STRENSIQ SOLUTION FOR INJECTION 100 MG/ML [SIN17126P]
Active ingredients: STRENSIQ SOLUTION FOR INJECTION 100 MG/ML
Product Info
STRENSIQ SOLUTION FOR INJECTION 100 MG/ML
[SIN17126P]
Product information
Active Ingredient and Strength | ASFOTASE ALFA - 100 MG/ML |
Dosage Form | INJECTION, SOLUTION |
Manufacturer and Country | ALEXION PHARMA INTERNATIONAL OPERATIONS LIMITED (AAMF) - IRELAND |
Registration Number | SIN17126P |
Licence Holder | ASTRAZENECA SINGAPORE PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | A16AB13 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
STRENSIQ is indicated for long-term enzyme replacement therapy in patients with pediatric-onset hypophosphatasia (HPP).
Dosing
4.2 Posology and method of administration
Posology
Treatment should be initiated by a physician experienced in the management of patients with metabolic or bone disorders.
STRENSIQ is for subcutaneous injection only.
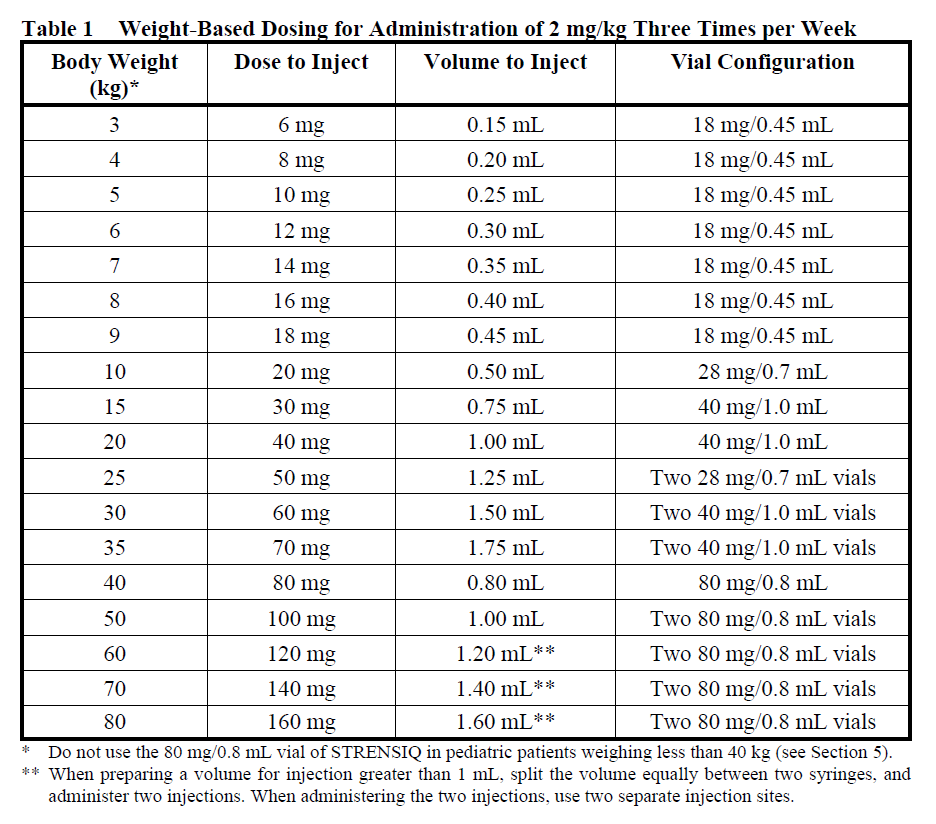
