STEQEYMA® SOLUTION FOR INJECTION IN PRE-FILLED SYRINGE 90MG/1ML [SIN17309P]
Active ingredients: STEQEYMA® SOLUTION FOR INJECTION IN PRE-FILLED SYRINGE 90MG/1ML
Product Info
STEQEYMA® SOLUTION FOR INJECTION IN PRE-FILLED SYRINGE 90MG/1ML
[SIN17309P]
Product information
Active Ingredient and Strength | USTEKINUMAB - 90 MG/ML |
Dosage Form | INJECTION, SOLUTION |
Manufacturer and Country | CELLTRION PHARM, INC. - KOREA, REPUBLIC OF |
Registration Number | SIN17309P |
Licence Holder | CELLTRION HEALTHCARE SINGAPORE PRIVATE LIMITED |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L04AC05 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Indications
Plaque Psoriasis:
STEQEYMA® is indicated for the treatment of adult patients (18 years or older) with moderate to severe plaque psoriasis who failed to respond to, or who have a contraindication to, or are intolerant to other systemic therapies including cyclosporin, methotrexate or PUVA.
Pediatric Plaque Psoriasis:
STEQEYMA® is indicated for the treatment of pediatric patients (children and adolescents) (6 years and older) with moderate to severe plaque psoriasis who are inadequately controlled by, or are intolerant to, other systemic therapies or phototherapies.
Psoriatic Arthritis (PsA):
STEQEYMA®, alone or in combination with methotrexate (MTX), is indicated for:
the treatment of adult patients (18 years or older) with active psoriatic arthritis when the response to previous non-biological disease-modifying anti-rheumatic drug (DMARD) therapy has been inadequate
inhibiting the progression of structural damage
Crohn’s Disease:
STEQEYMA® is indicated for the treatment of adults with moderately to severe active Crohn’s disease who have:
failed or were intolerant to treatment with immunomodulators or corticosteroids, but never failed treatment with a tumor necrosis factor (TNF) blocker or failed or were intolerant to treatment with one or more TNF blockers
Dosing
Dosage and Administration
Dosage – Adults
Plaque Psoriasis
For the treatment of plaque psoriasis, ustekinumab is administered by subcutaneous injection. Ustekinumab is intended for use under the guidance and supervision of a physician experienced in the diagnosis and treatment of psoriasis.
For patients weighing ≤100 kg, the recommended dose is 45 mg initially and 4 weeks later, followed by dosing every 12 weeks.
For patients weighing >100 kg, alternatively a dose of 90 mg initially and 4 weeks later, followed by dosing every 12 weeks may be used.
In patients weighing >100 kg, 45mg was also shown to be efficacious. However, 90mg resulted in greater efficacy in these patients.
Re-treatment
Re-treatment with a dosing regimen of Weeks 0 and 4, followed by 12 weeks dosing after interruption of therapy has been shown to be safe and effective.
Psoriatic Arthritis
For the treatment of psoriatic arthritis, ustekinumab is administered by subcutaneous injection. The recommended dose of ustekinumab is 45 mg administered at Weeks 0 and 4, then every 12 weeks thereafter. Alternatively, 90 mg may be used in patients with a body weight greater than 100 kg.
Consideration should be given to discontinuing treatment in patients who have shown no response up to 28 weeks of treatment.
