LORMALZI CONCENTRATE FOR SOLUTION FOR INFUSION 350MG/20ML [SIN17201P]
Active ingredients: LORMALZI CONCENTRATE FOR SOLUTION FOR INFUSION 350MG/20ML
Product Info
LORMALZI CONCENTRATE FOR SOLUTION FOR INFUSION 350MG/20ML
[SIN17201P]
Product information
Active Ingredient and Strength | DONANEMAB - 350 MG/20 ML |
Dosage Form | INFUSION, SOLUTION CONCENTRATE |
Manufacturer and Country | LILLY FRANCE - FRANCE |
Registration Number | SIN17201P |
Licence Holder | DKSH SINGAPORE PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | N06DX05 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Donanemab is indicated to slow disease progression in adult patients with Alzheimer’s disease (AD). Treatment with donanemab should be initiated in patients with evidence of amyloid beta pathology and either mild cognitive impairment or mild dementia.
Dosing
4.2 Posology and method of administration
Treatment should be initiated by a physician experienced in the diagnosis and treatment of Alzheimer’s disease. The infusion of donanemab should be initiated and supervised by a healthcare professional.
Beta amyloid evidence
Beta amyloid evidence consistent with AD should be confirmed using a validated test.
Posology
The recommended dose of donanemab is 350 mg for the first dose, 700 mg for the second dose, and 1050 mg for the third dose, followed by 1400 mg, every 4 weeks. Treatment should be maintained until amyloid plaques are cleared, as confirmed using a validated method, up to a maximum of 18 months.
Treatment should be continued for up to 18 months if monitoring of amyloid plaque clearance with a validated method is not possible (see section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
The benefit-risk of treatment should be reassessed at regular intervals on an individual basis. Consideration should be given to discontinuing treatment if donanemab would no longer be expected to be effective.
Monitoring and dosing interruption for amyloid related imaging abnormalities
Obtain a recent (within 1 year) brain magnetic resonance imaging (MRI) prior to initiating treatment. Perform an MRI prior to the second dose, prior to the third dose, prior to the fourth dose, and prior to the seventh dose (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
The recommendations for dosing interruptions for patients with amyloid-related imaging abnormalities-oedema/effusions (ARIA-E) and amyloid-related imaging abnormalities haemorrhage/hemosiderin deposition (ARIA-H) are provided in Table 1.
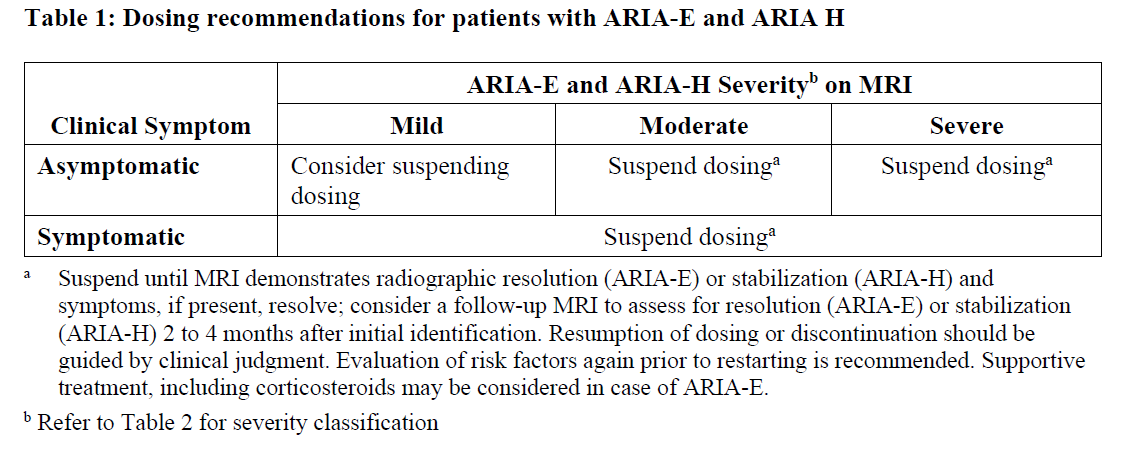
Discontinue donanemab if intracerebral haemorrhage greater than 1 cm occurs.
Method of administration
Lormalzi 350 mg is for intravenous infusion only. Each vial is for single use only. It should be administered over at least 30 minutes. Patients should be observed post-infusion for a minimum of 30 minutes. For instructions on dilution of the medicinal product before administration, see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Parenteral medicinal products should be inspected visually for particulate matter and discolouration prior to administration, whenever solution and container permit. Do not use donanemab if it is cloudy or there are visible particles.
Missed dose
If an infusion is missed, the missed dose should be administered at the next possible occasion. Then, resume the recommended dosing regimen every 4 weeks.
Paediatric population
There is no relevant use of Lormalzi in the paediatric population for the treatment of Alzheimer’s disease.
Contraindications
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
