NABOTA POWDER FOR SOLUTION FOR INJECTION 100 UNITS/VIAL [SIN16680P]
Active ingredients: NABOTA POWDER FOR SOLUTION FOR INJECTION 100 UNITS/VIAL
Product Info
NABOTA POWDER FOR SOLUTION FOR INJECTION 100 UNITS/VIAL
[SIN16680P]
Product information
Active Ingredient and Strength | CLOSTRIDIUM BOTULINUM TOXIN TYPE A - 100 UNITS/VIAL |
Dosage Form | INJECTION, POWDER, LYOPHILIZED, FOR SOLUTION |
Manufacturer and Country | DAEWOONG PHARMACEUTICAL CO., LTD. - KOREA, REPUBLIC OF |
Registration Number | SIN16680P |
Licence Holder | HYPHENS PHARMA PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | M03AX01 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
4.1.1. Glabellar lines
Temporary improvement in the appearance of moderate to severe glabellar lines (vertical lines between the eyebrows) associated with corrugator muscle and/or procerus muscle activity in adults below 65 years of age.
4.1.2. Focal upper limb spasticity
Upper limb spasticity associated with stroke in adults aged over 18 years.
4.1.3. Lateral canthal lines (crow's feet lines)
Temporary improvement in the appearance of moderate to severe lateral canthal lines (crow's feet lines) associated with orbicularis oculi muscle activity in adults below 65 years of age.
4.1.4. Blepharospasm
Treatment of benign essential blepharospasm in adults aged over 18 years.
4.1.5. Benign masseter muscle hypertrophy (square jaw)
Temporary improvement in marked* to very marked** benign masseter muscle hypertrophy associated with conditions such as bruxism (including teeth clenching and grinding) in adults below 65 years of age.
* Marked:
At rest, the lower face contour, including the masseter muscle, jaw, and jawline, is square, and the surface of the masseter muscle is severely convex. At maximum clenching, the lower face contour shows a severe difference compared to at rest, and the masseter muscle mass is firm or hard and palpable.** Very marked:
At rest, the lower face contour, including the masseter muscle, jaw, and jawline, is trapezoidal, and the surface of the masseter muscle is severely convex. At maximum clenching, the lower face contour shows a severe difference compared to at rest, and the masseter muscle is hard and palpable
Dosing
4.2 Posology and method of administration
For intramuscular use only.
4.2.1. Glabellar lines
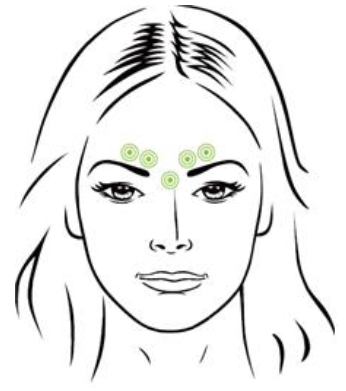
Reconstitute by diluting with preservative-free, sterile saline solution to make 100units/2.5mL (4units/0.1mL). Using a sterile 30-gauge needle, inject a dose of 0.1mL into each of the 5 injection sites: 2 injections in each corrugator muscle and 1 injection in the procerus muscle for a total dose of 20 Units.
Physical manipulation (such as rubbing) of the injection site in the immediate post-administration period should be avoided.
In order to reduce the complication of ptosis, injections near the levator palpebrae superioris muscle must be avoided, particularly in patients with larger brow-depressor complexes (depressor supercilii). Injections into inner corrugators muscle and central eyebrow should be placed at least 1 cm above the bony supraorbital ridge.
Careful attention should be paid to avoid injection of this product into the blood vessel. The thumb or index finger should be placed firmly below the orbital rim in order to prevent extravasation below the orbital rim. The needle should be oriented superiorly and medially during the injection and careful attention should be paid to inject accurate volume.
Glabellar facial lines arise from the activity of corrugator muscle and orbicularis oculi muscle. These muscles move the brow medially, and the procerus muscle and depressor supercilii muscle pull the brow inferiorly. This creates a frown or “furrowed brow”. The location, size, and use of the muscles vary markedly among individuals. An effective dose for facial lines is determined by gross observation of the patient’s ability to activate the superficial muscles injected.
Each treatment lasts approximately three to four months. More frequent injection of this product is not recommended because the safety and efficacy are not established.
Typically, the initial doses of botulinum toxin induce chemical denervation of the injected muscles one to two days after injection, increasing in intensity during the first week.
4.2.2. Focal upper limb spasticity
The exact dosage and number of injection sites may be tailored to the individual based on the size, number and location of the muscles involved, the severity of spasticity, the presence of local muscle weakness, and the patient's response to previous treatment. Clinical improvement of spasticity was observed within 4 weeks after injection, which lasted for 8 to 12 weeks.
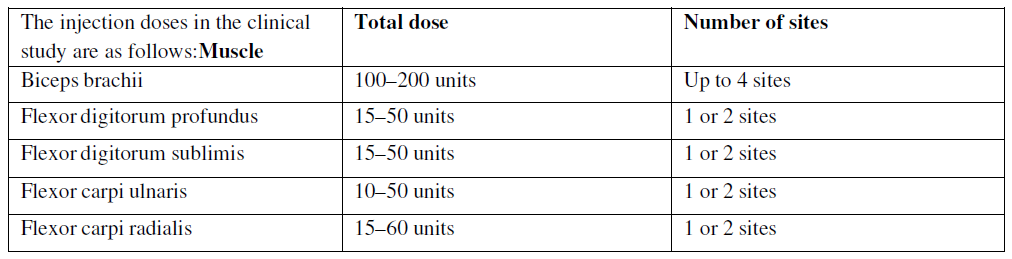
In the clinical study, doses ranging up to 360 units were divided among selected muscles.
