TALZENNA CAPSULE 0.5MG [SIN17240P]
Active ingredients: TALZENNA CAPSULE 0.5MG
Product Info
TALZENNA CAPSULE 0.5MG
[SIN17240P]
Product information
Active Ingredient and Strength | TALAZOPARIB TOSYLATE EQV TALAZOPARIB - 0.5 MG |
Dosage Form | CAPSULE |
Manufacturer and Country | EXCELLA GMBH & CO. KG - GERMANY |
Registration Number | SIN17240P |
Licence Holder | PFIZER PRIVATE LIMITED |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01XK04 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1. Therapeutic indications
Breast cancer
TALZENNA is indicated for the treatment of adult patients with germline breast cancer susceptibility gene (BRCA)-mutated (gBRCAm) human epidermal growth factor receptor 2 (HER2)-negative locally advanced or metastatic breast cancer who have previously been treated with chemotherapy. These patients could have received chemotherapy in the neoadjuvant, adjuvant, locally advanced or metastatic setting unless patients were not suitable for these treatments.
Prostate cancer
TALZENNA is indicated in combination with enzalutamide for the treatment of adult patients with metastatic castration-resistant prostate cancer (mCRPC) in whom chemotherapy is not clinically indicated.
Dosing
4.2. Posology and method of administration
Treatment with TALZENNA should be initiated and supervised by a physician experienced in the use of anticancer medicinal products.
Treatment of gBRCAm HER2-negative locally advanced or metastatic breast cancer
Detection of mutations in hereditary breast cancer-related BRCA1 and BRCA2 genes should be determined by an experienced laboratory using a validated test method (see Section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Recommended dosing for gBRCAm HER2-negative locally advanced or metastatic breast cancer
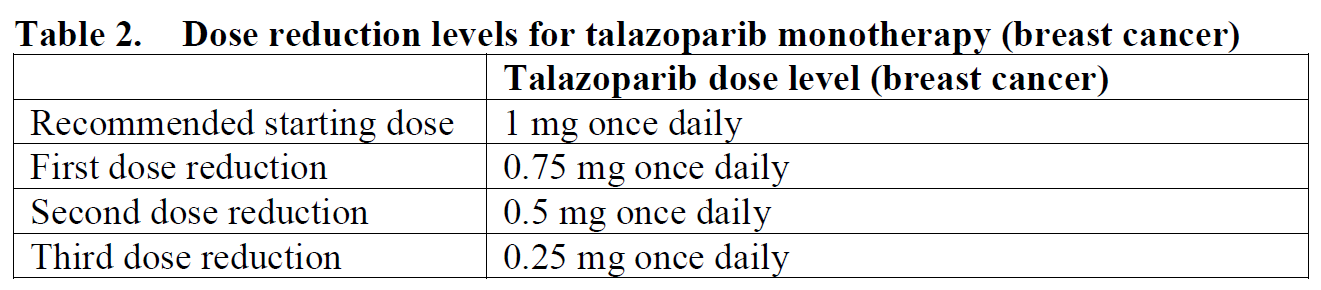
The recommended dose of TALZENNA is 1 mg capsule taken orally once daily. Patients should be treated until disease progression or unacceptable toxicity occurs.
Treatment of mCRPC
Recommended dosing for mCRPC
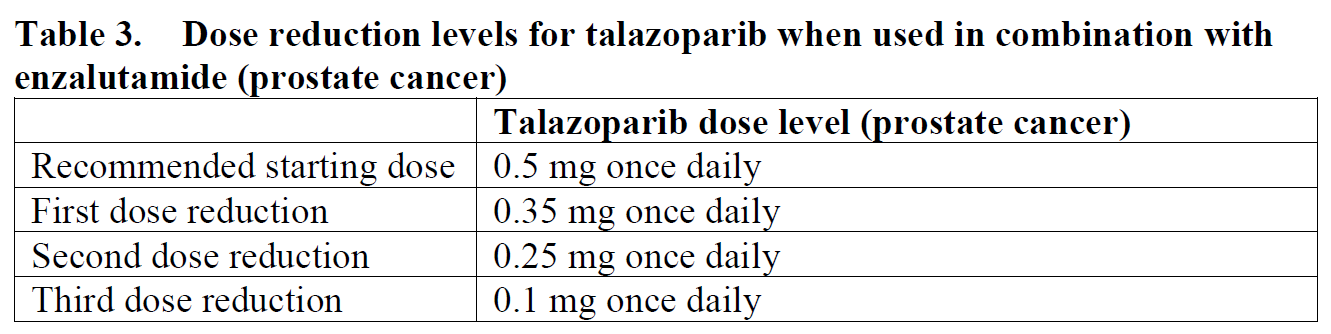
The recommended dose of TALZENNA is 0.5 mg administered orally once daily in combination with enzalutamide 160 mg orally once daily. Patients should be treated until disease progression or unacceptable toxicity occurs.
