PY TEST CAPSULES & PY TEST KIT [SIN13330P]
Active ingredients: PY TEST CAPSULES & PY TEST KIT
Product Info
PY TEST CAPSULES & PY TEST KIT
[SIN13330P]
Product information
Active Ingredient and Strength | 14-C UREA - 37 (+/-20%) KβQ (1 MCG +/-20%) |
Dosage Form | CAPSULE |
Manufacturer and Country | AVENT, INC. (A WHOLLY SUBSIDIARY OF AVANOS MEDICAL, INC.) - UNITED STATES |
Registration Number | SIN13330P |
Licence Holder | ALL EIGHTS (SINGAPORE) PRIVATE LIMITED |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | NA |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 THERAPEUTIC INDICATIONS
PYtest® (C14-Urea breath test) is indicated for use in the detection of gastric urease as an aid in the diagnosis of H. pylori infection in the human stomach.
Dosing
4.2 DOSE AND METHOD OF ADMINISTRATION
PYtest® (C14-Urea breath test) is a qualitative and non-invasive method for the diagnosis of Helicobacter pylori (H. pylori).
Dosage
Adults: One PYtest® capsule.
Children: Clinical studies in children have not been conducted.
Method of Administration
For oral administration.
Procedural notes
Inform the patient to fast for 4 hours prior to the test
The patient should not have taken antibiotics and bismuth containing products for 1 month, proton pump inhibitors for 1 week and sucralfate for 2 weeks prior to the test
Have patient sitting at rest while doing the test
The capsule should not be handled directly as this may interfere with the test result
To avoid contamination by bacteria in the mouth, the capsule should be swallowed intact. Do not chew capsule.
See also section 4.4. Special warnings and precautions for use – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Step by Step Procedure for Balloon:
Materials needed but not provided:
clock or suitable timepiece capable of measuring 10 minutes in minute intervals.
Water (40mL)
Materials provided:
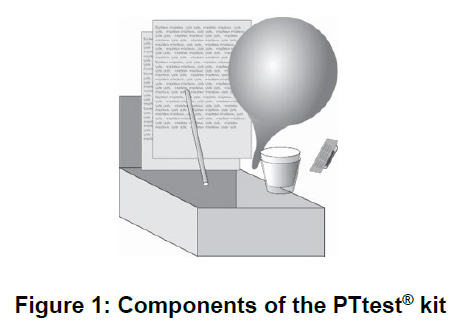
As shown in Figure 1 below, the PYtest® kit contains:
PYtest® capsule
Paper Cup (x2)
Straw
Balloon
Package seals
Return Carton
The PYtest® Kit (C14-Urea breath test) is designed for use with the PYtest® capsule, a gelatin capsule for oral administration containing 37 kBq of 14C labelled urea. The urea is absorbed on sugar spheres and coloured yellow with fluorescein.
Breath Sample Collection
Label the balloon with the required details. (Recommend using a permanent marker)
Check that all materials are present.
Sit the patient down.
Place the capsule into one of the paper cups provided.
Fill the second cup 2/3 full (20mL) with water.
Instruct the patient to swallow the capsule whole and to drink the water to wash the capsule down.
Three minutes later, fill a paper cup 2/3 full (20mL) with water and instruct the patient to drink it. This is to ensure the capsule has been ‘washed’ into the stomach.
With the patient still sitting, wait for a further seven minutes.
On the neck of the balloon, separate the larger flap from the smaller flap at the black line. This can be achieved by gently squeezing the sides of the flaps together whilst pushing the flaps apart until an opening appears.
Insert approximately one third of the straw into the opening.
Instruct the patient to take a deep breath, get them to hold it for 5–10 seconds then ask them to blow slowly through the straw into the balloon until it is firm. If the balloon is not filled with the first attempt, a second breath can be used.
Hold the neck of the balloon at the base and remove the straw. The balloon will seal.
Record the finish time on the balloon.
Discard waste (e.g., capsule packaging, used straws) according to your facility’s regulations.
Sample Preparation
The breath sample is transferred to collection fluid to trap the labelled CO2. The liquid sample is then analysed in a liquid scintillation counter.
Quality Control
A minimum of 1 mmol of CO2 is required to perform analysis of a breath sample. The amount of breath required to provide 1 mmol of CO2 varies depending on the amount of CO2 the patient is producing. Since a full balloon typically contains at least 1 mmol of CO2, the balloon should be completely filled.
Post treatment evaluation
A negative PYtest® 28 days after completing antibiotic treatment is evidence that the infection has been eradicated.
