CEFAZOLIN SANDOZ 1 G/VIAL [SIN07879P]
Active ingredients: CEFAZOLIN SANDOZ 1 G/VIAL
Product Info
CEFAZOLIN SANDOZ 1 G/VIAL
[SIN07879P]
Product information
Active Ingredient and Strength | CEFAZOLIN SODIUM EQV CEFAZOLIN - 1 G/VIAL |
Dosage Form | INJECTION, POWDER, FOR SOLUTION |
Manufacturer and Country | SANDOZ GMBH - AUSTRIA |
Registration Number | SIN07879P |
Licence Holder | SANDOZ SINGAPORE PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J01DB04 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Indications
Cefazolin is indicated in a number of infections caused by cefazolin-sensitive micro-organisms.
These include:
Infections of the respiratory tract like bronchitis and pneumonia
Infections of the urogenital tract like pyelonephritis, cystitis, urethritis and prostatitis
Infections of the skin and soft tissues
Infections of the bile ducts
Bone and joint infections
Endocarditis
Systemic septic infections
Perioperative prophylaxis (hysterectomy, cholecystectomy, open heart surgery, bone and joint surgery).
Dosing
Dosage
Adults are given 500 mg to 1 g of cefazolin b.i.d. or t.i.d. for uncomplicated infections, 500 mg to 1 g t.i.d. or q.i.d. for moderately severe and severe infections and 1 g to 1.5 q.i.d. for severe life-threatening infections. Rarely, doses of up to 12 g daily were administered.
In adult patients with renal insufficiency the dosage schedule below should be followed:
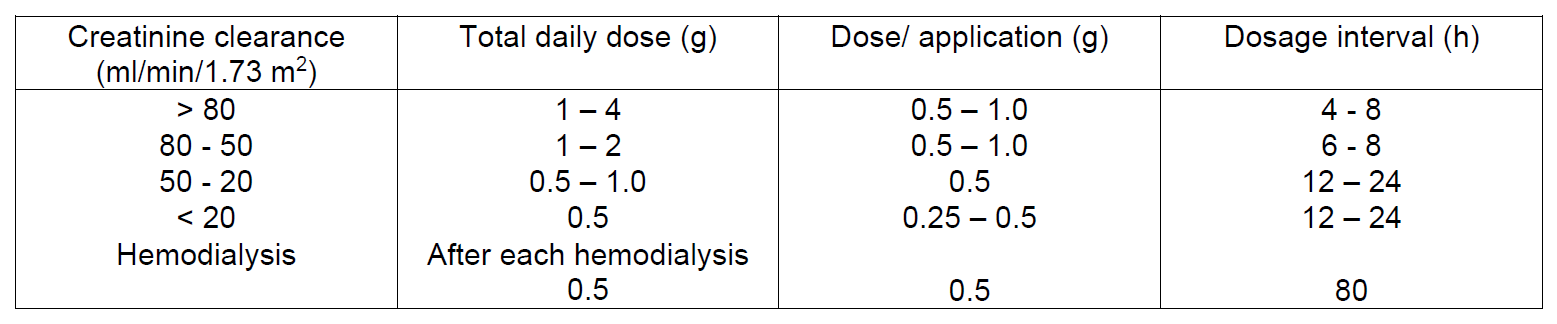
For perioperative use to prevent infections doses depend on the type and duration of surgery. The doses below are recommended:
30 minutes to 1 hour prior to surgery an initial dose of 1 g is administered i.v. or i.m. This is sufficient for minor procedures of short duration.
For longer operations (2 hours or more) another dose of 500 mg to 1 g is administered i.v. or i.m. intraoperatively. The dosage level and the timing depend on the type and duration of surgery.
Postoperatively 500 mg to 1 g are administered i.v. or i.m. at intervals of 6 to 8 hours for 24 hours.
If potential infections are likely to be very dangerous for the patient (e.g. after cardiac surgery or major orthopedic surgery like total joint replacement), it is advisable to continue postoperative dosing (see item 3.) for 3 to 5 days.
