TECVAYLI SOLUTION FOR INJECTION 90MG/ML [SIN16949P]
Active ingredients: TECVAYLI SOLUTION FOR INJECTION 90MG/ML
Product Info
TECVAYLI SOLUTION FOR INJECTION 90MG/ML
[SIN16949P]
Product information
Active Ingredient and Strength | TECLISTAMAB - 90 MG/ML |
Dosage Form | INJECTION, SOLUTION |
Manufacturer and Country | PATHEON MANUFACTURING SERVICES LLC (BULK PRODUCTION/PRIMARY PACKAGER) - UNITED STATES |
Registration Number | SIN16949P |
Licence Holder | JOHNSON & JOHNSON INTERNATIONAL (SINGAPORE) PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01FX24 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Indications
TECVAYLI® as monotherapy is indicated for the treatment of adult patients with relapsed or refractory multiple myeloma who have received at least three prior therapies, including a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 monoclonal antibody, and have demonstrated disease progression on the last therapy.
Dosing
Dosage and Administration
Dosage – Adults
TECVAYLI® should be administered by subcutaneous injection only.
Administer pretreatment medications prior to each dose of the TECVAYLI® step-up dosing schedule (see Dosage and Administration – Pretreatment medications).
Recommended dosing schedule
The recommended dosing schedule for TECVAYLI® is provided in Table 1. The recommended dosage of TECVAYLI® is step-up doses of 0.06 mg/kg and 0.3 mg/kg followed by 1.5 mg/kg once weekly until disease progression or unacceptable toxicity.
In patients who have a complete response or better for a minimum of 6 months, a reduced dosing frequency of 1.5 mg/kg every two weeks until disease progression or unacceptable toxicity may be considered (see Clinical Studies – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Administer TECVAYLI® according to the step-up dosing schedule in Table 1 to reduce the incidence and severity of cytokine release syndrome (CRS). Due to the risk of cytokine release syndrome (CRS), instruct patients to remain within proximity of a healthcare facility and monitor patients for signs and symptoms daily for 48 hours after administration of all doses within the TECVAYLI® step-up dosing schedule (see Dosage and Administration – Administration and Warnings and Precautions - Cytokine Release Syndrome – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Failure to follow the recommended doses or dosing schedule for initiation of therapy or re-initiation of therapy after dose delays may result in increased frequency and severity of adverse events related to mechanism of action, particularly cytokine release syndrome (see Dosage and Administration - Dosage modifications and Warnings and Precautions – Cytokine Release Syndrome – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
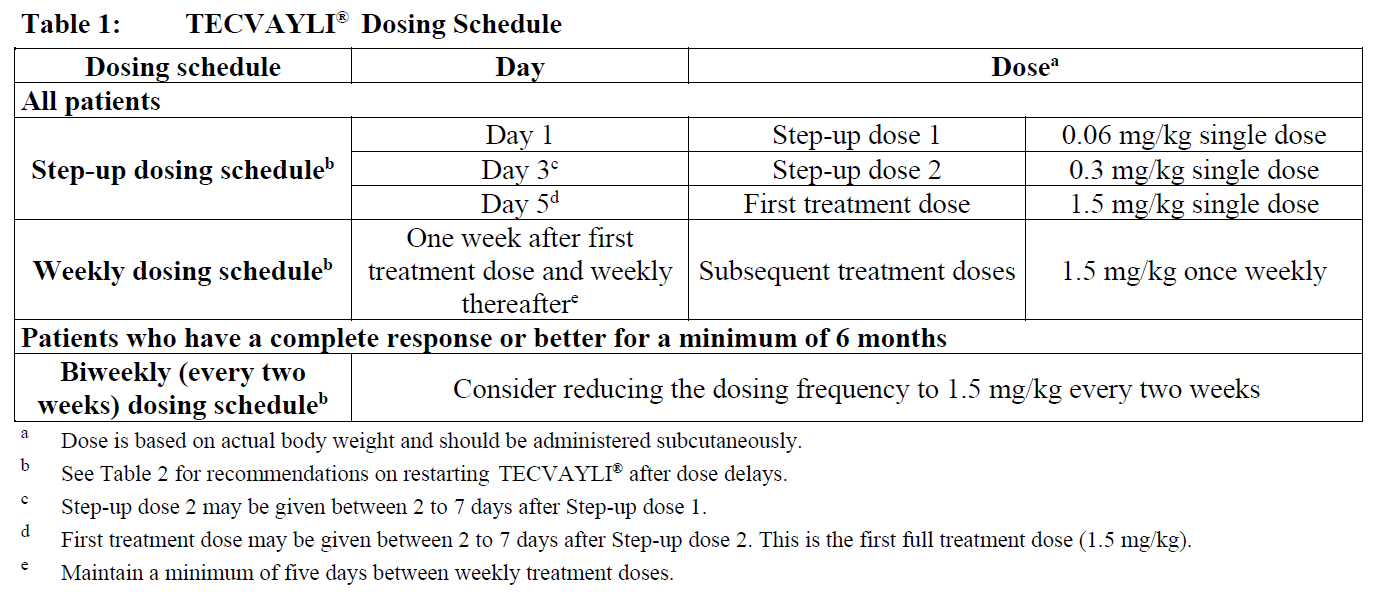
Refer to Tables 6, 7 and 8 to determine the dosage based on predetermined weight ranges (see Dosage and Administration – Preparation of TECVAYLI®).
