ITOVEBI FILM COATED TABLETS 9 MG [SIN17404P]
Active ingredients: ITOVEBI FILM COATED TABLETS 9 MG
Product Info
ITOVEBI FILM COATED TABLETS 9 MG
[SIN17404P]
Product information
Active Ingredient and Strength | INAVOLISIB - 9 MG |
Dosage Form | TABLET, FILM COATED |
Manufacturer and Country | F.HOFFMANN-LA ROCHE AG - SWITZERLAND |
Registration Number | SIN17404P |
Licence Holder | ROCHE SINGAPORE PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01EM06 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
2.1 THERAPEUTIC INDICATION(S)
Itovebi, in combination with palbociclib and fulvestrant, is indicated for the treatment of adult patients with PIK3CA-mutated, hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative, locally advanced or metastatic breast cancer, following recurrence on or within 12 months of completing adjuvant endocrine therapy.
Dosing
2.2 DOSAGE AND ADMINISTRATION
2.2.1 General
Patients with HR-positive, HER2-negative, locally advanced or metastatic breast cancer should be selected for treatment with Itovebi based on the presence of one or more PIK3CA mutations using a validated assay. PIK3CA mutation status should be established prior to initiation of Itovebi therapy.
2.2.2 Dose Recommendation
The recommended dose of is 9 mg taken orally once daily with or without food.
Itovebi should be administered in combination with palbociclib and fulvestrant. The recommended dose of palbociclib is 125 mg taken orally once daily for 21 consecutive days followed by 7 days off treatment to comprise a complete cycle of 28 days. Refer to the prescribing information for palbociclib and fulvestrant being used for complete dosing information.
Treatment of pre/perimenopausal women with Itovebi should also include a luteinizing hormone-releasing hormone (LHRH) agonist in accordance with local clinical practice.
For male patients, consider treatment with an LHRH agonist according to local clinical practice.
2.2.3 Duration of Treatment
It is recommended that patients are treated with Itovebi until disease progression or unacceptable toxicity.
2.2.4 Delayed or Missed Doses
Patients should be encouraged to take their dose at approximately the same time each day. If a dose of Itovebi is missed, it can be taken within 9 hours after the time it is usually taken. After more than 9 hours, the dose should be skipped for that day. On the next day, Itovebi should be taken at the usual time. If the patient vomits after taking the Itovebi dose, the patient should not take an additional dose on that day and should resume the usual dosing schedule the next day at the usual time.
2.2.5 Dose Modification
Management of adverse reactions may require temporary interruption, dose reduction, or discontinuation of treatment with Itovebi.
The recommended dose reduction guidelines for adverse reactions are listed in Table 1.
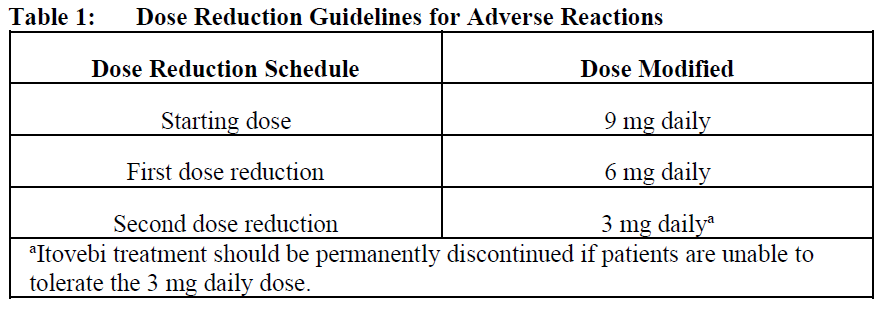
The dose of Itovebi may be re-escalated to a maximum daily dose of 9 mg based on clinical evaluation of the patient by the treating physician.
