STOBOCLO® SOLUTION FOR INJECTION IN PRE-FILLED SYRINGE 60MG/ML [SIN17387P]
Active ingredients: STOBOCLO® SOLUTION FOR INJECTION IN PRE-FILLED SYRINGE 60MG/ML
Product Info
STOBOCLO® SOLUTION FOR INJECTION IN PRE-FILLED SYRINGE 60MG/ML
[SIN17387P]
Product information
Active Ingredient and Strength | DENOSUMAB - 60 MG/ML |
Dosage Form | INJECTION, SOLUTION |
Manufacturer and Country | CELLTRION PHARM INC. (CLT-PHARM) - KOREA, REPUBLIC OF |
Registration Number | SIN17387P |
Licence Holder | CELLTRION HEALTHCARE SINGAPORE PRIVATE LIMITED |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | M05BX04 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
1 INDICATIONS AND USAGE
1.1 Treatment of Postmenopausal Women with Osteoporosis at High Risk for Fracture
Stoboclo is indicated for the treatment of postmenopausal women with osteoporosis at high risk for fracture, defined as a history of osteoporotic fracture, or multiple risk factors for fracture; or patients who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, denosumab reduces the incidence of vertebral, nonvertebral, and hip fractures [see Clinical Studies (14.1) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
1.2 Treatment to Increase Bone Mass in Men with Osteoporosis
Stoboclo is indicated for treatment to increase bone mass in men with osteoporosis at high risk for fracture, defined as a history of osteoporotic fracture, or multiple risk factors for fracture; or patients who have failed or are intolerant to other available osteoporosis therapy [see Clinical Studies (14.2) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
1.3 Treatment of Glucocorticoid-Induced Osteoporosis
Stoboclo is indicated for the treatment of glucocorticoid-induced osteoporosis in men and women at high risk of fracture who are either initiating or continuing systemic glucocorticoids in a daily dosage equivalent to 7.5 mg or greater of prednisone and expected to remain on glucocorticoids for at least 6 months. High risk of fracture is defined as a history of osteoporotic fracture, multiple risk factors for fracture, or patients who have failed or are intolerant to other available osteoporosis therapy [see Clinical Studies (14.3) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
1.4 Treatment of Bone Loss in Men Receiving Androgen Deprivation Therapy for Prostate Cancer
Stoboclo is indicated as a treatment to increase bone mass in men at high risk for fracture receiving androgen deprivation therapy for nonmetastatic prostate cancer. In these patients denosumab also reduced the incidence of vertebral fractures [see Clinical Studies (14.4) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
1.5 Treatment of Bone Loss in Women Receiving Adjuvant Aromatase Inhibitor Therapy for Breast Cancer
Stoboclo is indicated as a treatment to increase bone mass in women at high risk for fracture receiving adjuvant aromatase inhibitor therapy for breast cancer [see Clinical Studies (14.5) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
Dosing
2 DOSAGE AND ADMINISTRATION
2.1 Pregnancy Testing Prior to Initiation of Stoboclo
Pregnancy must be ruled out prior to administration of Stoboclo. Perform pregnancy testing in all females of reproductive potential prior to administration of Stoboclo. Based on findings in animals, denosumab products can cause fetal harm when administered to pregnant women [see Use in Specific Populations (8.1, 8.3) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
2.2 Laboratory Testing in Patients with Advanced Chronic Kidney Disease Prior to Initiation of Stoboclo
In patients with advanced chronic kidney disease [i.e., estimated glomerular filtration rate (eGFR) < 30 mL/min/1.73 m2], including dialysis-dependent patients, evaluate for the presence of chronic kidney disease mineral and bone disorder (CKD-MBD) with intact parathyroid hormone (iPTH), serum calcium, 25(OH) vitamin D, and 1,25 (OH)2 vitamin D prior to decisions regarding Stoboclo treatment. Consider also assessing bone turnover status (serum markers of bone turnover or bone biopsy) to evaluate the underlying bone disease that may be present [see Warnings and Precautions (5.1) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
2.3 Recommended Dosage
Stoboclo should be administered by a healthcare provider.
The recommended dose of Stoboclo is 60 mg administered as a single subcutaneous injection once every 6 months. Administer Stoboclo via subcutaneous injection in the upper arm, the upper thigh, or the abdomen. All patients should receive calcium 1000 mg daily and at least 400 international units vitamin D daily [see Warnings and Precautions (5.1) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
If a dose of Stoboclo is missed, administer the injection as soon as the patient is available. Thereafter, schedule injections every 6 months from the date of the last injection.