Results
Interpretation of results (10 minute sample)
Table caption
< 50 DPM | Negative for H. pylori |
50–199 DPM | Borderline positive for H. pylori |
≥200 DPM | Positive for H. pylori |
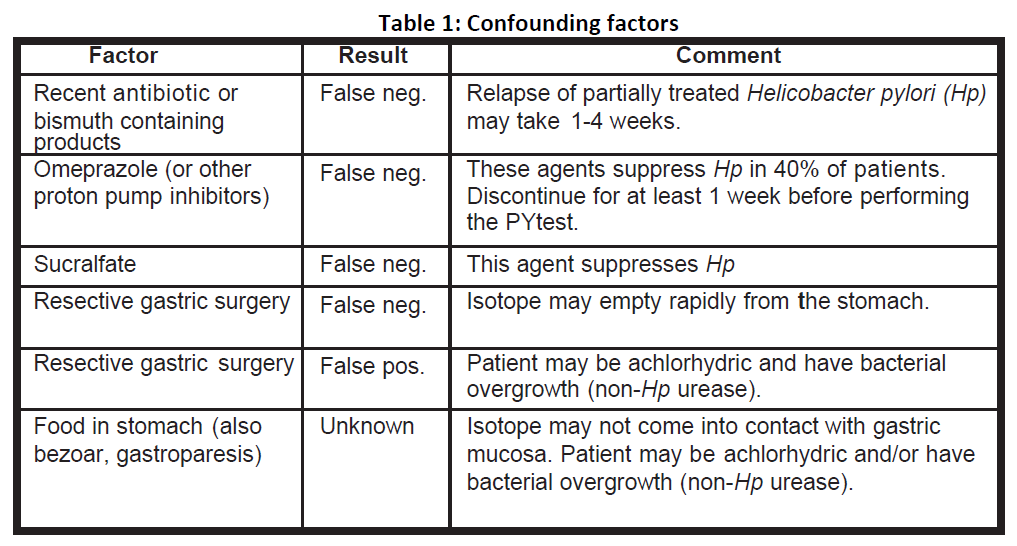
Borderline positive results should be evaluated by repeating the PYtest® or using an alternative diagnostic method. If repeat breath testing is undertaken, careful history to exclude confounding factors should be obtained. If confounding factors are present, wait an appropriate time (refer to Table 1 below) before repeating the PYtest®. The cut-off point of 50 DPM was determined to be the mean +3SD of results obtained in-patients who did not have H. pylori.
Expected values
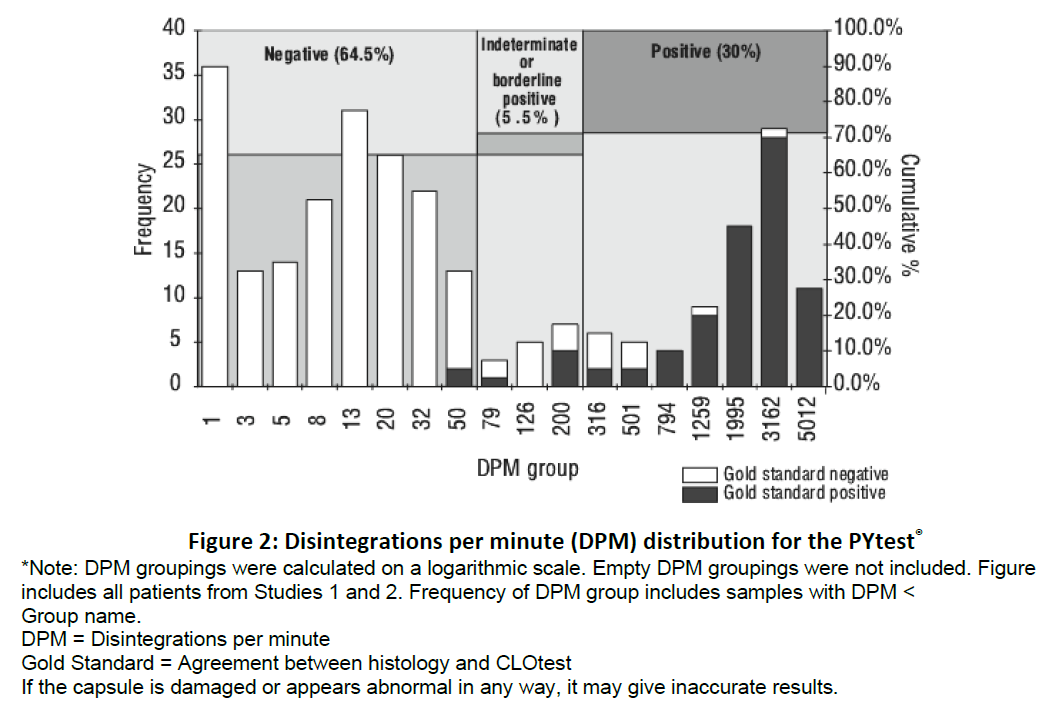
As shown in Figure 2 below, approximately 30% of patients tested will be positive for H. pylori.
Contraindications
4.3 CONTRAINDICATIONS
Hypersensitivity to the active substance, or any of the excipients.
