DIGOKERN TABLETS 0.25MG [SIN17078P]
Active ingredients: DIGOKERN TABLETS 0.25MG
Product Info
DIGOKERN TABLETS 0.25MG
[SIN17078P]
Product information
Active Ingredient and Strength | DIGOXIN - 0.25 MG |
Dosage Form | TABLET |
Manufacturer and Country | KERN PHARMA, S.L. - SPAIN |
Registration Number | SIN17078P |
Licence Holder | APOTHECA MARKETING PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | C01AA05 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Cardiac failure:
Digoxin is indicated in the treatment of chronic heart failure where systolic dysfunction is the main problem. The greatest therapeutic benefit is obtained in patients with ventricular dilatation.
Digoxin is specifically indicated when heart failure is accompanied by atrial fibrillation.
Supraventricular arrhythmias:
Digoxin is indicated in the treatment of certain supraventricular arrhythmias, particularly chronic atrial flutter and fibrillation.
Dosing
4.2 Dosage and Administration
The dose of digoxin for each patient has to be tailored individually according to age, lean body weight and renal function. Suggested doses are intended only as an initial guide. The score is used to split and facilitate swallowing but not to divide the tablet into equal doses.
The difference in bioavailability between injectable digoxin and oral formulations must be considered when changing from one dosage form or brand to another. For example if patients are switched from oral to the i.v. formulation the dosage should be reduced by approximately 33 %.
Monitoring
Serum concentrations of digoxin may be expressed in Conventional Units of ng/ml or SI Units of nmol/l. To convert ng/ml to nmol/l, multiply ng/ml by 1.28. The serum concentration of digoxin can be determined by radioimmunoassay. Blood should be taken 6 hours or more after the last dose of digoxin.
There are no rigid guidelines as to the range of serum concentrations that are most efficacious. A post hoc analysis of heart failure patients in the Digitalis Investigation Group trial demonstrated that at low serum digoxin concentrations (0.5–0.9 ng/ml), the use of digoxin was associated with reductions in mortality and hospitalisation. Patients with higher digoxin levels (> 1ng/ml) had a higher incidence of morbidity and mortality, although at these concentrations digoxin reduces heart failure hospitalisation. Therefore, the optimal trough digoxin serum level may be 0.5 ng/mL (0.64 nanomol/L) to 1.0 ng/mL (1.28 nanomol/L).
Digoxin toxicity is more commonly associated with serum digoxin concentration greater than 2 ng/mL. However, serum digoxin concentration should be interpreted in the clinical context. Toxicity may occur with lower digoxin serum concentrations. In deciding whether a patient’s symptoms are due to digoxin, the clinical state together with the serum potassium level and thyroid function are important factors (see Overdose – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Other glycosides, including metabolites of digoxin, can interfere with the assays that are available and one should always be wary of values which do not seem commensurate with the clinical state of the patient.
Populations
• Adults and children over 10 years
Rapid Oral Loading:
If medically appropriate, rapid digitalisation may be achieved in a number of ways, such as the following: 750 to 1500 micrograms (0.75 to 1.5 mg) as a single dose.
Where there is less urgency, or greater risk of toxicity, e.g. in the elderly, the oral loading dose should be given in divided doses 6 hours apart, with approximately half the total dose given as the first dose. Clinical response should be assessed before giving each additional dose (see Special warnings and precautions for use – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Slow Oral Loading:
In some patients, for example those with mild heart failure, digitalisation may be achieved more slowly with doses of 250 to 750 micrograms (0.25 to 0.75 mg) daily for 1 week followed by an appropriate maintenance dose. A clinical response should be seen within one week.
NOTE: The choice between slow and rapid oral loading depends on the clinical state of the patient and the urgency of the condition.