2.4 Preparation and Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. Stoboclo is a clear, colorless to pale yellow solution. Do not use if the solution is discolored or cloudy or if the solution contains particles or foreign particulate matter.
Prior to administration, Stoboclo may be removed from the refrigerator and brought to room temperature up to 25°C (77°F) by standing in the original container. This generally takes 15 to 30 minutes. Do not warm Stoboclo in any other way [see How Supplied/Storage and Handling (16) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
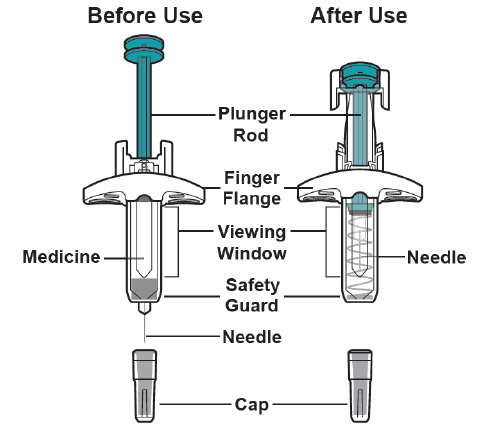
Instructions for Administration of Prefilled Syringe with Safety Guard
IMPORTANT: To reduce the risk of accidental needle stick injury, each prefilled syringe has a safety guard that is automatically activated to cover the needle after you have given the injection.
Do not pull back on the plunger rod at any time.
Step 1: Remove the Needle Cap
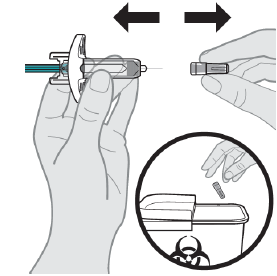
Dispose of the needle cap right away in the nearest sharps disposal container.
DO NOT re-cap the prefilled syringe.
Step 2: Administer Subcutaneous Injection
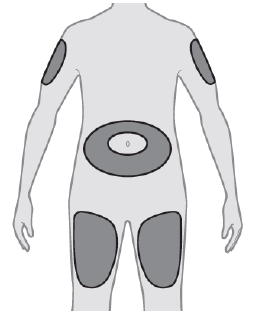
Choose an appropriate injection site. The recommended injection sites for Stoboclo include: the outer area of the upper arms OR the upper legs (thighs) OR around the stomach area (abdomen).
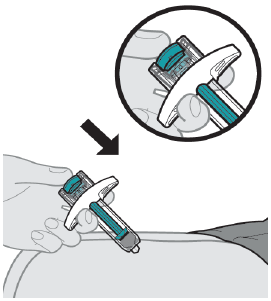
Inject all of the liquid by using your thumb to push the plunger rod all the way down.
If the plunger rod is not fully pressed, the safety guard will not extend to cover the needle when it is removed.
Step 3: Remove the prefilled syringe from the injection site
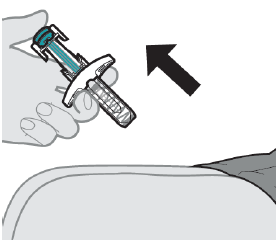
After the prefilled syringe is empty, slowly remove the needle by lifting your thumb from the plunger rod until the needle is completely covered by the safety guard.
Do not rub the injection site.
Immediately dispose of the prefilled syringe in the nearest sharps disposal container.
Contraindications
4 CONTRAINDICATIONS
Stoboclo is contraindicated in:
Patients with hypocalcemia: Pre-existing hypocalcemia must be corrected prior to initiating therapy with Stoboclo [see Warnings and Precautions (5.1) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
Pregnant women: Denosumab products may cause fetal harm when administered to a pregnant woman. In women of reproductive potential, pregnancy testing should be performed prior to initiating treatment with Stoboclo [see Use in Specific Populations (8.1) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
Patients with hypersensitivity to denosumab products: Stoboclo is contraindicated in patients with a history of systemic hypersensitivity to any component of the product. Reactions have included anaphylaxis, facial swelling, and urticaria [see Warnings and Precautions (5.3), Adverse Reactions (6.2) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
