OBETIDE SOLUTION FOR INJECTION IN PRE-FILLED PEN 6MG/ML [SIN17342P]
Active ingredients: OBETIDE SOLUTION FOR INJECTION IN PRE-FILLED PEN 6MG/ML
Product Info
OBETIDE SOLUTION FOR INJECTION IN PRE-FILLED PEN 6MG/ML
[SIN17342P]
Product information
Active Ingredient and Strength | LIRAGLUTIDE - 6 MG/ML |
Dosage Form | INJECTION |
Manufacturer and Country | SUN PHARMACEUTICAL INDUSTRIES LIMITED - INDIA |
Registration Number | SIN17342P |
Licence Holder | RANBAXY (MALAYSIA) SDN. BHD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | A10BJ02 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
INDICATIONS
Adults
OBETIDE is indicated as an adjunct to a reduced-calorie diet and increased physical activity for weight management in adult patients with an initial Body Mass Index (BMI) of:
≥ 30 kg/m2 (obesity), or
≥ 27 kg/m2 to < 30 kg/m2 (overweight) in the presence of at least one weight related comorbidity such as dysglycaemia (pre-diabetes or type 2 diabetes mellitus), hypertension, dyslipidaemia, or obstructive sleep apnoea.
Treatment with OBETIDE should be discontinued after 12 weeks on the 3.0 mg/day dose if patients have not lost at least 5% of their initial body weight.
Adolescents
OBETIDE can be used as an adjunct to a healthy nutrition and increased physical activity for weight management in adolescent patients from the age of 12 years and above with:
an inadequate response to reduced calorie diet and increased physical activity alone, and
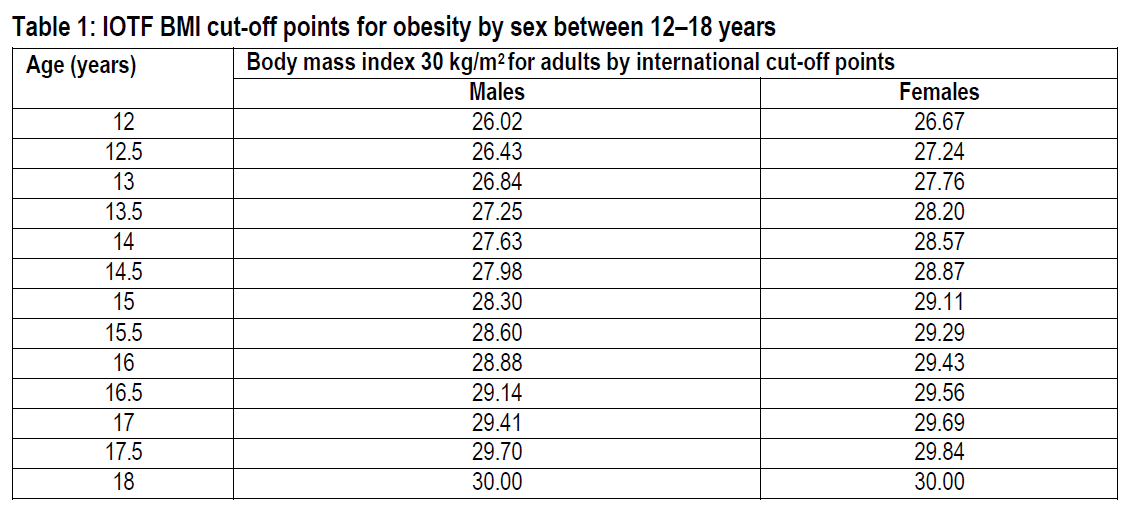
obesity (BMI corresponding to ≥30 kg/m2 for adults by international cut-off points)* and
body weight above 60 kg
Limitations of Use: The safety and effectiveness of OBETIDE in pediatric patients with type 2 diabetes have not been established.
Treatment with OBETIDE should be discontinued and re-evaluated if patients have not lost at least 4% of their BMI or BMI z score after 12 weeks on the 3.0 mg/day or maximum tolerated dose.
*IOTF BMI cut-off points for obesity by sex between 12–18 years (see table 1):
Dosing
DOSE AND METHOD OF ADMINISTRATION
OBETIDE is available at the strength of 6mg/ml, 3ml only and may not be suitable for all the dosing recommendations mentioned below. In such cases, other approved strength must be used.