Maintenance Dose:
The maintenance dose should be based on the percentage of the peak body stores lost each day through elimination. The following formula has had wide clinical use:
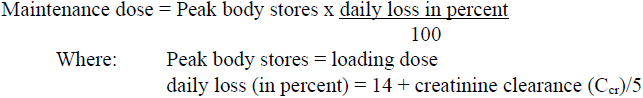
Ccr is creatinine clearance corrected to 70 kg bodyweight or 1.73 m2 body surface area. If only serum creatinine (Scr) concentrations are available, Ccr (corrected to 70 kg bodyweight) may be estimated in men as:
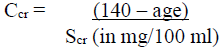
NOTE: Where serum creatinine values are obtained only in micromol/l, these can be converted into mg/100 ml (mg %) as follows:
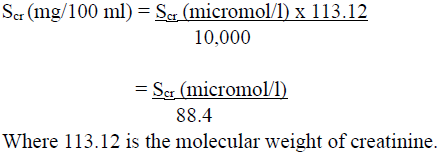
For women, this result should be multiplied by 0.85.
Note: These formulae cannot be used for creatinine clearance in children.
In practice this will mean that most patients with heart failure will be maintained on 125 to 250 micrograms (0.125 to 0.25 mg) of digoxin per day. However, in those who show increased sensitivity to the adverse effects of digoxin, a dose of 62.5 micrograms (0.0625 mg) daily or less may suffice. Conversely, some patients may require a higher dose.
• Neonates, infants and children up to 10 years of age
(if cardiac glycosides have not been given in the preceding two weeks)
An age-appropriate formulation should be used in this patient population.
If cardiac glycosides have been given in the two weeks preceding commencement of digoxin therapy, it should be anticipated that optimum loading doses of digoxin will be less than those recommended below. In the newborn, particularly in the premature infant, renal clearance of digoxin is diminished and suitable dose reductions must be observed, over and above general dosage instructions.
Beyond the immediate newborn period, children generally require proportionally larger doses than adults on the basis of body weight or body surface area, as indicated in the schedule below. Children over 10 years of age require adult doses in proportion to their body weight.
Oral Loading Dose:
This should be administered in accordance with the following schedule:
Table caption
Pre-term neonates < 1.5 kg | - 25 micrograms/kg per 24 hours. |
Pre-term neonates 1.5 kg to 2.5 kg | - 30 micrograms/kg per 24 hours. |
Term neonates to 2 years | - 45 micrograms/kg per 24 hours |
2 to 5 years | - 35 micrograms/kg per 24 hours. |
5 to 10 years | - 25 micrograms/kg per 24 hours. |
The loading dose should be administered in divided doses with approximately half of the total dose given as the first dose and further fractions of the total dose given at intervals of 4 to 8 hours, assessing the clinical response before giving each additional dose.
Maintenance
The maintenance dose should be administered in accordance with the following schedule:
Pre-term neonates:
daily dose = 20% of 24-hour loading dose.
Term neonates and children up to 10 years:
daily dose = 25% of 24-hour loading dose.
These dosage schedules are meant as guidelines and careful clinical observation and monitoring of serum digoxin levels (see Monitoring above) should be used as a basis for adjusting the dosage in these paediatric patient groups.
• Elderly
The tendency to impaired renal function and low lean body mass in the elderly influences the pharmacokinetics of digoxin such that high serum digoxin levels and associated toxicity can occur quite readily, unless doses of digoxin lower than those in non-elderly patients are used. Serum digoxin levels should be checked regularly and hypokalaemia avoided.
• Dose Recommendations in Specific Patients Groups
See Special warnings and precautions for use – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Contraindications
4.3 Contraindications
DigoKern is contraindicated in intermittent complete heart block or second-degree atrioventricular block, especially if there is a history of Stokes-Adams Syndrome.
DigoKern is contraindicated in arrhythmias originating through intoxication with cardiac glycosides.
DigoKern is contraindicated in supraventricular arrhythmias associated with an atrioventricular accessory pathway, as in the Wolff-Parkinson-White syndrome, unless the electrophysiological characteristics of the accessory pathway and any harmful effects of digoxin on these characteristics have been assessed. If the existence of an accessory pathway is confirmed or suspected, and there is no history of prior supraventricular arrhythmias, DigoKern is similarly contraindicated.
DigoKern is contraindicated in ventricular tachycardia or in ventricular fibrillation.
DigoKern is contraindicated in hypertrophic obstructive cardiomyopathy, unless there is concomitant atrial fibrillation and heart failure, but even then caution should be exercised if digoxin is to be used.
DigoKern is contraindicated in patients known to be hypersensitive to digoxin, to other digitalis glycosides or to any of the excipients listed in Section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
