ZERPIDIO (SERPLULIMAB) CONCENTRATE FOR SOLUTION FOR INFUSION 100MG/10ML [SIN17246P]
Active ingredients: ZERPIDIO (SERPLULIMAB) CONCENTRATE FOR SOLUTION FOR INFUSION 100MG/10ML
Product Info
ZERPIDIO (SERPLULIMAB) CONCENTRATE FOR SOLUTION FOR INFUSION 100MG/10ML
[SIN17246P]
Product information
Active Ingredient and Strength | SERPLULIMAB - 100 MG/10 ML |
Dosage Form | INJECTION, SOLUTION, CONCENTRATE |
Manufacturer and Country | SHANGHAI HENLIUS BIOPHARMACEUTICAL CO. LTD. - CHINA |
Registration Number | SIN17246P |
Licence Holder | INNOGENE KALBIOTECH PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01FF12 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Therapeutic Indications
Zerpidio in combination with carboplatin and etoposide is indicated for the first-line treatment of adult patients with extensive-stage small cell lung cancer (ES-SCLC).
Dosing
Posology and Method of Administration
Treatment must be initiated and supervised by physicians experienced in the treatment of cancer.
Posology
The recommended dose of Zerpidio is 4.5 mg/kg every 3 weeks or 300 mg every 3 weeks* until disease progression or unacceptable toxicity.
Atypical responses (i.e., an initial transient increase in tumour size or small new lesions within the first few months of treatment, followed by tumour shrinkage) have been observed. It is recommended to continue treatment for clinically stable patients with initial evidence of disease progression until disease progression is confirmed.
Dose escalation or reduction of Zerpidio is not recommended. Dose withholding or discontinuation may be required based on individual safety and tolerability.
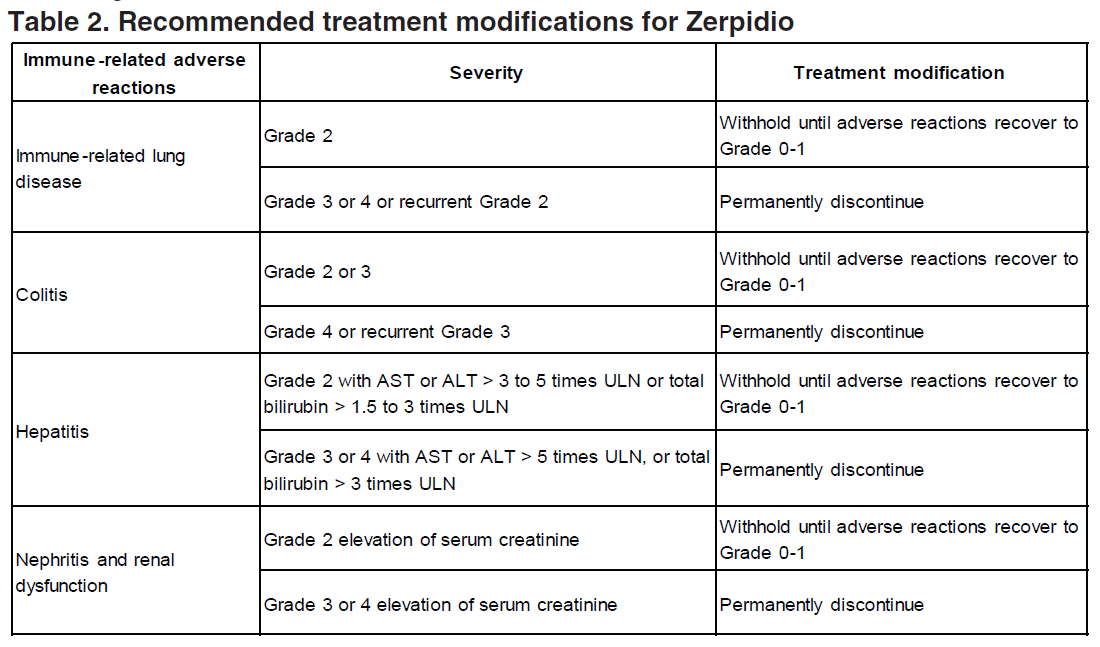
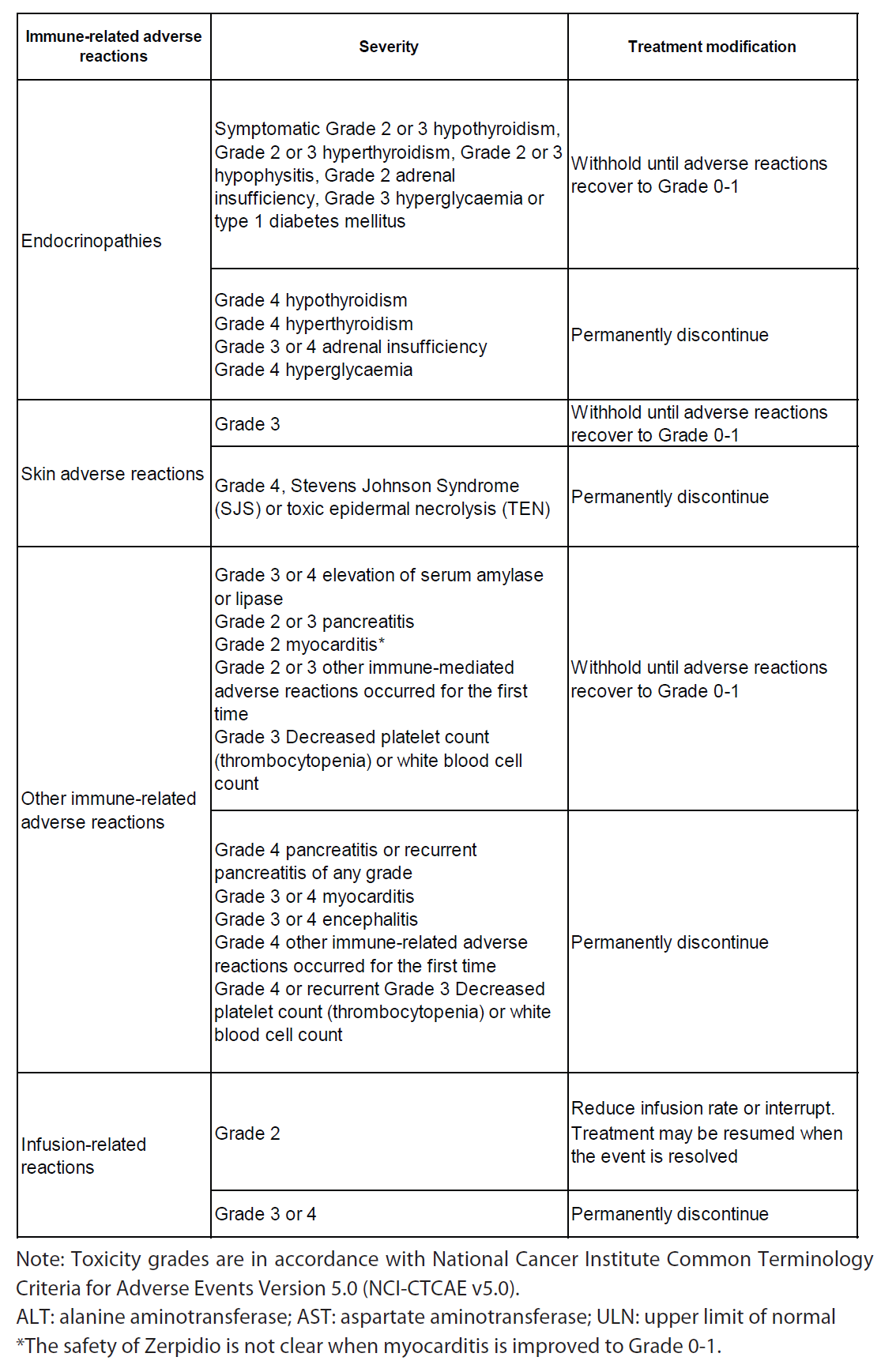
Guidelines for management of immune-related adverse reactions are described in Table 2 (see section Special Warnings and Precautions for Use – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
*The recommended alternative flat dosing of 300 mg every 3 weeks has not been investigated in patients with ES-SCLC and is based on population pharmacokinetic modelling and simulations.
Special Populations
Paediatric population
The safety and efficacy of Zerpidio in children and adolescents below 18 years of age have not been established. No data are available.
Elderly
No dose adjustment is required for elderly patients (≥ 65 years). Zerpidio must be administered with caution in this population.
Renal impairment
