CURAM 1000MG+200MG POWDER FOR INTRAVENOUS SOLUTION [SIN13390P]
Active ingredients: CURAM 1000MG+200MG POWDER FOR INTRAVENOUS SOLUTION
Product Info
CURAM 1000MG+200MG POWDER FOR INTRAVENOUS SOLUTION
[SIN13390P]
Product information
Active Ingredient and Strength | AMOXICILLIN SODIUM EQV. 1 G OF AMOXICILLIN - 1.06 G |
Dosage Form | INJECTION, POWDER, FOR SOLUTION |
Manufacturer and Country | SANDOZ GMBH - AUSTRIA |
Registration Number | SIN13390P |
Licence Holder | SANDOZ SINGAPORE PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J01CR02 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Curam 1000mg + 200mg Powder for intravenous solution may be administered for the short-term treatment of primarily serious infections due to micro-organisms that are resistant to amoxicillin and other B-lactam antibiotics as a result of the formation of a clavulanic acid-susceptible ß-lactamase, such as:
Upper respiratory tract infections (including ENT) e.g. recurrent tonsillitis, sinusitis, otitis media.
Lower respiratory tract infections e.g. acute exacerbation of chronic bronchitis, lobar and bronchopneumonia.
Genito-urinary tract infections e.g. cystitis, urethritis, pyelonephritis.
Skin and soft tissue infections e.g. boils, abscesses, cellulites, wound infections.
Bone and joint infections e.g. osteomyelitis.
Other infections e.g. intra-abdominal sepsis.
Curam 1000mg + 200mg Powder for intravenous solution can be used for prophylaxis against infections in major surgery such as gastrointestinal, pelvic, head and neck, cardiac, renal, joint replacement and biliary tract.
Dosing
4.2 Posology and method of administration
Dosage
The duration of treatment is dependent on the indication and the severity of the infection. Curam 1000mg + 200mg Powder for intravenous solution may not be used for more than 14 days without monitoring of the patient’s liver function.
Adults and children over 12 years
Normal renal function
Non-serious infections: Amoxicillin/Clavulanic acid 1000/200 mg 8 hourly by intravenous injection. In more serious infections, increase frequency to six hourly intervals, If possible, preference should be given to the oral form of Amoxicillin/Clavulanic acid.
Children (3 months – 12 years)
Table caption
- Serious infections | Usually 25/5 mg/kg per slow intravenous injection or infusion (infusion time at least 30 minutes) every six hours. |
- Non-serious infections | Up to a maximum 25/5 mg/kg per slow intravenous injection or infusion (infusion time at least 30 minutes), every eight hours. If possible the oral form of Amoxicillin/Clavulanic acid is preferable. |
Children (0 – 3 months) | 25/5 mg/kg per infusion every 12 hours in premature infants and full term infants during the perinatal period, increasing to eight hours thereafter. |
Dosage in the case of renal impairment
Elimination of clavulanic acid and amoxicillin via the kidneys is delayed in patients with renal impairment. Depending on the degree of renal impairment, the total daily dose of Curam 1000mg + 200mg Powder for intravenous solution (expressed in quantities of amoxicillin) should not exceed that indicated in following table.
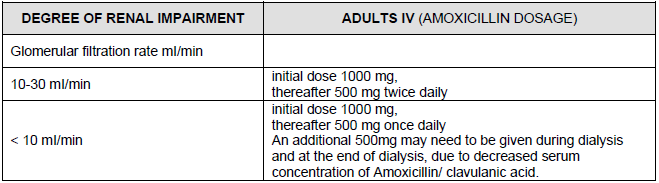
Similar reduction in dosage should be made for children.
Adult dose for surgical prophylaxis
Amoxicillin/Clavulanic acid 1000/200 mg Powder for solution for injection/infusion at the induction of anaesthesia. Operations where there is an increased risk of infection, e.g. colorectal surgery, may require 3 up to 4 doses in a 24-hour period 8 hourly.
Contraindications
4.3 Contraindications
Hypersensitivity to the active substances, to any of the penicillins or to any of the excipients
History of a severe immediate hypersensitivity reaction (e.g. anaphylaxis) to another beta-lactam agent (e.g. a cephalosporin, carbapenem or monobactam)
History of jaundice/hepatic impairment due to amoxicillin/clavulanic acid (see section 4.8 Undesirable effects – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information)
