LOQTORZI CONCENTRATE FOR SOLUTION FOR INFUSION 240MG/6ML [SIN17212P]
Active ingredients: LOQTORZI CONCENTRATE FOR SOLUTION FOR INFUSION 240MG/6ML
Product Info
LOQTORZI CONCENTRATE FOR SOLUTION FOR INFUSION 240MG/6ML
[SIN17212P]
Product information
Active Ingredient and Strength | TORIPALIMAB - 240 MG/6 ML |
Dosage Form | INFUSION, SOLUTION CONCENTRATE |
Manufacturer and Country | SUZHOU UNION BIOPHARM CO., LTD - CHINA |
Registration Number | SIN17212P |
Licence Holder | TA BIOSCIENCES PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01FF13 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 THERAPEUTIC INDICATIONS
LOQTORZI, in combination with cisplatin and gemcitabine, is indicated for the first-line treatment of adult patients with recurrent, not amenable to surgery or radiotherapy, or metastatic nasopharyngeal carcinoma.
Dosing
4.2 DOSE AND METHOD OF ADMINISTRATION
The recommended dosing regimen of LOQTORZI is 240 mg every 3 weeks (Q3W). Treatment should continue until disease progression, unacceptable toxicity or up to a maximum duration of 24 months.
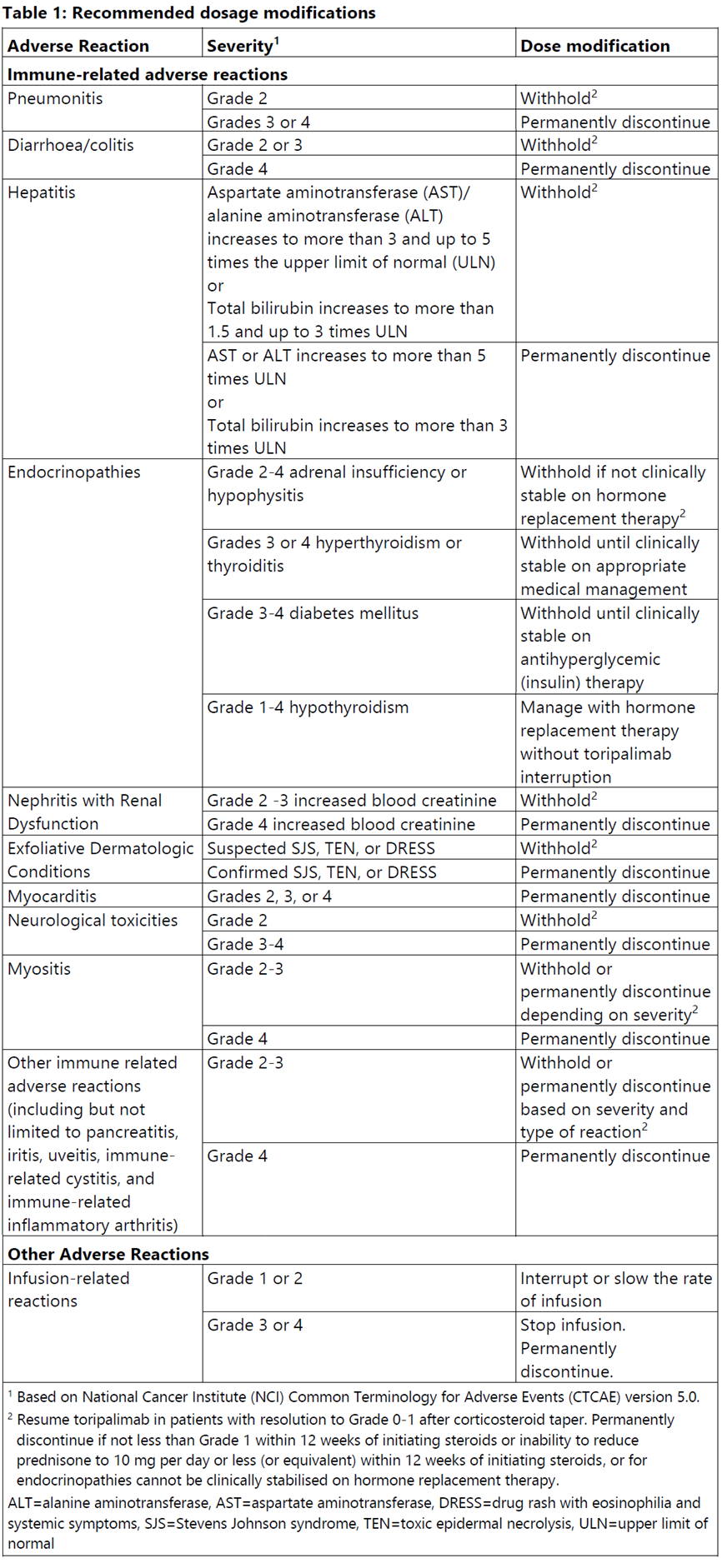
Dosage modifications
No dose reductions of LOQTORZI are recommended. Withold or discontinue LOQTORZI to manage adverse reactions as described in Table 1.
Administration
Administer diluted solution intravenously via an infusion pump using an in-line aseptic filter (0.2 micron or 0.22 micron pore size).
First Infusion: Infuse over at least 60 minutes.
Subsequent infusions: If no infusion-related reactions occurred during the first infusion, subsequent infusions may be administered over 30 minutes. (See Dosage modifications)
Do not co-administer other medicines through the same intravenous line.
When administered on the same day as chemotherapy, LOQTORZI should be administered prior to chemotherapy.
Refer to the Product Information for cisplatin and gemcitabine for recommended dosing information.
Preparation for intravenous infusion
Visually inspect the solution for particulate matter and discoloration. The solution is clear to slightly opalescent, colourless to slightly yellow. Discard the vial if visible particles are observed.
Withdraw the required volume of LOQTORZI and inject slowly into a 100 mL or 250 mL infusion bag containing sodium chloride 9 mg/mL (0.9%) solution for injection for infusion using aseptic technique. Mix diluted solution by gentle inversion. Do not shake. The final concentration of the diluted solution should be between 1 mg/mL to 3 mg/mL.
Each vial is for use in one patient on one occasion only. Discard any unused portion left in the vial.
Refer to section 6.4 Special precautions for storage, if the diluted solution is not administered immediately – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Special populations
Elderly
No dose adjustment is recommended for patients who are 65 years of age or older (see section 5.2 Pharmacokinetic properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal impairment
No dose adjustment is needed for patients with mild or moderate renal impairment. There are no data in patients with severe renal impairment for dosing recommendations (see section 5.2 Pharmacokinetic properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Hepatic impairment
No dose adjustment is recommended for patients with mild hepatic impairment. There are no data in patients with moderate or severe hepatic impairment for dosing recommendations (see section 5.2 Pharmacokinetic properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Paediatric population
The safety and efficacy of toripalimab in children and adolescents under 18 years of age have not been established. No data are available.
Contraindications
4.3 CONTRAINDICATIONS
Severe hypersensitivity to the active ingredient or to any excipients listed in section 6.1 List of excipients – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
