VFEND POWDER FOR ORAL SUSPENSION 40MG/ML [SIN13033P]
Active ingredients: VFEND POWDER FOR ORAL SUSPENSION 40MG/ML
Product Info
VFEND POWDER FOR ORAL SUSPENSION 40MG/ML
[SIN13033P]
Product information
Active Ingredient and Strength | VORICONAZOLE - 40 MG/ML |
Dosage Form | POWDER, FOR SUSPENSION |
Manufacturer and Country | FAREVA AMBOISE - FRANCE |
Registration Number | SIN13033P |
Licence Holder | PFIZER PRIVATE LIMITED |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | NA |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Voriconazole is a broad spectrum, triazole antifungal agent and is indicated as follows:
Treatment of invasive aspergillosis;
Treatment of candidaemia in non-neutropenic patients;
Treatment of fluconazole-resistant serious invasive Candida infections (including C. krusei);
Treatment of serious fungal infections caused by Scedosporium spp. and Fusarium spp.;
Prophylaxis in patients who are at high risk of developing invasive fungal infections, such as allogeneic haematopoietic stem cell transplant (HSCT) recipients.
Dosing
4.2 Posology and method of administration
Film-coated tablets:
Voriconazole film-coated tablets are to be taken at least one hour before, or one hour following a meal.
Powder for oral suspension:
Voriconazole oral suspension is to be taken at least one hour before, or two hours following a meal.
Powder for solution for infusion:
Voriconazole requires reconstitution and dilution (see Section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information) prior to administration as an intravenous infusion.
Voriconazole powder for solution for infusion is not recommended for bolus injection.
It is recommended that voriconazole be administered at a maximum rate of 3 mg/kg per hour over 1 to 3 hours.
Blood products and concentrated electrolytes
Voriconazole must not be infused concomitantly with any blood product or any short-term infusion of concentrated electrolytes, even if the two infusions are running in separate intravenous lines (or cannulas). Electrolyte disturbances such as hypokalaemia, hypomagnesaemia and hypocalcaemia should be corrected prior to initiation of voriconazole therapy (see Section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Intravenous solutions containing (non-concentrated) electrolytes
Voriconazole can be infused at the same time as other intravenous solutions containing (non-concentrated) electrolytes, but must be infused through a separate line.
Total parenteral nutrition (TPN)
Voriconazole can be infused at the same time as total parenteral nutrition, but must be infused in a separate line. If infused through a multiple-lumen catheter, TPN needs to be administered using a different port from the one used for voriconazole (see Section 6.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Other intravenous products
Voriconazole must not be infused into the same line or cannula concomitantly with other intravenous products.
Use in adults
Therapy must be initiated with the specified loading dose regimen of either intravenous or oral voriconazole to achieve plasma concentrations on Day 1 that are close to steady state. On the basis of the high oral bioavailability (96%; see Section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information), switching between intravenous and oral administration is appropriate when clinically indicated.
Detailed information on dosage recommendations is provided in the following table:
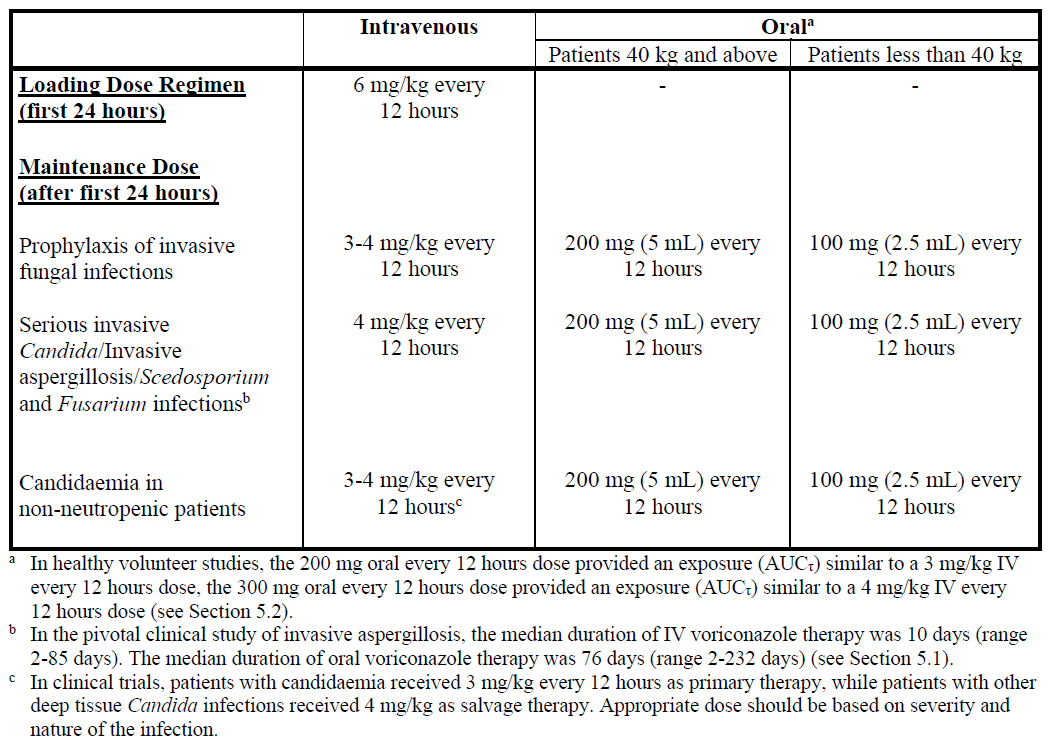
Dosage adjustment
Film-coated tablets/Powder for oral suspension:
If patient response is inadequate, the maintenance dose may be increased from 200 mg (5 mL) every 12 hours (similar to 3 mg/kg IV every 12 hours) to 300 mg (7.5 mL) every 12 hours (similar to 4 mg/kg IV every 12 hours) for oral administration. For patients less than 40 kg the oral dose may be increased from 100 mg (2.5 mL) to 150 mg (3.75 mL) every 12 hours.