For guidance regarding restarting therapy with TECVAYLI® after dose delays, (see Dosage and Administration - Restarting TECVAYLI® after dose delays).
Pretreatment medications
Administer the following pretreatment medications 1 to 3 hours before each dose of the TECVAYLI® step-up dosing schedule to reduce the risk of cytokine release syndrome (see Warnings and Precautions - Cytokine Release Syndrome and Adverse Reactions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Corticosteroid (oral or intravenous dexamethasone, 16 mg)
Antihistamine (oral or intravenous diphenhydramine, 50 mg or equivalent)
Antipyretics (oral or intravenous acetaminophen, 650 mg to 1000 mg or equivalent)
Administration of pretreatment medications may be required prior to administration of subsequent doses of TECVAYLI® in the following patients: (see Dosage and Administration – Dosage modifications).
Patients who repeat doses within the TECVAYLI® step-up dosing schedule following a dose delay (see Dosage and Administration – Restarting TECVAYLI® after dose delays).
Patients who experienced CRS following the prior dose of TECVAYLI® (see Dosage and Administration – Dosage modifications).
Prophylaxis for herpes zoster virus reactivation
Prior to starting treatment with TECVAYLI®, anti-viral prophylaxis should be considered for the prevention of herpes zoster virus reactivation per local institutional guidelines.
Restarting TECVAYLI® after dose delays
If a dose of TECVAYLI® is delayed, restart therapy based on the recommendations listed in Table 2 and resume the treatment schedule accordingly (see Dosage and Administration-Dosage – Adults). Administer pretreatment medications as indicated in Table 2 and monitor patients following administration of TECVAYLI® accordingly (see Dosage and Administration – Pretreatment medications and Dosage and Administration - Administration).
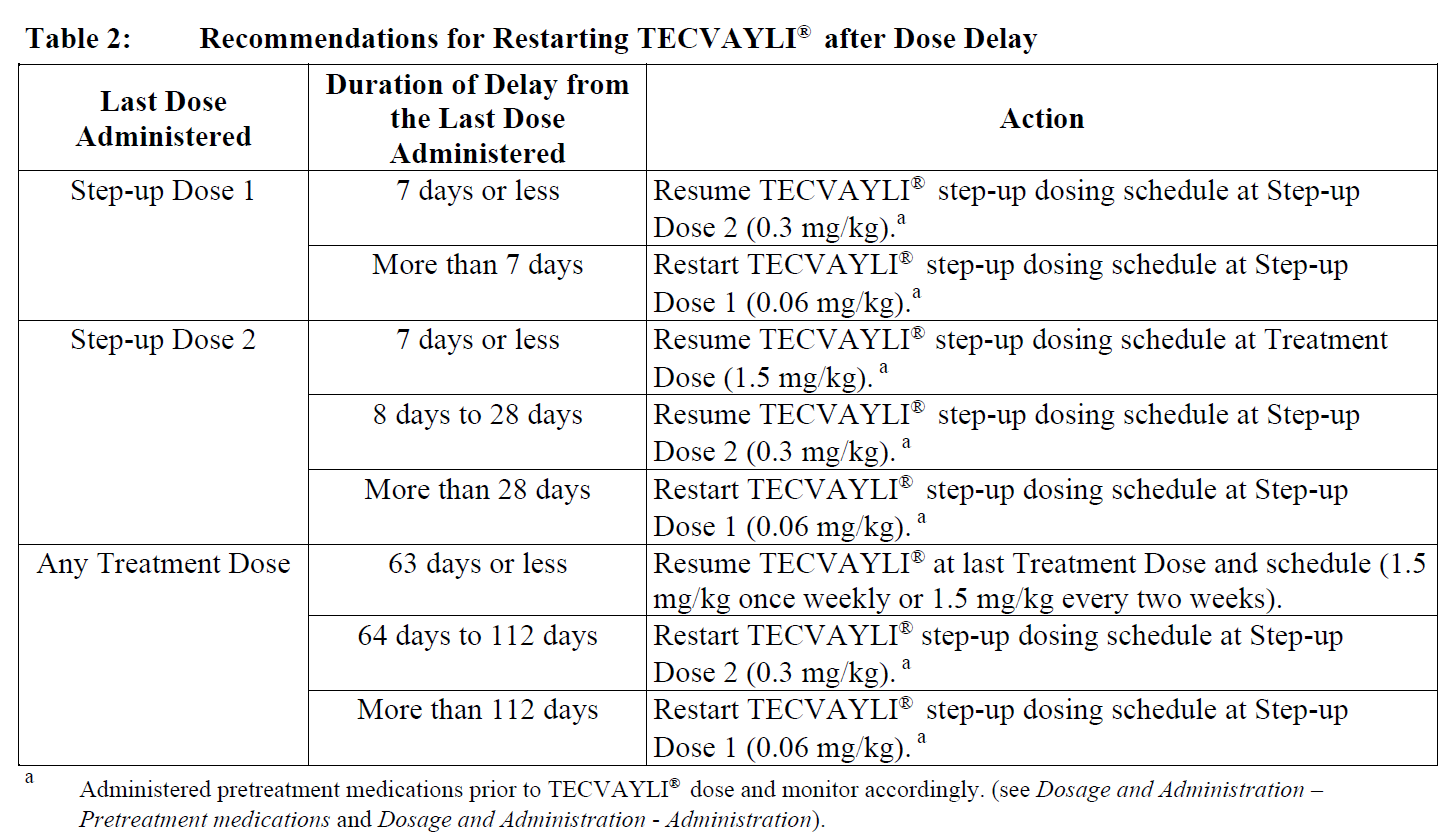
Dosage modifications
Do not skip step-up doses of TECVAYLI®.
Dose reductions of TECVAYLI® are not recommended.
Dose delays may be required to manage toxicities related to TECVAYLI® (see Warnings and Precautions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
See Table 3 for recommended actions for adverse reactions following administration of TECVAYLI®.
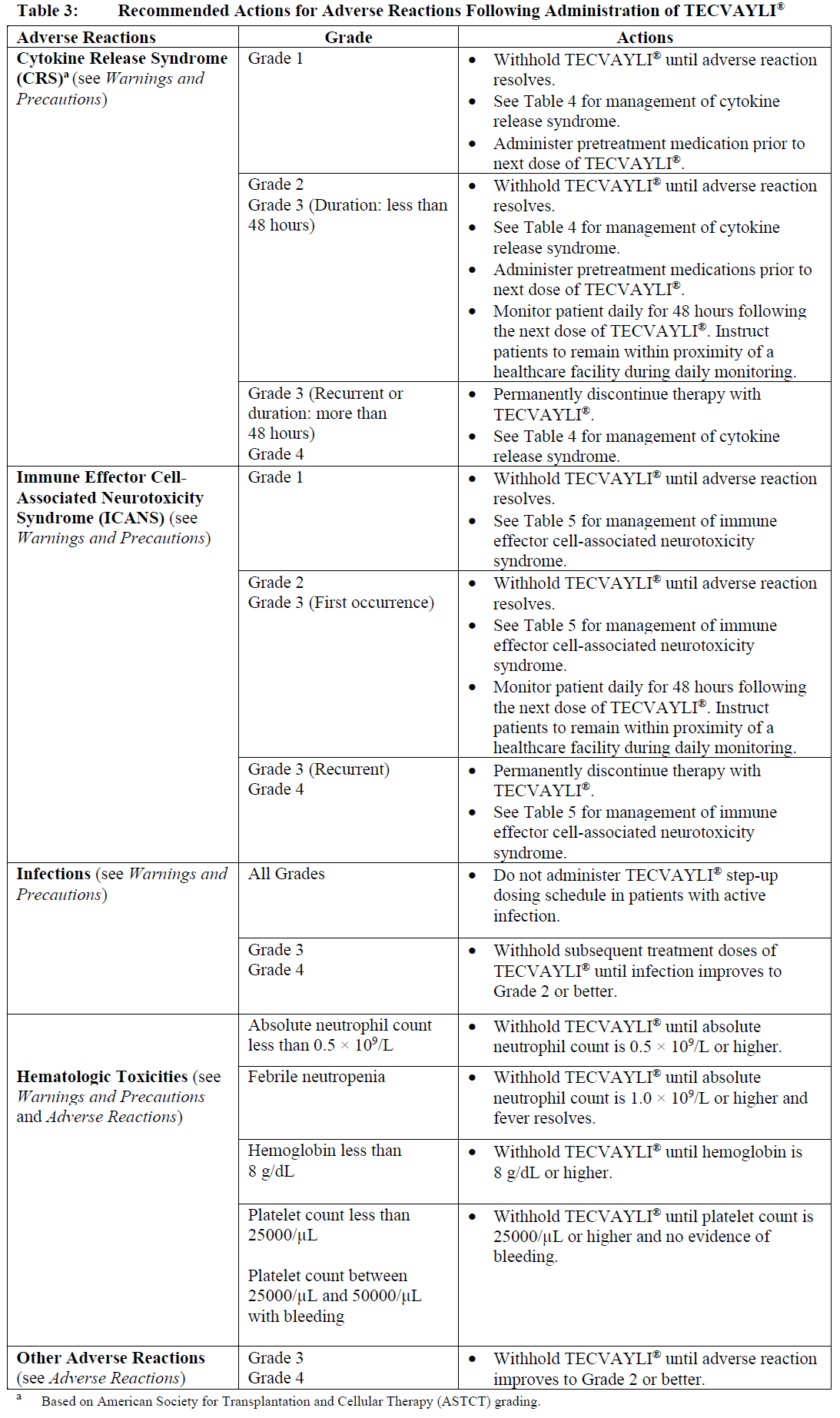