No dose adjustment is needed for patients with mild (CRCL=60–89mL/min) or moderate (CRCL=30–59 mL/min) renal impairment. There are insufficient data in patients with severe (CRCL=15–29 mL/min) renal impairment for dosing recommendations.
Hepatic impairment
No dose adjustment is needed for patients with mild (BIL ≤ ULN and AST > ULN or BIL > 1 to 1.5 × ULN and any AST*) hepatic impairment. There are insufficient data in patients with moderate (BIL > 1.5 to 3 × ULN and any AST*) hepatic impairments and no data are available in severe (BIL > 3 × ULN and any AST*) hepatic impairments for dosing recommendations.
*Hepatic impairment is not defined solely based on BIL and AST levels.
Method of administration
Zerpidio is for intravenous use.
The initial infusion rate should be set up to 100 mL per hour. If the first infusion is well tolerated, all subsequent infusions may be shortened to 30 minutes.
Preparation and administration
Confirm the dose of the product and calculate the required volume of Zerpidio.
To make a total volume of 100 mL, use a sterile syringe to extract the normal saline (100 mL of 0.9% sodium chloride solution) at the volume which equals to the required Zerpidio volume and discard it.
Use a syringe to withdraw the required volume of Zerpidio from the vial and inject it into normal saline.
Set the initial infusion rate to 100 mL per hour (25 drops per minute is recommended, and the infusion rate can be adjusted if infusion related reactions occur). If there is no infusion related adverse reactions in the first infusion, the duration of subsequent administration can be shortened to 30 minutes.
At the end of infusion, the infusion tube is flushed with normal saline according to the routine operation procedure of the hospital.
Any unused medicinal product or waste material should be disposed of in accordance with local requirements.
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products. Zerpidio should not be infused concomitantly in the same intravenous line with other medicinal products.
Diluted solution
From a microbiological point of view, the prepared solution for infusion should be used immediately. If not used immediately, in-use storage times and conditions prior to use are the responsibility of the user and would normally no longer than 24 hours at 2 °C–8 °C. The 24-hour hold may include up to 6 hours at room temperature (≤ 25°C). Aseptic handling should be ensured during the preparation of infusion.
Contraindications
Contraindications
Hypersensitivity to active substance or to any of the excipients listed in section List of excipients – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