If patients are unable to tolerate treatment at these higher doses, reduce the oral dose by 50 mg (1.25 mL) steps to a minimum of 200 mg (5 mL) every 12 hours (or 100 mg [2.5 mL] every 12 hours for patients less than 40 kg).
Phenytoin may be co-administered with voriconazole if the maintenance dose of voriconazole is increased from 200 mg (5 mL) to 400 mg (10 mL) orally every 12 hours (from 100 mg [2.5 mL] to 200 mg [5 mL] orally, every 12 hours in patients less than 40 kg) (see Sections 4.4 and 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
When voriconazole is co-administered with adjusted doses of efavirenz, voriconazole maintenance dose should be increased to 400 mg (10 mL) every 12 hours (see Sections 4.3, 4.4 and 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Treatment duration depends upon patients’ clinical and mycological response.
Intravenous administration:
If patient response at 3 mg/kg every 12 hours is inadequate, the intravenous maintenance dose may be increased to 4 mg/kg every 12 hours.
If patients are unable to tolerate 4 mg/kg every 12 hours, reduce the intravenous maintenance dose to a minimum of 3 mg/kg every 12 hours.
Phenytoin may be co-administered with voriconazole if the maintenance dose of voriconazole is increased to 5 mg/kg intravenously every 12 hours (see Sections 4.4 and 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Treatment duration depends upon patients’ clinical and mycological response.
Use in the elderly
No dose adjustment is necessary for elderly patients.
Use in patients with renal impairment
Film-coated tablets/Powder for oral suspension:
The pharmacokinetics of orally administered voriconazole are not affected by renal impairment. Therefore, no adjustment is necessary for oral dosing for patients with mild to severe renal impairment.
Powder for solution for infusion:
In patients with moderate to severe renal dysfunction (creatinine clearance <50 mL/min), accumulation of the intravenous vehicle, sulphobutylether β-cyclodextrin sodium (SBECD) occurs. Oral voriconazole should be administered to these patients, unless an assessment of the risk benefit to the patient justifies the use of intravenous voriconazole. Serum creatinine levels should be closely monitored in these patients and, if increases occur, consideration should be given to changing to oral voriconazole therapy.
Voriconazole is haemodialysed with a clearance of 121 mL/min. A four-hour haemodialysis session does not remove a sufficient amount of voriconazole to warrant dose adjustment.
The intravenous vehicle, SBECD, is haemodialysed with a clearance of 55 mL/min.
Use in patients with hepatic impairment
No dose adjustment is necessary in patients with acute hepatic injury, manifested by elevated liver function tests (ALT, AST). Continued monitoring of liver function tests for further elevations is recommended.
It is recommended that the standard loading dose regimens be used but that the maintenance dose be halved in patients with mild to moderate hepatic cirrhosis (Child-Pugh A and B) receiving voriconazole.
Voriconazole has not been studied in patients with severe chronic hepatic cirrhosis (Child-Pugh C).
Voriconazole has been associated with elevations in liver function tests and clinical signs of liver damage, such as jaundice, and must only be used in patients with severe hepatic impairment if the benefit outweighs the potential risk. Patients with severe hepatic impairment must be carefully monitored for drug toxicity.