Crohn’s Disease
In patients with Crohn’s disease, the recommended treatment regimen is a single intravenous (IV) tiered dose of ustekinumab based on body weight (Table 1), followed by 90 mg subcutaneous dosing 8 weeks later, then every 8 weeks thereafter (see Instructions for Use, Handling and Disposal – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
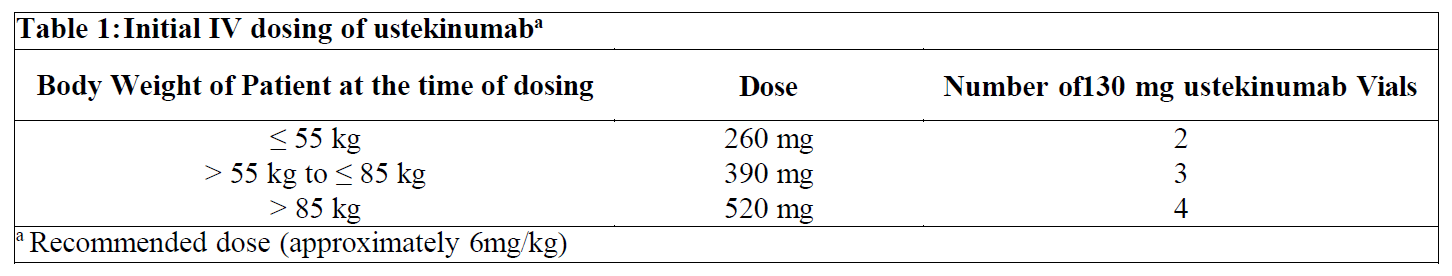
For some patients, a single IV dose based on body weight (Table 1) followed by 90 mg subcutaneous dosing 8 weeks later, then every 12 weeks thereafter may be acceptable according to clinical judgment. Patients who inadequately respond to 90 mg subcutaneous dosing every 12 weeks may benefit from an increase in dosing frequency to every 8 weeks (see Clinical Studies – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Immunomodulators and/or corticosteroids may be continued during treatment with ustekinumab. In patients who have responded to treatment with ustekinumab corticosteroids may be reduced or discontinued in accordance with standard of care.
If therapy in Crohn’s disease is interrupted, resumption of treatment with subcutaneous dosing every 8 weeks is safe and effective.
Dosage – Pediatric Population (6 years and older)
Plaque Psoriasis
The recommended dose of Steqeyma based on body weight is shown below (Table 2). Steqeyma should be administered at Weeks 0 and 4, then every 12 weeks thereafter. There is no dose form for Steqeyma that allows weight-based dosing for pediatric patients below 60 kg. For pediatric plaque psoriasis, Steqeyma is available only as 45 mg and 90 mg solution for injection in pre-filled syringe.

Patients weighing less than 60 kg should be accurately dosed on a mg/kg basis using another ustekinumab product for which a 45 mg solution for injection in vial presentation enabling weight-based dosing is available. Consideration should be given to discontinuing treatment in patients who have shown no response up to 28 weeks of treatment.
General Consideration for Administration
Subcutaneous administration
Ustekinumab is intended for use under the guidance and supervision of a physician. In pediatric patients, it is recommended that ustekinumab be administered by a health-care provider. Patients or their caregivers may inject ustekinumab if a physician determines that it is appropriate and with medical follow-up as necessary, after proper training in subcutaneous injection technique and disposal (see Instructions for Use, Handling and Disposal – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Comprehensive instructions for the subcutaneous administration of ustekinumab are given in the “Patient Information Leaflet”. Patients should be instructed to inject the prescribed amount of ustekinumab according to the directions provided in the patient information leaflet.
Do not inject ustekinumab into areas where the skin is tender, bruised, red, hard, thick, scaly, or affected by psoriasis.
Prior to subcutaneous administration, visually inspect the solution in the ustekinumab vial for particulate matter and discoloration. The product is colorless to light yellow and may contain a few small translucent or white particles of protein. This appearance is not unusual for proteinaceous solutions. The product should not be used if solution is discolored or cloudy, or if other particulate matter is present. ustekinumab does not contain preservatives; therefore, any unused product remaining in the vial and syringe should not be used.
Intravenous infusion (Crohn’s Disease)
Ustekinumab 130 mg vial is for IV infusion only. Intravenous infusion of ustekinumab should be administered by qualified health-care professionals (for preparation, see Instructions for Use, Handling and Disposal – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Special populations
Pediatrics
Studies of ustekinumab in pediatric patients below 6 years of age have not been conducted. No studies have been conducted in pediatric patients with psoriatic arthritis, Crohn’s disease.
Elderly
Of the 6710 patients exposed to ustekinumab, a total of 353 were 65 years or older (183 patients with psoriasis, 69 patients with psoriatic arthritis, 58 with Crohn’s disease). No major age-related differences in clearance or volume of distribution were observed in clinical studies. Although no overall differences in safety or efficacy were observed between older and younger patients in clinical studies in approved indications, the number of patients aged 65 and over is not sufficient to determine whether they respond differently from younger patients.
Renal impairment
Specific studies have not been conducted in patients with renal insufficiency.
Hepatic impairment
Specific studies have not been conducted in patients with hepatic insufficiency.
Contraindications
Contraindications
Severe hypersensitivity to ustekinumab or to any of the excipients (see Warnings and Precautions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
