MICAFUNGIN PHARMAZAC POWDER FOR CONCENTRATE FOR SOLUTION FOR INFUSION 50MG/VIAL [SIN17264P]
Active ingredients: MICAFUNGIN PHARMAZAC POWDER FOR CONCENTRATE FOR SOLUTION FOR INFUSION 50MG/VIAL
Product Info
MICAFUNGIN PHARMAZAC POWDER FOR CONCENTRATE FOR SOLUTION FOR INFUSION 50MG/VIAL
[SIN17264P]
Product information
Active Ingredient and Strength | MICAFUNGIN SODIUM EQUIVALENT TO MICAFUNGIN - 50 MG/VIAL |
Dosage Form | INJECTION, POWDER, LYOPHILIZED, FOR SOLUTION |
Manufacturer and Country | ROMPHARM COMPANY SRL - ROMANIA |
Registration Number | SIN17264P |
Licence Holder | SCIGEN PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J02AX05 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Micafungin Pharmazac is indicated for:
Adults and paediatric patients 4 months and older for:
treatment of invasive candidiasis
treatment of oesophageal candidiasis in patients for whom intravenous therapy is appropriate.
prophylaxis of Candida infection in patients undergoing allogeneic haematopoietic stem cell transplantation or patients who are expected to have neutropenia (absolute neutrophil count < 500 cells/microlitre) for 10 or more days.
Micafungin has not been adequately studied in patients with endocarditis, osteomyelitis, and meningitis due to Candida infections.
The decision to use Micafungin should take into account a potential risk for the development of liver tumors. Micafungin should therefore only be used if other antifungals are not appropriate.
Dosing
4.2 Posology and method of administration
Treatment with Micafungin Pharmazac should be initiated by a physician experienced in the management of fungal infections.
Posology
Consideration should be given to official/national guidance on the appropriate use of antifungal agents. Treatment with Micafungin should be initiated by a physician experienced in the management of fungal infections.
Specimens for fungal culture and other relevant laboratory studies (including histopathology) should be obtained prior to therapy to isolate and identify causative organism(s). Therapy may be instituted before the results of the cultures and other laboratory studies are known. However, once these results become available, antifungal therapy should be adjusted accordingly.
Micafungin should be administered once daily by intravenous infusion. The dosage depends on the indication and body weight of the patient as shown in the table below.
Use in adults, adolescents ≥ 16 years of age and elderly

A loading dose is not required. Typically, 85% of the steady-state concentration is achieved after three daily Micafungin doses.
Treatment duration
Invasive candidiasis: The treatment duration of Candida infection should be a minimum of 14 days. The antifungal treatment should continue for at least one week after two sequential negative blood cultures have been obtained and after resolution of clinical signs and symptoms of infection.
Oesophageal candidiasis: Micafungin should be administered for at least one week after resolution of clinical signs and symptoms.
Prophylaxis of Candida infections: Micafungin should be administered for at least one week after neutrophil recovery.
Dosage for paediatric patients 4 months and older
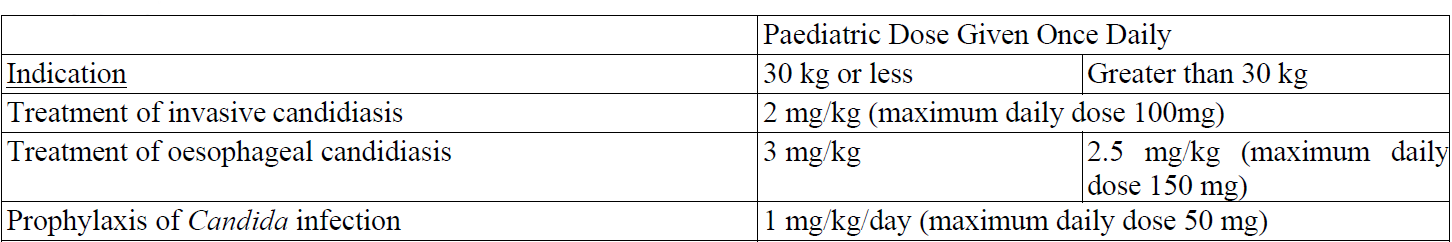
Hepatic impairment
No dose adjustment is necessary in patients with mild or moderate hepatic impairment (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). There are currently insufficient data available for the use of micafungin in patients with severe hepatic impairment and its use is not recommended in these patients (see sections 4.4 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal impairment
No dose adjustment is necessary in patients with renal impairment (creatinine clearance < 30mL/min) (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Method of administration
For intravenous use.
After reconstitution and dilution, the solution should be administered by intravenous infusion over approximately 1 hour. More rapid infusions may result in more frequent histamine mediated reactions. For reconstitution instructions see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Contraindications
4.3 Contraindications
Hypersensitivity to the active substance, to other echinocandins or to any of the excipients listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