A sterile 24–30 gauge needle is used for superficial muscles, and a needle of appropriate length is used for muscle tissues. Localization of the involved muscles with techniques such as electromyographic guidance or nerve stimulation is recommended.
4.2.3. Lateral canthal lines (crow's feet lines)
Lateral canthal lines arise largely from the activity of the orbicularis oculi muscles, which are around the eyes and responsible for blinking and eyelid closure. Forceful contraction of the orbicularis oculi results in lateral and radially oriented folds (crow's feet lines), which originate from the lateral canthus. The distribution of these radial lines differs among patients.
Injections should be given with the needle bevel tip pointed up and oriented away from the eye. Using a 30–33 gauge needle, inject a dose of 0.1 mL (4 units) into 3 sites per side (a total of 6 sites and 24 units for both sides) in the lateral orbicularis oculi muscle.
The first injection should be approximately 1.5–2.0 cm temporal to the lateral canthus and just temporal to the orbital rim. If the lines in the lateral canthus at maximum smile are above and below the lateral canthus, inject as shown in the figure.
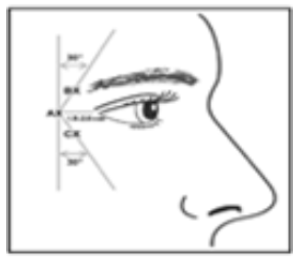
The duration of effect of NABOTA for lateral canthal lines is approximately 3 months. The safety and efficacy of the injection interval have not been clinically evaluated.
4.2.4. Blepharospasm
For blepharospasm, reconstitute by dilution (see Dilution Table) and inject using a sterile 27–30 gauge needle without electromyographic indicator. The recommended initial dose for injection into the medial and lateral pretarsal orbicularis oculi of the upper lid and into the lateral pretarsal orbicularis oculi of the lower lid is 2.5 units (0.1 mL at each site). If blepharospasm blocks vision, injections into the medial corrugator supercilii muscle and superolateral portion of orbicularis oculi are possible. At repeat treatment sessions, the same dose as the most recent total injection dose may be injected up to 5 units per injection site (a total of 50 units).
The safety and efficacy of a single dose of NABOTA were assessed for blepharospasm for 12 weeks.
The safety and efficacy of the injection interval have not been clinically evaluated.
4.2.5. Benign masseter muscle hypertrophy
Inject a dose of 0.2 mL (8 units) into each of 3 sites on each side of the bilateral masseter muscles (a total of 6 sites and 1.2 mL (48 units) for both sides) using a 30–33 gauge needle.
Before the injection, palpate the masseter muscle to mark the anterior and posterior borders when the patient clenches their teeth.
Consider the anterior and posterior borders as left and right boundaries, respectively. Consider the line from tragus to angle of mouth to be a superior border and the mandible border line to be an inferior border and mark a zone 1 cm inside each border.
Inject into 2 sites in the inferior border of the mandible and 1 site above them corresponding to a vertex of an imaginary triangle around the most prominent bulges within the rectangle imagined by the borders (a total of 3 sites). The injection sites should be 1 cm apart.
Make sure to carefully administer the product 1 cm inward from the left/right and anterior/posterior borderlines after determining the injection sites by identifying the location of the masseter muscles and referring to the figure below, as the administration may cause discomfort in chewing and changes in facial expression when smiling. Be careful to avoid injection into the muscles around the mouth.
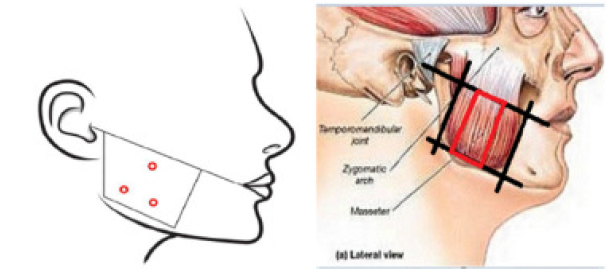
Injection sites of botulinum toxin for MMH and anatomical image of masseter muscle
Efficacy and long-term safety information on Benign masseter muscle hypertrophy has been obtained through the Open Label Extension (OLE) study, including the second administration of investigational drug at 24–28 weeks, totalling a period of 48–52 weeks.
Elderly patients
NABOTA is not recommended for use in patients over 65 years of age.
Preparation and Dilution Technique
Prior to injection, reconstitute the product with a preservative-free, sterile saline. 0.9% sodium chloride injection is the recommended diluent. Draw up the proper amount of diluent in the syringe of appropriate size. Since this product is denatured by bubbling or similar violent agitation, the diluent should be injected gently into the vial. Discard the vial if a vacuum does not pull the diluent into the vial. Record the date and time of reconstitution on the label. This product should be administered within 24 hours after reconstitution. During this period, reconstituted product should be stored in a refrigerator (2–8°C). Reconstituted product should be clear, colourless and free of particulate matter. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Because this product and the diluent do not contain any preservative, one vial of this product should be used for a single patient treatment only during a single session.

Contraindications
4.3 Contraindications
Patients who are hypersensitive to any ingredient in the formulation of this product
Patients who have neuromuscular junctional disorders (e.g. myasthenia gravis, Lambert-Eaton syndrome, or amyotrophic lateral sclerosis). The diseases may be exacerbated due to the muscle relaxation activity of this drug product.
Pregnant women, women of childbearing potential or nursing mothers.
Infection or inflammation at the proposed injection sites.