Patients receiving TALZENNA and enzalutamide should also receive a gonadotropin-releasing hormone (GnRH) analog concurrently or should have had bilateral orchiectomy.
The 0.35 mg, 0.25 mg and 0.1 mg capsules are available for dose reduction.
Missing dose
TALZENNA may be taken with or without food. If the patient vomits or misses a dose, an additional dose should not be taken. The next prescribed dose should be taken at the usual time.
Dose modifications and reductions
To manage adverse reactions, consider interruption of treatment or dose reduction based on severity and clinical presentation. Recommended dose reductions are indicated in Tables 2 and 3.
Complete blood count should be obtained prior to starting TALZENNA therapy and monitored monthly and as clinically indicated (see Table 1 and Section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).

gBRCAm HER2-negative locally advanced or metastatic breast cancer
mCRPC
Please refer to the enzalutamide prescribing information for dose modifications for adverse reactions associated with enzalutamide.
Concomitant treatment with inhibitors of P-glycoprotein (P-gp)
gBRCAm HER2-negative locally advanced or metastatic breast cancer
Strong inhibitors of P-gp may lead to increased talazoparib exposure. Concomitant use of strong P-gp inhibitors during treatment with talazoparib should be avoided. Coadministration should only be considered after careful evaluation of the potential benefits and risks. If coadministration with a strong P-gp inhibitor is unavoidable, the TALZENNA dose should be reduced to the next lower dose. When the strong P-gp inhibitor is discontinued, the TALZENNA dose should be increased (after 3 to 5 half-lives of the P-gp inhibitor) to the dose used prior to the initiation of the strong P-gp inhibitor (see Section 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
mCRPC
The effect of coadministration of P-gp inhibitors on talazoparib exposure when talazoparib is given in combination with enzalutamide has not been studied. Therefore, concomitant use of P-gp inhibitors during treatment with talazoparib should be avoided (see Section 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Concomitant treatment with inhibitors of Breast Cancer Resistance Protein (BCRP)
The effect of coadministration of BCRP inhibitors with TALZENNA has not been studied. Therefore, concomitant use of strong BCRP inhibitors during treatment with talazoparib should be avoided (see Section 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Special populations
Hepatic impairment
No dose adjustment is required for patients with mild hepatic impairment (total bilirubin ≤1 × upper limit of normal [ULN] and aspartate aminotransferase (AST) >ULN, or total bilirubin >1.0 to 1.5 × ULN and any AST), moderate hepatic impairment (total bilirubin >1.5 to 3.0 × ULN and any AST), or severe hepatic impairment (total bilirubin >3.0 × ULN and any AST) (see Section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). TALZENNA in combination with enzalutamide is not recommended for use in patients with severe hepatic impairment (Child-Pugh classification C), as pharmacokinetics and safety have not been established in these patients (see Section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal impairment
gBRCAm HER2-negative locally advanced or metastatic breast cancer
No dose adjustment is required for patients with mild renal impairment (60 mL/min ≤ creatinine clearance [CrCL] < 90 mL/min). For patients with moderate renal impairment (30 mL/min ≤ CrCL < 60 mL/min), the recommended dose of TALZENNA is 0.75 mg once daily. For patients with severe renal impairment (15 mL/min ≤ CrCL < 30 mL/min), the recommended dose of TALZENNA is 0.5 mg once daily. TALZENNA has not been studied in patients requiring hemodialysis (see Section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
mCRPC
No dose adjustment is necessary for patients with mild renal impairment (60 mL/min ≤ creatinine clearance [CrCL] <90 mL/min). For patients with moderate renal impairment (30 mL/min ≤ CrCL <60 mL/min), the recommended dose of TALZENNA is 0.35 mg once daily in combination with enzalutamide orally once daily. For patients with severe renal impairment (15 mL/min ≤ CrCL <30 mL/min), the recommended dose of TALZENNA is 0.25 mg once daily in combination with enzalutamide once daily. TALZENNA has not been studied in patients with CrCL <15 mL/min or patients requiring hemodialysis (see Section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Elderly population
No dose adjustment is necessary in elderly (≥65 years of age) patients (see Section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Pediatric population
The safety and efficacy of TALZENNA in children and adolescents <18 years of age have not been established.
Contraindications
4.3. Contraindications
None.