Management of severe adverse reactions
Cytokine release syndrome (CRS)
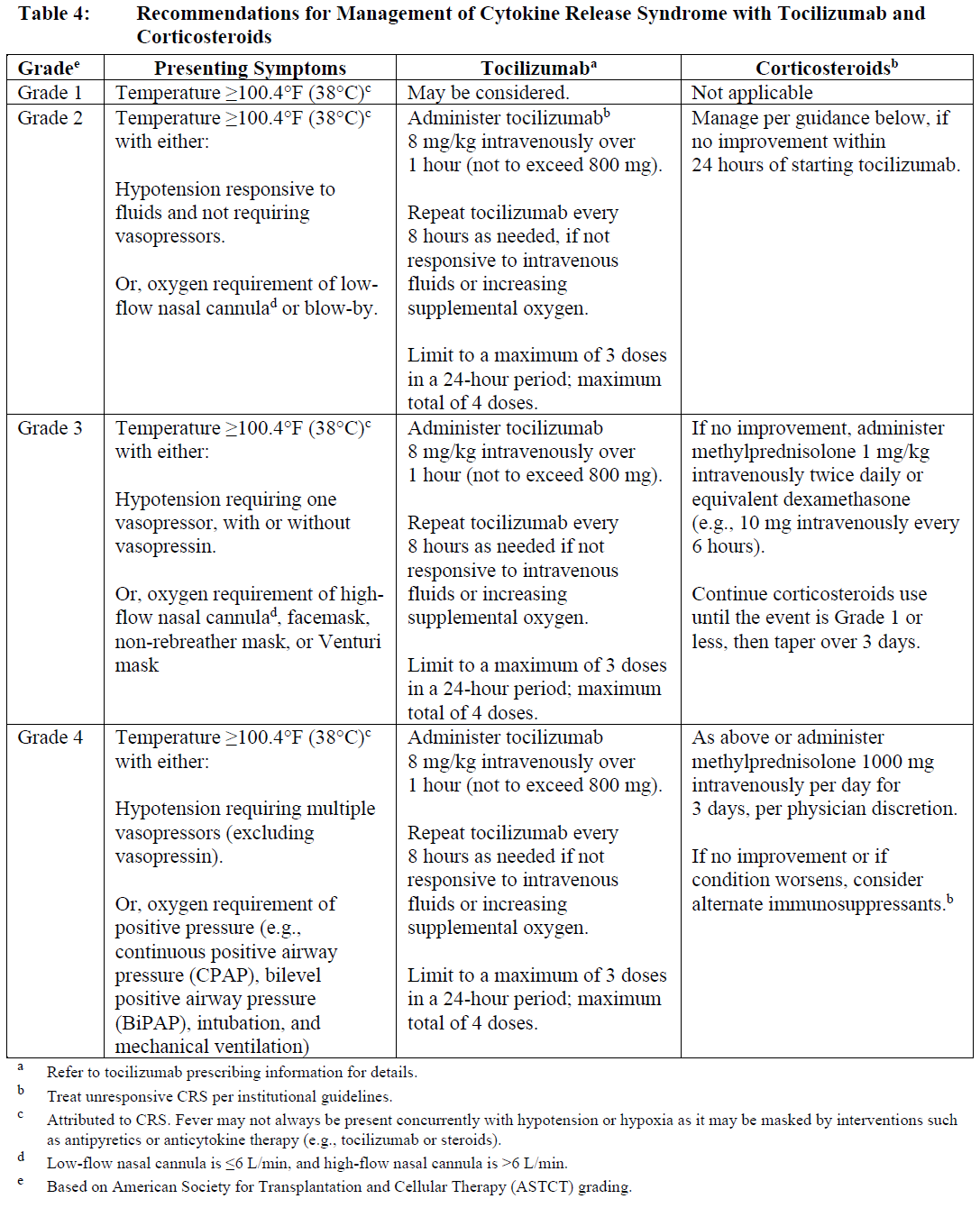
Identify CRS based on clinical presentation (see Warnings and Precautions - Cytokine Release Syndrome – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). Evaluate and treat other causes of fever, hypoxia, and hypotension.
If CRS is suspected, withhold TECVAYLI® until the adverse reaction resolves (see Table 3) and manage according to the recommendations in Table 4. Administer supportive care for CRS (including but not limited to anti-pyretic agents, intravenous fluid support, vasopressors, supplemental oxygen, etc.) as appropriate. Consider laboratory testing to monitor for disseminated intravascular coagulation (DIC), hematology parameters, as well as pulmonary, cardiac, renal, and hepatic function.
Neurologic toxicities
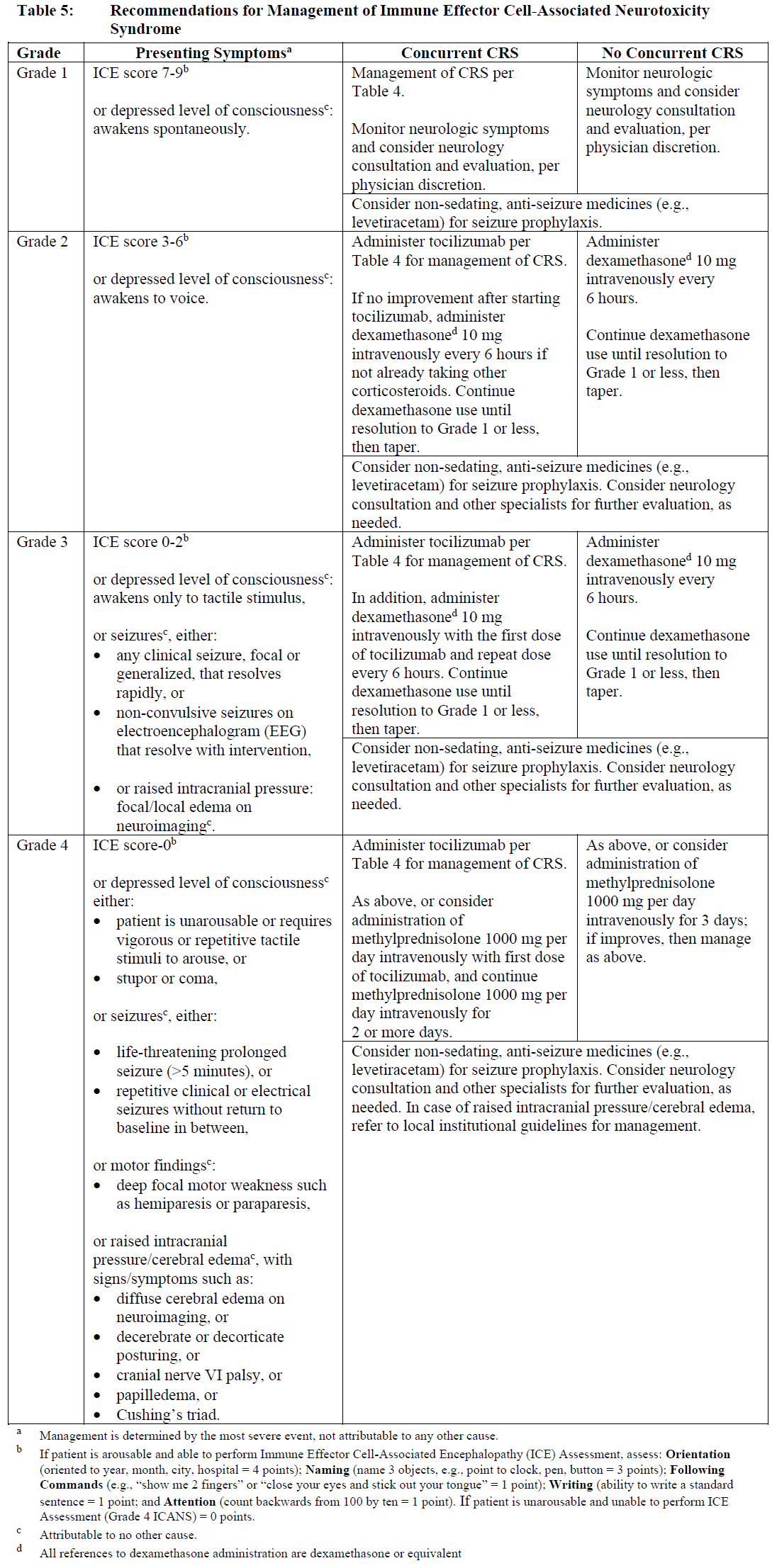
General management for neurologic toxicity (e.g., Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS) with or without concurrent CRS) is summarized in Table 5.
