DAPTOMYCIN/ANFARM POWDER FOR SOLUTION FOR INJECTION/INFUSION 500MG/VIAL [SIN17204P]
Active ingredients: DAPTOMYCIN/ANFARM POWDER FOR SOLUTION FOR INJECTION/INFUSION 500MG/VIAL
Product Info
DAPTOMYCIN/ANFARM POWDER FOR SOLUTION FOR INJECTION/INFUSION 500MG/VIAL
[SIN17204P]
Product information
Active Ingredient and Strength | DAPTOMYCIN - 500 MG/VIAL |
Dosage Form | INJECTION, POWDER, LYOPHILIZED, FOR SOLUTION |
Manufacturer and Country | ANFARM HELLAS S.A - GREECE |
Registration Number | SIN17204P |
Licence Holder | GOLDPLUS UNIVERSAL PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J01XX09 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Daptomycin/Anfarm is indicated for the treatment of the following infections (see sections 4.4 and 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Adult and paediatric (1 to 17 years of age) patients with complicated skin and skin structure infections (cSSSI) caused by susceptible isolates of the following Gram-positive bacteria: Staphylococcus aureus (including methicillin-resistant isolates), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus dysgalactiae subsp. equisimilis, and Enterococcus faecalis (vancomycin-susceptible isolates only).
Adult patients with Staphylococcus aureus bloodstream infections (bacteremia), including those with right-sided infective endocarditis (SAB/RIE), caused by methicillin-susceptible and methicillin-resistant isolates. See sections 4.4 and 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Pediatric patients (1 to 17 years of age) with S. aureus bloodstream infections (bacteremia) caused by methicillin-susceptible and methicillin-resistant isolates.
Consideration should be given to official guidance on the appropriate use of antibacterial agents.
Dosing
4.2 Posology and method of administration
Posology
Adults
Complicated Skin and Skin Structure Infections:
Daptomycin/Anfarm 4 mg/kg is administered to adult patients intravenously in 0.9% sodium chloride for injection once every 24 hours for 7 to 14 days, either by injection over a 2-minute period or by infusion over a 30-minute period. Do not dose Daptomycin/Anfarm more frequently than once a day, and measure creatine phosphokinase (CPK) levels at baseline and at regular intervals (at least weekly). [See 3 INSTRUCTIONS FOR USE, 3.1 Preparation of Daptomycin/Anfarm for Administration – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.]
Staphylococcus aureus Bloodstream Infections (Bacteremia):
Daptomycin/Anfarm 6 mg/kg is administered to adult patients intravenously in 0.9% sodium chloride for injection once every 24 hours for 2 to 6 weeks, either by injection over a 2-minute period or by infusion over a 30-minute period. Duration of treatment is based on the treating physician’s working diagnosis. Do not dose Daptomycin/Anfarm more frequently than once a day, and measure CPK levels at baseline and at regular intervals (at least weekly). [See 3 INSTRUCTIONS FOR USE, 3.1 Preparation of Daptomycin/Anfarm for Administration – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.]
Renal impairment
Daptomycin is eliminated primarily by the kidneys; therefore, an adjustment of Daptomycin/Anfarm dosage interval is recommended for adult patients with creatinine clearance (CLCR) <30 mL/min, including patients receiving hemodialysis or continuous ambulatory peritoneal dialysis (CAPD). The recommended dosing regimen for these adult patients is 4 mg/kg (cSSSI) or 6 mg/kg (S. aureus bloodstream infections) once every 48 hours. When possible, administer Daptomycin/Anfarm following the completion of hemodialysis on hemodialysis days. In adult patients with renal impairment, monitor both renal function and CPK more frequently than once weekly. No dosage interval adjustment is required for adult patients with CLCR ≥30 mL/min.
Due to limited clinical experience (see table and footnotes below) Daptomycin/Anfarm should only be used in adult patients with any degree of renal impairment (CrCl < 80 ml/min) when it is considered that the expected clinical benefit outweighs the potential risk. The response to treatment, renal function and creatine phosphokinase (CPK) levels should be closely monitored in all patients with any degree of renal impairment (see also sections 4.4 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). The dosage regimen for daptomycin in paediatric patients with renal impairment has not been established.