Use in paediatrics
Use in children (2 to <12 years) and young adolescents (12 to 14 years and <50 kg):
The recommended dosing regimen is as follows:
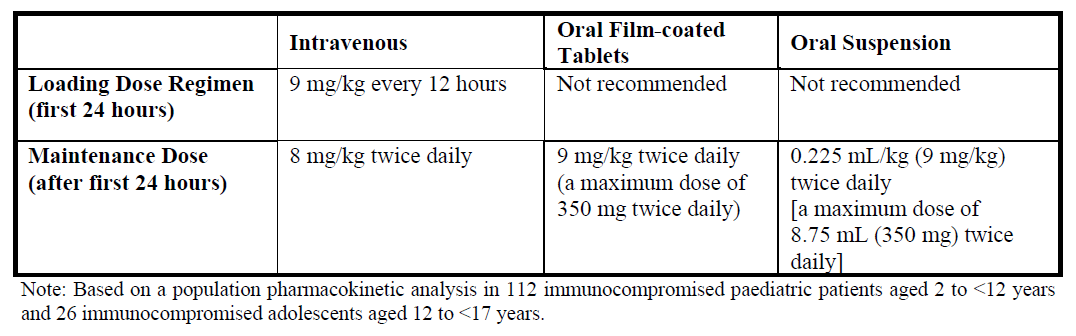
It is recommended to initiate the therapy with intravenous regimen, and oral regimen should be considered only after there is a significant clinical improvement. It should be noted that an 8 mg/kg intravenous dose will provide voriconazole exposure approximately 2-fold higher than a 9 mg/kg oral dose.
The oral dose recommendation for children is based on studies in which voriconazole was administered as the powder for oral suspension formulation. Bioequivalence between the powder for oral suspension and tablets has not been investigated in a paediatric population. Considering the assumed limited gastro-enteric transit time in paediatrics, the absorption of tablets may be different in paediatric compared to adult patients. It is therefore recommended to use the oral suspension formulation in children aged 2 to <12 years.
Safety and effectiveness in paediatric patients below the age of 2 years has not been established (see also Section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). Therefore, voriconazole is not recommended for children less than 2 years of age. Use in paediatric patients aged 2 to <12 years with hepatic or renal insufficiency has not been studied (see Sections 4.8 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). Oral bioavailability may be limited in paediatric patients 2 to <12 years with malabsorption and very low body weight for age. In that case, intravenous voriconazole administration is recommended.
Use in all other adolescents (12 to 14 years and ≥50 kg; 15 to 16 years regardless of body weight):
Voriconazole should be dosed as adults.
Dosage adjustment:
If patient response is inadequate, the dose may be increased by 1 mg/kg (0.025 mL/kg) steps (or by 50 mg [1.25 mL] steps if the maximum oral dose of 350 mg [8.75 mL] was used initially). If patients are unable to tolerate treatment, reduce the dose by 1 mg/kg (0.025 mL/kg) steps (or by 50 mg [1.25 mL] steps if the maximum oral dose of 350 mg [8.75 mL] was used initially).
Prophylaxis in adults and children
Prophylaxis should be initiated on the day of transplant and may be administered for up to 100 days. It may only be continued up to 180 days after transplantation in case of continuing immunosuppression or graft versus host disease (GvHD) (see Section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Dosage:
The recommended dosing regimen for prophylaxis is the same as for treatment in the respective age groups. Please refer to the treatment tables above.
Duration of prophylaxis:
The safety and efficacy of voriconazole use for longer than 180 days has not been adequately studied in clinical trials.
Contraindications
4.3 Contraindications
Voriconazole is contraindicated in patients with known hypersensitivity to voriconazole or to any of the excipients.
Interacting drugs listed in this section and Section 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information are a guide and not considered a comprehensive list of all possible drugs that may be contraindicated.
Co-administration of voriconazole is contraindicated with medicinal products that are highly dependent on CYP3A4 for metabolism, and for which elevated plasma concentrations are associated with serious and/or life-threatening reactions (see Section 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information):
Terfenadine
Astemizole
Cisapride
Pimozide
Lurasidone
Quinidine
Ivabradine
Ergot alkaloids (e.g., ergotamine, dihydroergotamine)
Sirolimus
Naloxegol: Co-administration may precipitate opioid withdrawal symptoms
Tolvaptan
Finerenone
Eplerenone
Voclosporin
Venetoclax: Co-administration contraindicated at initiation and during venetoclax dose titration phase as it may increase risk of tumour lysis syndrome.
Co-administration of voriconazole is contraindicated with medicinal products that induce CYP3A4 and significantly reduce its plasma concentrations of voriconazole:
Co-administration with rifabutin, rifampicin, carbamazepine, long-acting barbiturates (e.g., phenobarbital) and St. John’s Wort (see Section 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Efavirenz: Co-administration of standard doses of voriconazole with efavirenz doses of 400 mg QD or higher is contraindicated (see Section 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). For information on co-administration of voriconazole with lower doses of efavirenz, see Section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Ritonavir: Co-administration with high-dose ritonavir (400 mg and above twice daily) is contraindicated (see Section 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). For information on co-administration of voriconazole with lower doses of ritonavir, see Section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