At the first sign of neurologic toxicity including ICANS, consider neurology evaluation. Rule out other causes of neurologic symptoms. Provide intensive care and supportive therapy for severe or life-threatening neurologic toxicities (see Warnings and Precautions – Neurologic toxicities – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). Withhold TECVAYLI® as indicated in Table 3.
Special populations
Pediatrics
The safety and efficacy of TECVAYLI® have not been established in pediatric patients.
No data are available.
Elderly (65 years of age and older)
Of the 165 patients treated with TECVAYLI® in MajesTEC-1 at the recommended dose, 48% were 65 years of age or older, and 15% were 75 years of age or older. No overall differences in safety or effectiveness were observed between these patients and younger patients.
No dose adjustment is necessary (see Pharmacokinetic Properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal impairment
No formal studies of TECVAYLI® in patients with renal impairment have been conducted.
Based on population pharmacokinetic analyses, no dose adjustment is recommended for patients with mild or moderate renal impairment (see Pharmacokinetic Properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Hepatic impairment
No formal studies of TECVAYLI® in patients with hepatic impairment have been conducted.
Based on population pharmacokinetic analyses, no dose adjustment is recommended for patients with mild hepatic impairment (see Pharmacokinetic Properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Administration
It is very important that the instructions for preparation and administration provided in this section are strictly followed to minimize potential dosing errors with TECVAYLI® 10 mg/mL vial and TECVAYLI® 90 mg/mL vial.
TECVAYLI® should be administered via subcutaneous injection only. Do not administer TECVAYLI® intravenously.
TECVAYLI® should be administered by a healthcare professional with adequately trained medical personnel and appropriate medical equipment to manage severe reactions, including cytokine release syndrome (see Warnings and Precautions - Cytokine Release Syndrome – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
TECVAYLI® 10 mg/mL vial and TECVAYLI® 90 mg/mL vial are supplied as ready-to-use solution for injection that do not need dilution prior to administration.
