Dexmedetomidine HCl to some types of natural rubber. Although Dexmedetomidine HCl is dosed to effect, it is advisable to use administration components made with synthetic or coated natural rubber gaskets.
Contraindications
4 CONTRAINDICATIONS
None