Dosage guidelines for children
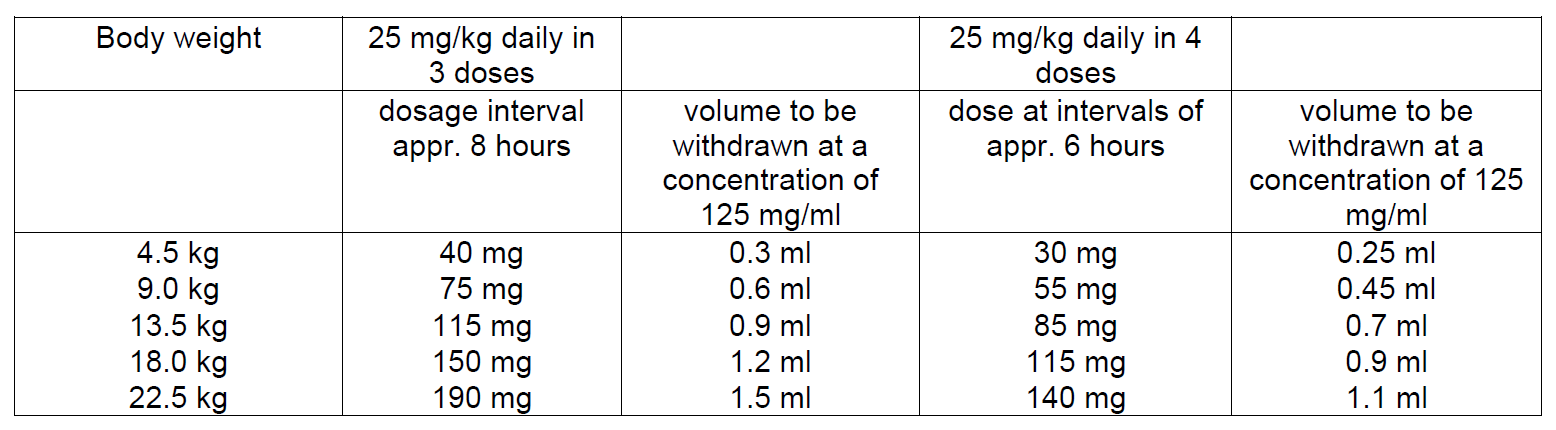
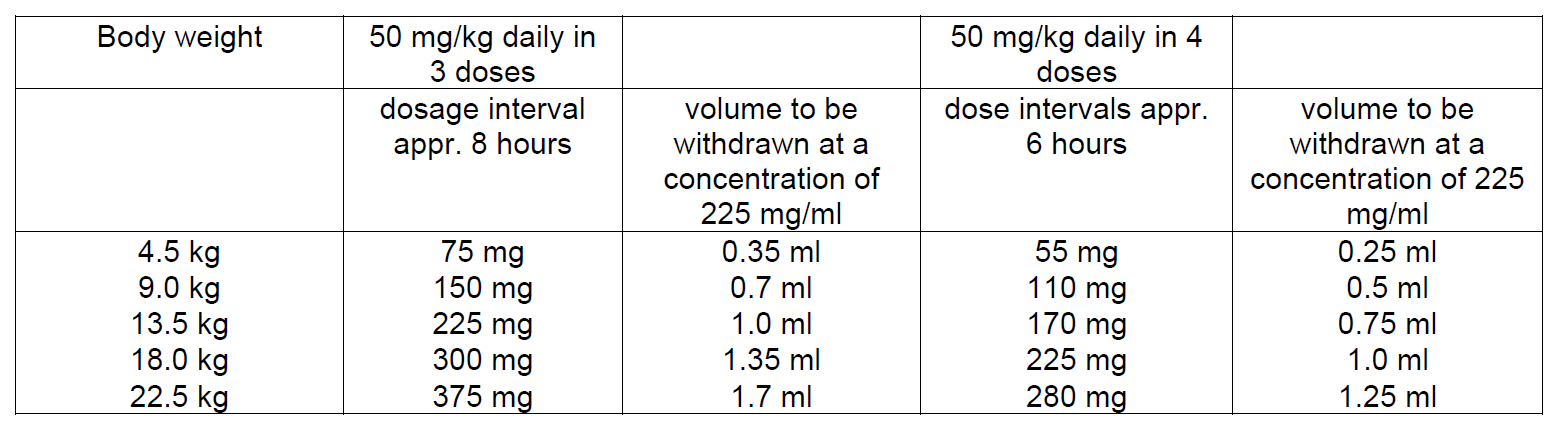
In neonates and infants below the age of 1 month the elimination half-life of cefazolin is usually much longer than in older children. To maintain drug safety serum drug levels should, therefore, be monitored in this age group.
Infants above age 1 month
A total daily dose of 25–50 mg/kg body weight divided in 3–4 fractions is effective in most mild to moderate infections. In severe infections the total dose may be increased to 100 mg/kg body weight.
Children with renal insufficiency
Children with a creatinine clearance (in ml/min/1.73 m2) between 70 and 40 are given 60% of the standard daily dose divided in 2 fractions. Children with a creatinine clearance between 40 and 20 are given 25% of the standard daily dose divided in 2 fractions.
Children with a creatinine clearance between 20 and 5 are given 10% of the standard daily dose at intervals of 24 hours. These doses are applied after the administration of a loading dose equivalent to the standard dose.
Children undergoing hemodialysis are given 7 mg/kg by weight at the beginning of treatment. As cefazolin serum levels drop by 35% to 65% during dialysis, a dose of 3 to 4 mg/kg by weight is administered between dialysis sessions (dialysis interval = 72 hours).
Elderly patients
No dose adjustments are needed in elderly patients with normal renal function.
Duration of treatment
The duration of treatment depends on the course of the disease. In keeping with the general principles of antibiotic therapy, cefazolin should be continued for at least 2 to 3 days after the fever has subsided or proof is obtained for the eradication of the causative agent.
Infections caused by β-hemolytic streptococci should prophylactically be treated for at least 10 days in order to prevent late complications.
Contraindications
Contraindications
The drug is contraindicated in patients allergic to cefazolin or other cephalosporins.
For administration to young children cefazolin should not be dissolved in lidocaine solutions. Special caution should be exercised in patients with an allergic diathesis, with bronchial asthma or hay fever.
Caution should also be exercised in patients with a history of gastrointestinal disease (colitis).
Pregnancy and lactation
The safety of cefazolin for use during pregnancy has not yet been established. The drug passes the placental barrier; minor amounts are present in breast milk. If cefazolin administration is considered during pregnancy, the benefits should be carefully weighed against the risks. Nursing mothers should suspend breast-feeding during cefazolin treatment.