TECVAYLI® vials of different concentrations should not be combined to achieve treatment dose.
Use aseptic technique to prepare and administer TECVAYLI®.
Preparation of TECVAYLI®
• Verify the prescribed dose for each TECVAYLI® injection. To minimize errors, use the following tables to prepare TECVAYLI® injection.
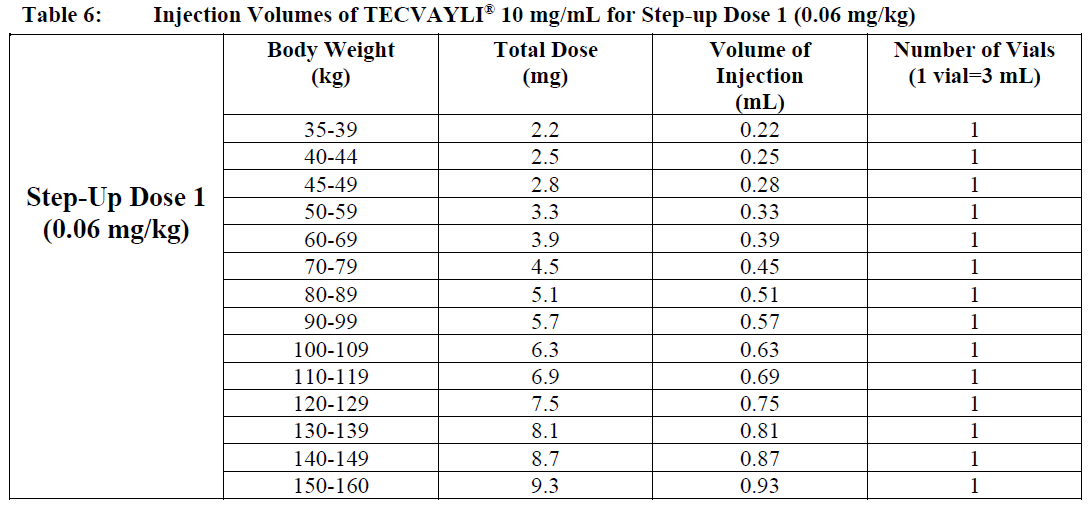
○ Use Table 6 to determine total dose, injection volume and number of vials required based on patient’s actual body weight for Step-up Dose 1 using TECVAYLI® 10 mg/mL.
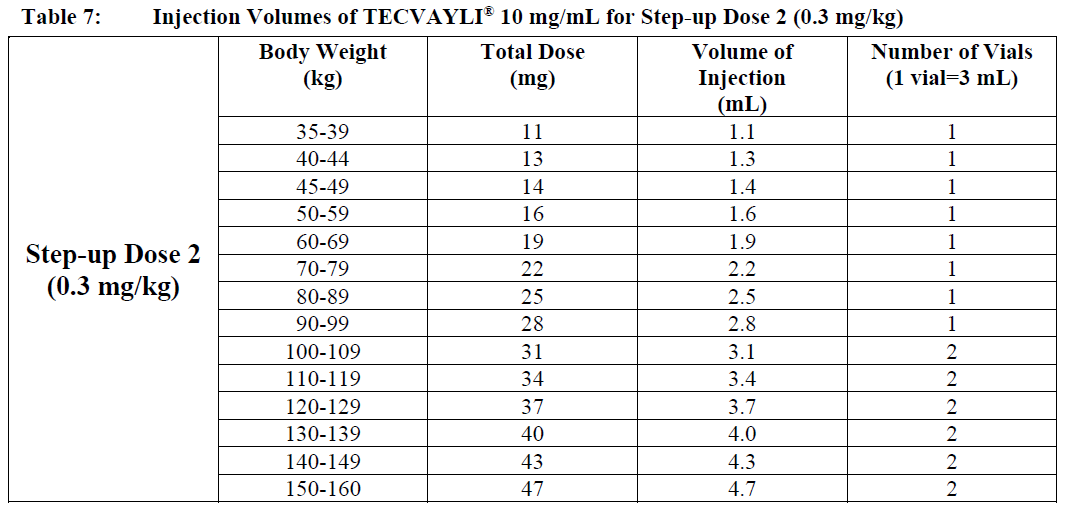
○ Use Table 7 to determine total dose, injection volume and number of vials required based on patient’s actual body weight for Step-up Dose 2 using TECVAYLI® 10 mg/mL.