Hyperglycemia
Before initiating treatment with Itovebi, fasting plasma glucose (FPG)/blood glucose (FBG) and HbA1C levels should be tested, and plasma/blood glucose levels should be optimized in all patients. Evaluate patients for renal impairment prior to treatment with Itovebi (see section 2.2.6.3 Special Dosage Instructions, Renal Impairment and section 3.2 Pharmacokinetic Properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). After initiating treatment with Itovebi, patient fasting glucose (FPG or FBG) levels should be monitored or self-monitored based on the recommended schedule (see section 2.4.1 Warnings and Precautions, General – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
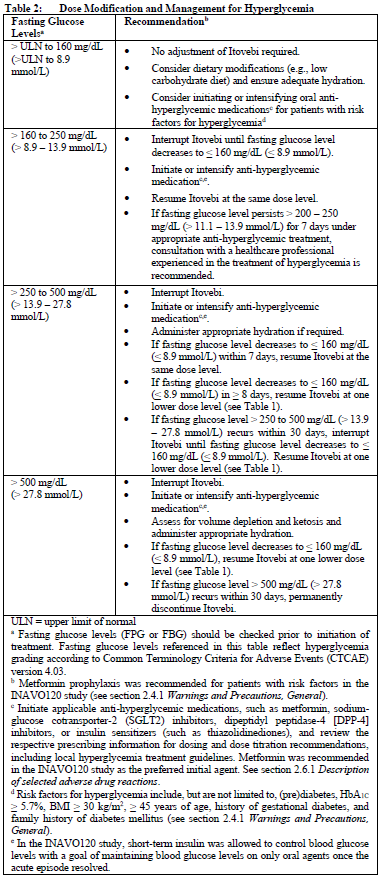
Stomatitis
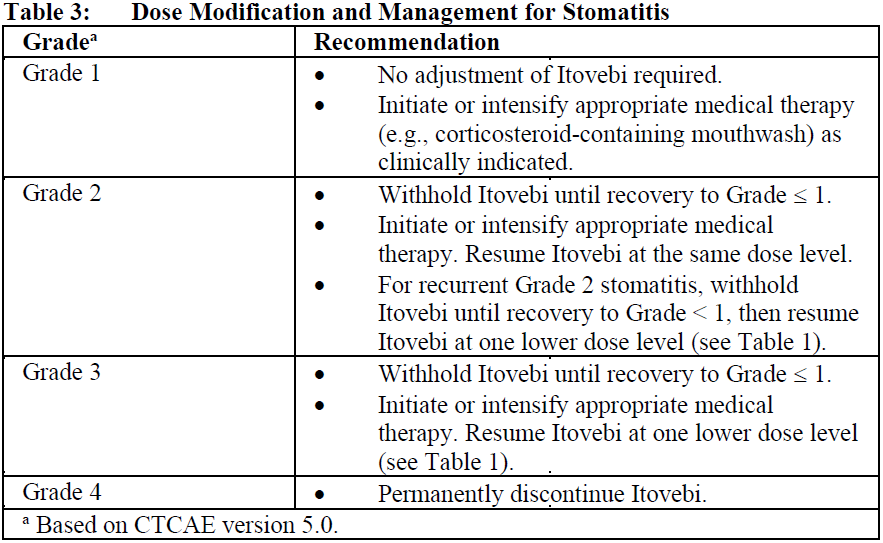
Other Adverse Reactions
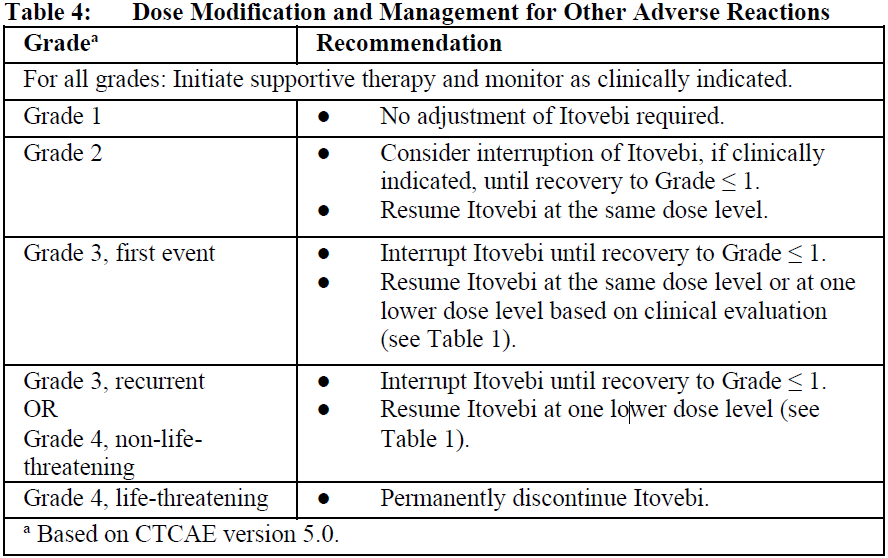
2.2.6 Special Dosage Instructions
2.2.6.1 Pediatric Use
The safety and efficacy of Itovebi has not been established in children and adolescents (< 18 years).
2.2.6.2 Geriatric Use
No dose adjustment of Itovebi is required in patients ≥ 65 years of age. For details on geriatric data, see section 2.5.5 Geriatric Use – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
2.2.6.3 Renal Impairment
No dose adjustment is required in patients with mild renal impairment (eGFR ≥ 60 to < 90mL/min). The recommended starting dose of Itovebi for patients with moderate renal impairment (eGFR 30 to < 60 mL/min) is 6 mg orally once daily, and 3 mg orally once daily for patients with severe renal impairment (eGFR < 30 mL/min). For details on renal impairment data, see section 2.5.6 Renal Impairment – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
2.2.6.4 Hepatic Impairment
No dose adjustment is required in patients with mild hepatic impairment (total bilirubin > ULN to ≤ 1.5 × ULN or AST > ULN and total bilirubin ≤ ULN). The safety and efficacy of Itovebi have not been studied in patients with moderate to severe hepatic impairment. For details on hepatic impairment data, see section 2.5.7 Hepatic Impairment – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Contraindications
2.3 CONTRAINDICATIONS
Itovebi is contraindicated in patients with a known hypersensitivity to inavolisib or any of the excipients.
