DATROWAY POWDER FOR CONCENTRATE FOR SOLUTION FOR INFUSION 100MG/VIAL [SIN17234P]
Active ingredients: DATROWAY POWDER FOR CONCENTRATE FOR SOLUTION FOR INFUSION 100MG/VIAL
Product Info
DATROWAY POWDER FOR CONCENTRATE FOR SOLUTION FOR INFUSION 100MG/VIAL
[SIN17234P]
Product information
Active Ingredient and Strength | DATOPOTAMAB DERUXTECAN - 100 MG/VIAL |
Dosage Form | INJECTION, POWDER, LYOPHILIZED, FOR SOLUTION |
Manufacturer and Country | BAXTER ONCOLOGY GMBH - GERMANY |
Registration Number | SIN17234P |
Licence Holder | ASTRAZENECA SINGAPORE PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | PENDING |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1. Therapeutic Indications
Breast Cancer
DATROWAY is indicated for the treatment of adult patients with unresectable or metastatic hormone receptor (HR)-positive, HER2-negative (IHC 0, IHC 1+ or IHC 2+/ISH−) breast cancer who have received prior endocrine-based therapy and chemotherapy for unresectable or metastatic disease.
Dosing
4.2. Posology and Method of Administration
Premedication and Prophylactic Medications
Prior to each infusion of DATROWAY, a premedication regimen for the prevention of infusion-related reactions that consists of an antihistamine agent and acetaminophen (with or without glucocorticoids) should be considered.
It is also recommended that patients receive prophylactic antiemetic agents (dexamethasone with 5-HT3 antagonists as well as other medicinal products, such as NK1 receptor antagonists), prior to infusion of DATROWAY and on subsequent days as needed.
For prophylactic treatment for keratitis and stomatitis, please refer to Special Warnings and Precautions for Use (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Posology
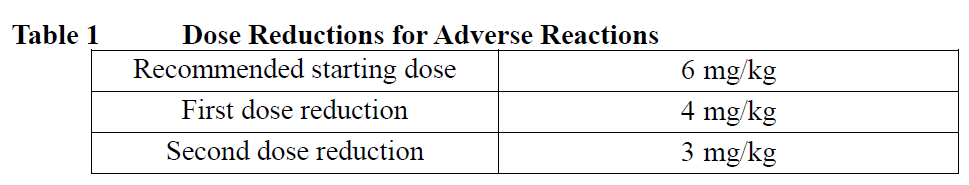
The recommended dose of DATROWAY is 6 mg/kg given as an intravenous infusion once every three weeks (21-day cycle) until disease progression or unacceptable toxicity.
First infusion: Administer infusion over 90 minutes. Observe patients during the infusion and for at least 30 minutes following the initial dose, for signs or symptoms of infusion-related reactions.
Subsequent infusions: Administer infusion over 30 minutes if prior infusions were tolerated. Observe patients during the infusion and for at least 30 minutes after infusion.
Dose Modifications
Dose modifications for infusion-related reactions
Slow or interrupt the infusion rate if the patient develops an infusion-related reaction. Permanently discontinue DATROWAY for life-threatening infusion-related reactions.
Dose modifications for adverse reactions