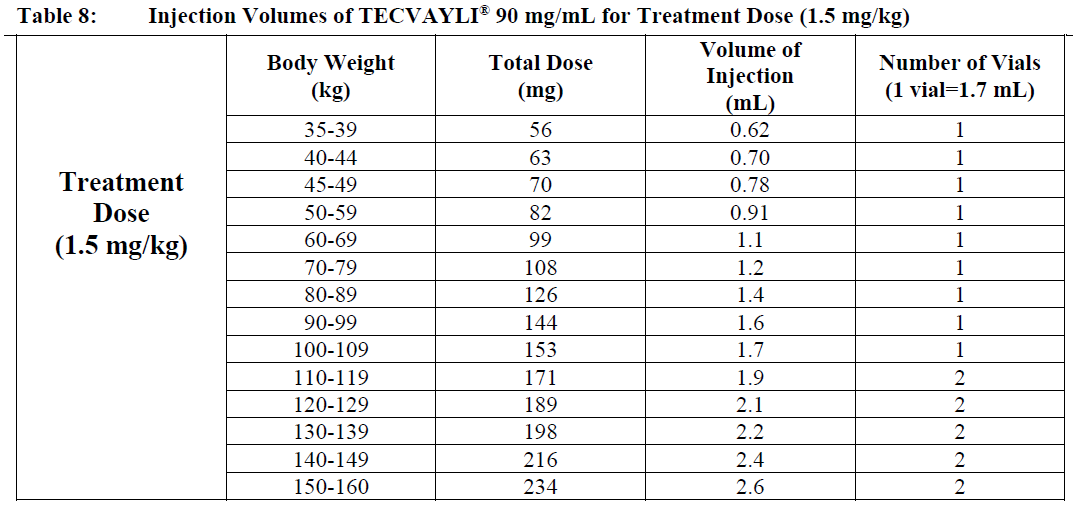
○ Use Table 8 to determine total dose, injection volume and number of vials required based on patient’s actual body weight for the Treatment Dose using TECVAYLI® 90 mg/mL.
• Remove the appropriate strength TECVAYLI® vial from refrigerated storage [2°C–8°C (36°F–46°F)] and equilibrate to ambient temperature [15°C–30°C (59°F–86°F)], as needed, for at least 15 minutes. Do not warm TECVAYLI® in any other way.
• Once equilibrated, gently swirl the vial for approximately 10 seconds to mix. Do not shake.
• Withdraw the required injection volume of TECVAYLI® from the vial(s) into an appropriately sized syringe using a transfer needle.
○ Each injection volume should not exceed 2.0 mL. Divide doses requiring greater than 2.0 mL equally into multiple syringes.
• TECVAYLI® is compatible with stainless steel injection needles and polypropylene or polycarbonate syringe material.
• Replace the transfer needle with an appropriately sized needle for injection.
• Visually inspect TECVAYLI® for particulate matter and discoloration prior to administration. Do not use if the solution is discolored, or cloudy, or if foreign particles are present.
○ TECVAYLI® solution for injection is colorless to light yellow.
Administration of TECVAYLI®
Inject the required volume of TECVAYLI® into the subcutaneous tissue of the abdomen (preferred injection site). Alternatively, TECVAYLI® may be injected into the subcutaneous tissue at other sites (e.g., thigh). If multiple injections are required, TECVAYLI® injections should be at least 2 cm apart.
Do not inject into tattoos or scars or areas where the skin is red, bruised, tender, hard or not intact.
Any unused medicinal product or waste material should be disposed in accordance with local requirements.
Storage
The prepared syringes should be administered immediately. If immediate administration is not possible, in-use storage times of the prepared syringe should be no longer than 20 hours at 2°C – 8°C or ambient temperature (15°C – 30°C). Discard after 20 hours, if not used.
Monitoring
Instruct patients to remain within proximity of a healthcare facility and monitor patients daily for 48 hours for signs and symptoms of CRS after administration of all doses within the TECVAYLI® step-up dosing schedule (see Table 1 and Dosage and Administration-Management of severe adverse reactions and Warnings and Precautions - Cytokine Release Syndrome – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Contraindications
Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in List of Excipients – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