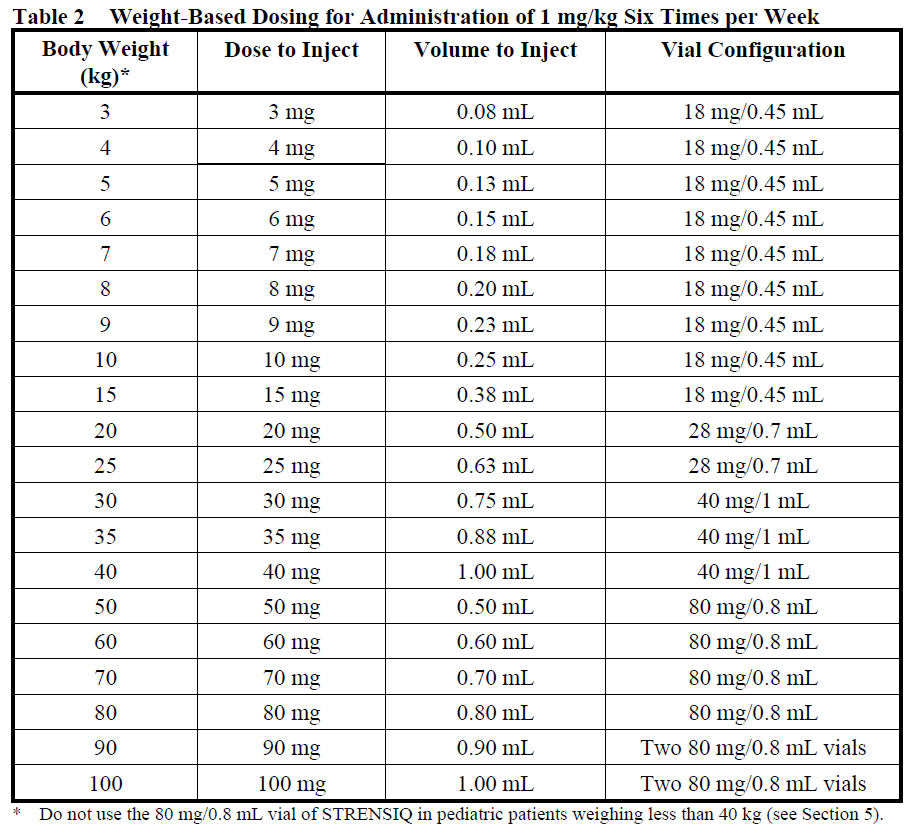
The dosing regimen consists of a total of 6 mg/kg/week of asfotase alfa administered by subcutaneous injection. The recommended dosage regimen of asfotase alfa is 2 mg/kg of body weight administered three times per week or 1 mg/kg of body weight administered six times per week.
Dosage for perinatal/infantile-onset HPP
The recommended dosage regimen of STRENSIQ for the treatment of perinatal/infantile-onset HPP is 6 mg/kg per week administered subcutaneously as either:
2 mg/kg three times per week, or
1 mg/kg six times per week. Injection site reactions may limit the tolerability of the six times per week regimen (see Section 4.8 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information)
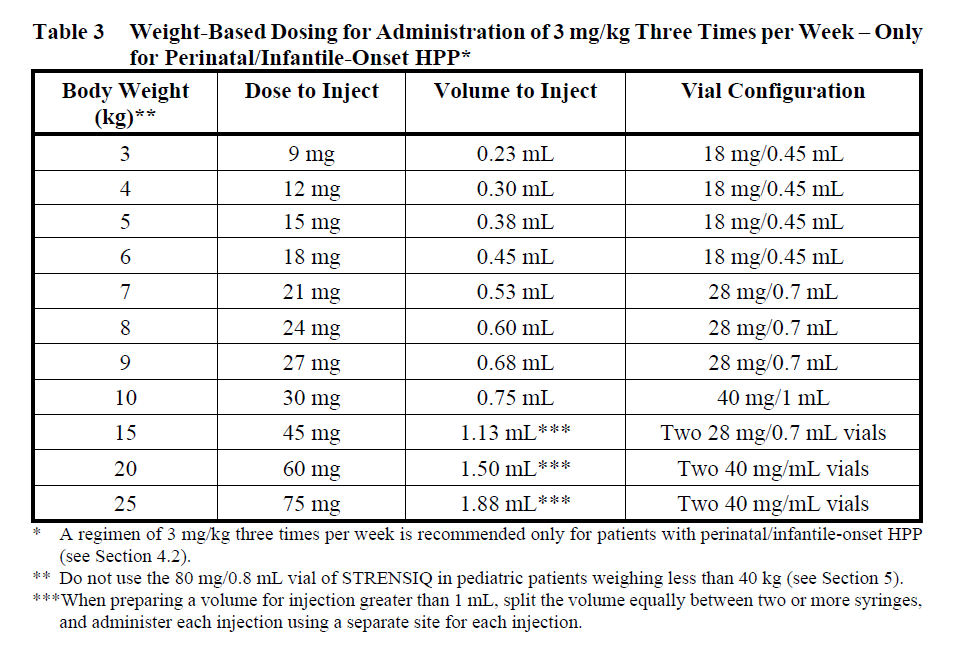
The dose of STRENSIQ may be increased for lack of efficacy (e.g., no improvement in respiratory status, growth, or radiographic findings) up to 9 mg/kg per week administered subcutaneously as 3 mg/kg three times per week.