Dose
Adults
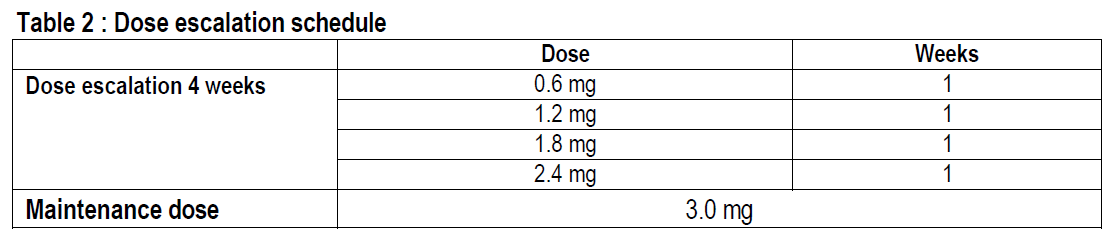
The starting dose is 0.6 mg once daily. The dose should be increased to 3.0 mg once daily in increments of 0.6 mg with at least one week intervals to improve gastro-intestinal tolerability (see table 2). If escalation to the next dose step is not tolerated for two consecutive weeks, consider discontinuing treatment. Daily doses higher than 3.0 mg are not recommended.
Adolescents (≥12 years)
For adolescents from the age of 12 to below 18 years old a similar dose escalation schedule as for adults should be applied (see table 2). The dose should be increased until 3.0 mg (maintenance dose) or maximum tolerated dose has been reached. Daily doses higher than 3.0 mg are not recommended.
Missed doses
If a dose is missed within 12 hours from when it is usually taken, the patient should take the dose as soon as possible. If there is less than 12 hours to the next dose, the patient should not take the missed dose and resume the once-daily regimen with the next scheduled dose. An extra dose or increase in dose should not be taken to make up for the missed dose.
Patients with type 2 diabetes mellitus
OBETIDE should not be used in combination with another GLP-1 receptor agonist.
When initiating OBETIDE, consider reducing the dose of concomitantly administered insulin or insulin secretagogues (such as sulfonylureas) to reduce the risk of hypoglycaemia. Blood glucose self-monitoring is necessary to adjust the dose of insulin or insulin-secretagogues (see WARNINGS AND PRECAUTIONS – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Special populations
Elderly patients (≥ 65 years old)
No dose adjustment is required based on age. Therapeutic experience in patients ≥75 years of age is limited and use in these patients is not recommended (see WARNINGS AND PRECAUTIONS and PHARMACODYNAMIC AND PHARMACOKINETIC PROPERTIES; Pharmacokinetic properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal impairment
No dose adjustment is required for patients with mild or moderate renal impairment (creatinine clearance ≥30 mL/min). OBETIDE is not recommended for use in patients with severe renal impairment (creatinine clearance <30 mL/min) including patients with end-stage renal disease (see WARNINGS AND PRECAUTIONS, UNDESIRABLE EFFECTS and PHARMACODYNAMIC AND PHARMACOKINETIC PROPERTIES; Pharmacokinetic properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Hepatic impairment
No dose adjustment is recommended for patients with mild or moderate hepatic impairment. OBETIDE is not recommended for use in patients with severe hepatic impairment and should be used cautiously in patients with mild or moderate hepatic impairment (see WARNINGS AND PRECAUTIONS and PHARMACODYNAMIC AND PHARMACOKINETIC PROPERTIES; Pharmacokinetic properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Paediatric population
No dose adjustment is required for adolescents from the age of 12 years and above. The safety and efficacy of OBETIDE in children below 12 years of age has not been established (see PHARMACODYNAMIC AND PHARMACOKINETIC PROPERTIES; Pharmacodynamic properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Method of administration
OBETIDE is for subcutaneous use only. It must not be administered intravenously or intramuscularly.
OBETIDE is administered once daily at any time, independent of meals. It should be injected in the abdomen, thigh or upper arm. The injection site and timing can be changed without dose adjustment. However, it is preferable that OBETIDE is injected around the same time of the day, when the most convenient time of the day has been chosen. Injection site should always be rotated to reduce the risk of injection site amyloid deposits (see UNDESIRABLE EFFECTS – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information)
Contraindications
CONTRAINDICATIONS
Hypersensitivity to liraglutide or to any of the excipients present in the formulation.