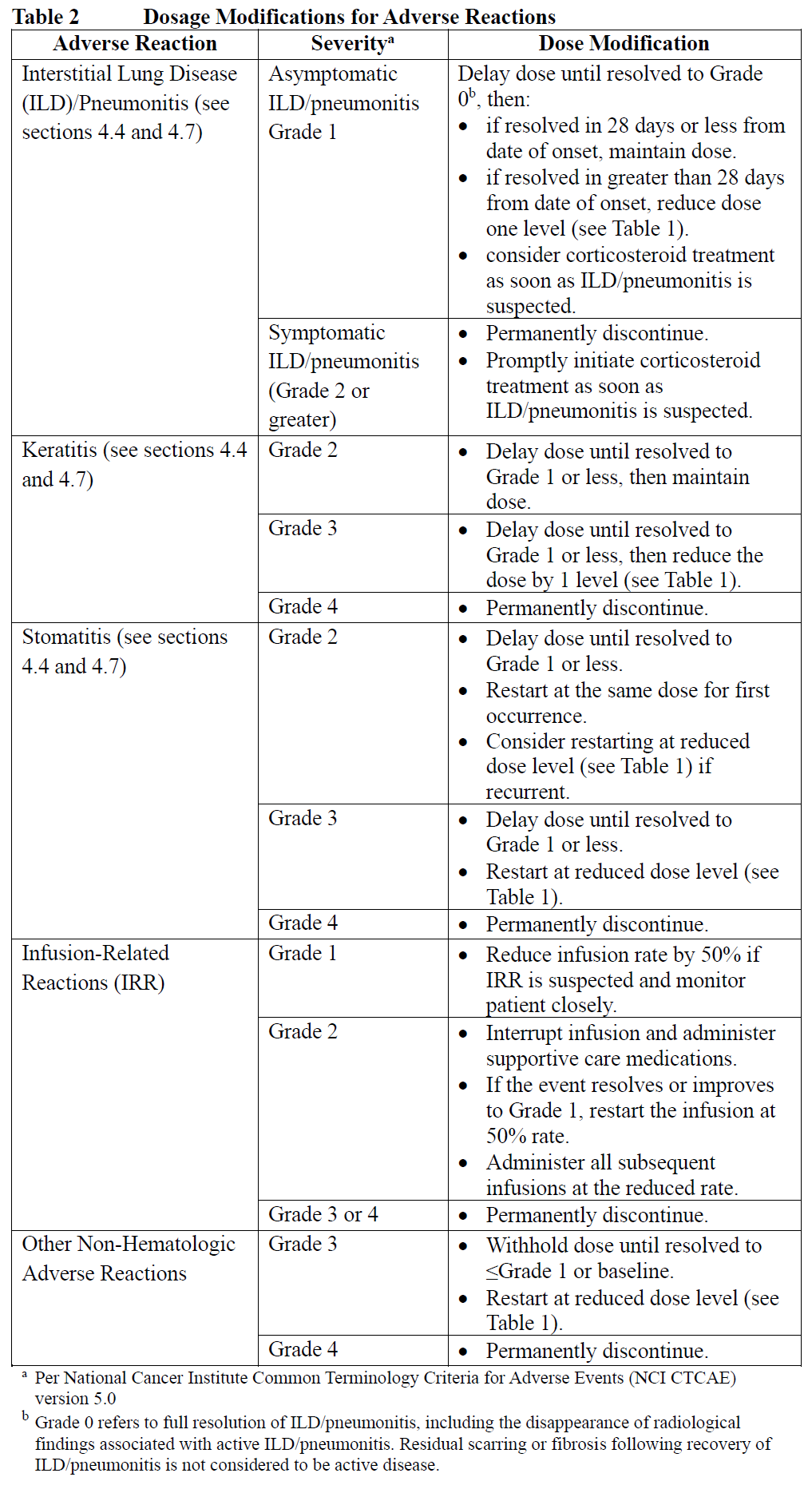
Management of adverse reactions may require dose delay, dose reduction, or treatment discontinuation per guidelines provided in Tables 1 and 2.
Do not re-escalate the DATROWAY dose after a dose reduction is made. After the dose reductions, if further toxicity meeting the requirement for dose reduction occurred, permanently discontinue DATROWAY.
Permanently discontinue for corneal perforation.
Delayed or Missed Dose
If a planned dose is delayed or missed, it should be administered as soon as possible without waiting until the next planned cycle. The schedule of administration should be adjusted to maintain a 3-week interval between doses.
Special Populations
Geriatrics
No dose adjustment of DATROWAY is required in patients aged 65 years or older.
Of 365 patients in TROPION-Breast01 randomized to DATROWAY 6 mg/kg, 24.9% were 65 years of age or older. No overall differences in efficacy were observed between patients ≥65 years of age and younger patients.
Of the 927 patients treated with DATROWAY 6 mg/kg in the clinical studies, 32.8% were 65 years of age or older. No clinically meaningful differences in safety were observed between patients ≥65 years of age and younger patients.
Population pharmacokinetic analysis indicates that age does not have a clinically meaningful effect on the pharmacokinetics of datopotamab deruxtecan.
Pediatrics
The safety and efficacy in children and adolescents below 18 years of age have not been established.
Renal Impairment
No dose adjustment is required in patients with mild to moderate (creatinine clearance [CLcr] 30 to <90 ml/min) renal impairment. There are limited data to make a recommendation on dose adjustment in patients with severe renal impairment.
Hepatic Impairment
No dose adjustment is required in patients with mild (total bilirubin ≤ULN and any AST >ULN or total bilirubin >1 to 1.5 times ULN and any AST) hepatic impairment. There are limited data to make a recommendation on dose adjustment in patients with moderate (total bilirubin >1.5 to 3 times ULN and any AST) and severe (total bilirubin >3 times ULN and any AST) hepatic impairment.
Method of Administration
DATROWAY is for intravenous use. It must be reconstituted and diluted by a healthcare professional and administered as an intravenous infusion. DATROWAY must not be administered as an intravenous push or bolus.
For instructions on reconstitution and dilution of DATROWAY before administration, see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Contraindications
4.3. Contraindications
None.