Dosage for juvenile-onset HPP
The recommended dosage regimen of STRENSIQ for the treatment of juvenile-onset HPP is 6 mg/kg per week administered subcutaneously as either:
2 mg/kg three times per week, or
1 mg/kg six times per week. Injection site reactions may limit the tolerability of the six times per week regimen
Special populations
Pediatric population
The safety and effectiveness of STRENSIQ have been established in pediatric patients. Currently available data are described in Section 5.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Use in the elderly
The safety and efficacy of STRENSIQ in patients older than 65 years have not been established.
Renal and hepatic impairment
The safety and efficacy of STRENSIQ in patients with renal or hepatic impairment have not been evaluated and no specific dose regimen can be recommended for these patients.
Method of administration
STRENSIQ is for subcutaneous injection only. The maximum volume of medication per injection should not exceed 1 mL. If more than 1 mL is required, multiple injections may be administered on the same day, at different sites.
Administer STRENSIQ within 3 hours upon removal of the vial(s) from refrigeration. Take the unopened STRENSIQ vial(s) out of the refrigerator 15 to 30 minutes before injecting to allow the liquid to reach room temperature. Do not warm STRENSIQ in any other way (for example, do not warm it in a microwave or in hot water).
Rotate the injection from among the following sites to reduce the risk of lipodystrophy: abdominal area, thigh, deltoid, or buttocks (see Section 4.4 and Section 4.8 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Do NOT administer injections in areas that are reddened, inflamed, or swollen.
Inject STRENSIQ subcutaneously into the determined site and properly dispose of the syringe and the needle.
STRENSIQ vials are single use only. Discard any unused product.
Preparation for administration
Each vial of STRENSIQ is intended for single use only.
Use aseptic technique.
Determine the total weekly volume needed for the prescribed dosage based on the patient’s weight and recommended dosage. Follow these steps to determine the patient dose:
Total weekly dose (mg) = patient’s weight (kg) x prescribed dose (mg/kg/week)
Total injection volume (mL) per week = Total dose (mg/week) divided by the STRENSIQ concentration (40 mg/mL or 100 mg/mL)
Round total injection volume to the nearest hundredth of a mL
Total number of vials per week = Total injection volume divided by vial volume (mL)
Determine the number of injection days per week (three or six per week).
Determine dose per injection day. Patient weights should be rounded to the nearest kilogram when determining dose. Use the following tables for guidance, for patients administering 2 mg/kg three times per week (Table 1), 1 mg/kg six times per week (Table 2) and for dose escalations to 3 mg/kg three times per week, recommended only for patients with perinatal/infantile-onset HPP (Table 3) (see Section 4.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
When preparing a volume for injection greater than 1 mL, split the volume equally between two or more syringes, and administer each injection using a separate site for each injection.
Inspect the solution in the vial(s) for particulate matter and discoloration. STRENSIQ is supplied as a clear, slightly opalescent or opalescent, colorless to slightly yellow aqueous solution; a few small translucent or white particles may be present. Discard any vials(s) not consistent with this appearance.
Assemble injection supplies. Administer STRENSIQ using sterile disposable 1 mL syringes and ½ inch injection needles, between 25 to 29 gauge are recommended. The use of two different gauge needles is recommended, a larger bore needle (e.g. 25 gauge) for withdrawal of the medication, and a smaller bore needle (e.g. 29 gauge) for the injection. For doses greater than 1 mL, the injection volume should be split equally between two 1 mL syringes. Always use a new syringe and needle for each injection.
Remove vial cap, aseptically prepare the vial and insert the syringe into the vial to withdraw the prescribed dose for administration.
Remove any air bubbles in the syringe and verify the correct dose.
Contraindications
4.3 Contraindications
Severe or life-threatening hypersensitivity to the active substance or to any of the excipients if hypersensitivity is not controllable (see Section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