Dose adjustments in adult patients with renal impairment by indication and creatinine clearance
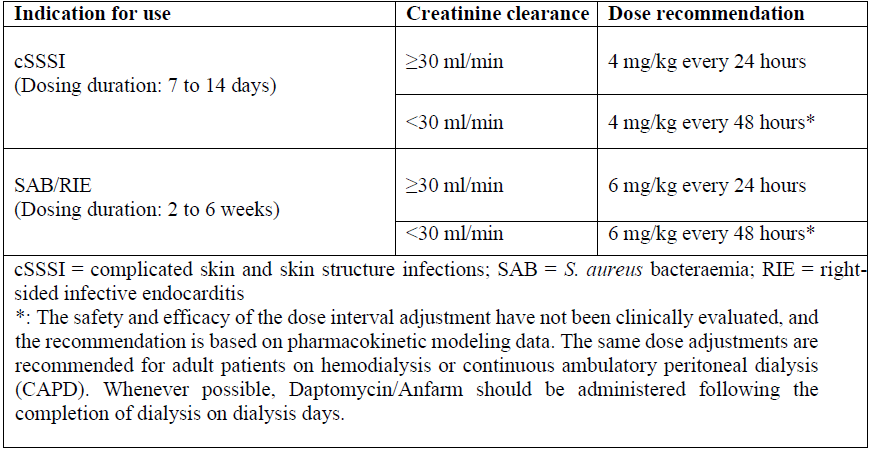
The dosage regimen for daptomycin in pediatric patients with renal impairment has not been established.
Hepatic impairment
No dose adjustment is necessary when administering Daptomycin/Anfarm to patients with mild or moderate hepatic impairment (Child–Pugh Class B) (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). No data are available in patients with severe hepatic impairment (Child–Pugh Class C).
Elderly patients
No adjustment of daptomycin dosage is warranted for elderly patients with CLCR ≥30 mL/min.
Paediatric population (1 to 17 years of age)
The recommended dosage regimens for paediatric patients based on age and indication are shown below.
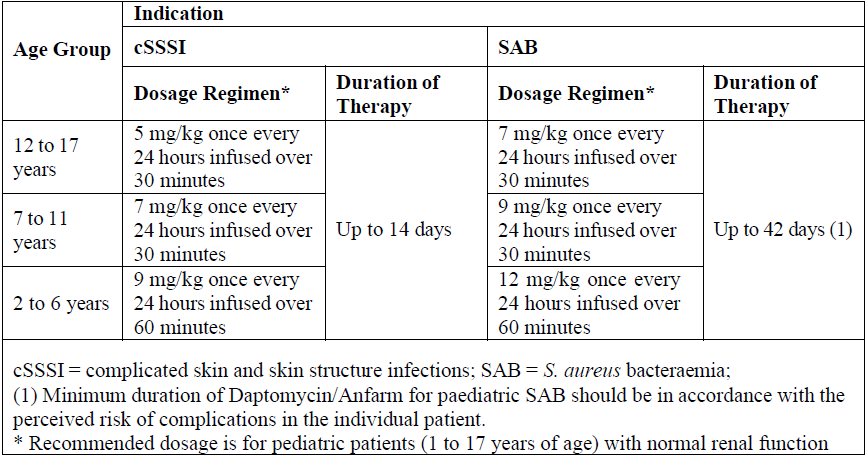
Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established.
Daptomycin/Anfarm is administered intravenously in 0.9% sodium chloride (see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). Daptomycin/Anfarm should not be used more frequently than once a day.
Creatine phosphokinase (CPK) levels must be measured at baseline and at regular intervals (at least weekly) during treatment (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Paediatric patients below the age of one year should not be given Daptomycin/Anfarm due to the risk of potential effects on muscular, neuromuscular and/or nervous systems (either peripheral and/or central) that were observed in neonatal dogs (see section 5.3 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Method of administration
In adults, Daptomycin/Anfarm is given by intravenous infusion (see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information) and administered over a 30-minute period or by intravenous injection (see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information) and administered over a 2-minute period.
In paediatric patients aged 1 to 17 years, Daptomycin/Anfarm is given by intravenous infusion over a 30-minute or 60-minute period (see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
For IV infusion over a period of 30 minutes in pediatric patients, reconstituted Daptomycin/Anfarm (concentration of 50 mg/mL) is further diluted, using aseptic technique, into a 50 mL IV infusion bag containing 0.9% sodium chloride for injection. The infusion rate should be maintained at 1.67 mL/min over the 30 minute period.
For IV infusion over a period of 60 minutes in pediatric patients, reconstituted Daptomycin/Anfarm (concentration of 50 mg/mL) is further diluted, using aseptic technique, into an IV infusion bag containing 25 mL of 0.9% sodium chloride for injection. The infusion rate should be maintained at 0.42 mL/min over the 60 minute period.
Unlike in adults, Daptomycin/Anfarm should not be administered by injection over a two (2) minute period in pediatric patients
For instructions on reconstitution and dilution of the medicinal product before administration, see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Contraindications
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
