REQUIP PD 24 HOUR PROLONGED RELEASE TABLETS (4MG) [SIN13764P]
Active ingredients: REQUIP PD 24 HOUR PROLONGED RELEASE TABLETS (4MG)
Product Info
REQUIP PD 24 HOUR PROLONGED RELEASE TABLETS (4MG)
[SIN13764P]
Product information
Active Ingredient and Strength | (ACTIVE LAYER) ROPINIROLE HYDROCHLORIDE EQV ROPINIROLE - 4 MG |
Dosage Form | TABLET, FILM COATED, EXTENDED RELEASE |
Manufacturer and Country | GLAXO WELLCOME S.A. - SPAIN |
Registration Number | SIN13764P |
Licence Holder | GLAXOSMITHKLINE PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | N04BC04 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Indications
REQUIP PD 24 HOUR is indicated for the treatment of Parkinson’s disease under the following conditions:
As monotherapy (without L-dopa) to substitute REQUIP immediate release tablet in PD patients already taking REQUIP immediate release tablet whom adequate symptomatic control has been established.
In combination with levodopa, over the course of the disease, when the effect of levodopa wears off or becomes inconsistent and fluctuations in the therapeutic effect occur (“end of dose” or “on-off” type fluctuations).
Dosing
Dosage and Administration
Pharmaceutical form
Film-coated, capsule-shaped tablets for oral administration. The tablet strengths are distinguished by colour and debossing.
2 mg: pink, capsule-shaped, film-coated tablets marked “GS” on one side and “3V2” on the other.
4 mg: light brown, capsule-shaped, film-coated tablets marked “GS” on one side and “WXG” on the other.
When switching treatment from another dopamine agonist to REQUIP PD 24 HOUR, the manufacturer’s guidance on discontinuation should be followed before initiating REQUIP PD 24 HOUR.
Individual dose titration against efficacy and tolerability is recommended.
Patients should be down-titrated if they experience disabling somnolence at any dose level. For other adverse events, down-titration followed by more gradual up-titration has been shown to be beneficial.
• Adults
REQUIP PD 24 HOUR should be taken as a single daily dose and should be taken at a similar time each day. The tablet(s) must be swallowed whole, and must not be chewed, crushed or divided. REQUIP PD 24 HOUR may be taken with or without food (see Pharmacokinetics – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Treatment initiation as adjunct to L-dopa
The dose should be titrated according to the individual clinical response and tolerability.
The starting dose is 2 mg taken once daily for 1 to 2 weeks, followed by increases of 2 mg/day at 1-week or longer intervals as appropriate, depending on therapeutic response and tolerability, up to a maximally recommended dose of 24 mg/day. Patients should be assessed for therapeutic response and tolerability at a minimal interval of 1 week or longer after each dose increment. Caution should be exercised during dose titration because too rapid a rate of titration may lead to dose selection that may not provide additional benefit, but that may increase the risk of adverse reactions.
The safety and efficacy of doses above 24 mg/day have not been established.
It may be possible to reduce gradually the L-dopa dose, depending on the clinical response. In clinical trials, the L-dopa dose was reduced gradually once the patient reached a dose of REQUIP PD 24 HOUR of 8 mg/day. On average, the L-dopa dose was reduced by approximately 30% in patients receiving REQUIP PD 24 HOUR as compared to approximately 20% in patients treated with placebo. In patients with advanced Parkinson’s disease receiving REQUIP PD 24 HOUR in combination with L-dopa, dyskinesias can occur during the initial titration of REQUIP PD 24 HOUR (see Adverse Reactions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Sudden discontinuation of REQUIP PD 24 HOUR is not recommended. REQUIP PD 24 HOUR should be discontinued gradually over a period of 7 days under the advice of the doctor (see Warnings and Precautions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
If treatment is interrupted for one day or more, re-initiation by dose titration should be considered (see above).
Switching from ropinirole immediate release tablets to ropinirole prolonged release tablets
Patients may be switched overnight from REQUIP immediate release (IR) tablets to REQUIP PD 24 HOUR PR tablets. The dose of REQUIP PD 24 HOUR PR tablets should be based on the total daily dose of REQUIP IR tablets that the patient was taking. If patients are taking a different total daily dose of ropinirole immediate release tablets to those typically prescribed doses as shown in the table, they should be switched to the nearest available dose of ropinirole prolonged release tablets as stated in the table.
The table below shows the recommended dose of REQUIP PD 24 HOUR PR tablets for patients switching from REQUIP IR tablets:
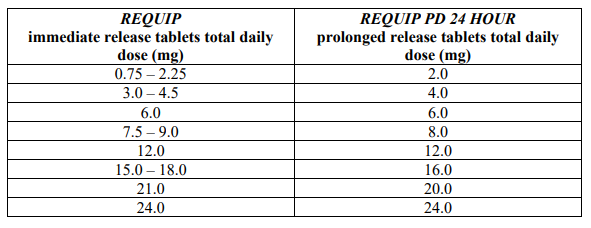
After switching to REQUIP PD 24 HOUR PR tablets, the dose may be adjusted depending on the therapeutic response (see “Treatment initiation” and “Therapeutic regimen” above).
• Elderly
The clearance of ropinirole is decreased in patients aged 65 years or above, but the dose of REQUIP PD 24 HOUR for elderly patients can be titrated in the normal manner based on therapeutic responses and tolerability.
• Children and Adolescents
The safety and efficacy of ropinirole have not been established in patients under 18 years of age; therefore, REQUIP PD 24 HOUR is not recommended for use in patients within this age group.
• Renal impairment
In patients with mild to moderate renal impairment (creatinine clearance 30 – 50 mL/min) no change in the clearance of ropinirole was observed, indicating that no dosage adjustment is necessary in this population.
A study into the use of ropinirole in patients with end stage renal disease (patients on haemodialysis) has shown that a dose adjustment in these patients is required as follows: The recommended initial dose of REQUIP PD 24 HOUR is 2 mg once daily. Further dose escalations should be based on tolerability and efficacy. The recommended maximum dose is 18 mg/day in patients receiving regular dialysis. Supplemental doses after dialysis are not required.
The use of ropinirole in patients with severe renal impairment (creatinine clearance less than 30 mL/min) without regular dialysis has not been studied.
• Hepatic impairment
The use of ropinirole in patients with hepatic impairment has not been studied.
Administration of REQUIP PD 24 HOUR to such patients is not recommended.
Contraindications
Contraindications
Hypersensitivity to ropinirole or to any of the excipients.
