REGPARA TABLETS 25 MG [SIN13993P]
Active ingredients: REGPARA TABLETS 25 MG
Product Info
REGPARA TABLETS 25 MG
[SIN13993P]
Product information
Active Ingredient and Strength | CINACALCET HYDROCHLORIDE 27.55 MG EQV. CINACALCET - 25 MG |
Dosage Form | TABLET, FILM COATED |
Manufacturer and Country | OHARA PHARMACEUTICAL CO., LTD (KAMI FACTORY) - JAPAN |
Registration Number | SIN13993P |
Licence Holder | DKSH SINGAPORE PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | H05BX01 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
INDICATIONS
Secondary hyperparathyroidism in patients undergoing maintenance dialysis.
Dosing
DOSAGE AND ADMINISTRATION
The starting dosage for adults is 25 mg of cinacalcet once daily, to be orally administered.
With careful management of the patient's serum parathyroid hormone (PTH) and calcium levels, the dose may then be adjusted within a range of 25–75 mg once daily. If no improvement found in PTH, the dose may be increased up to 100 mg once daily. If dose increase is required, dose should be increased by 25 mg at a time, at intervals of at least 3 weeks.
1. Regpara has an effect of decreasing calcium in blood. Therefore, it should be confirmed that the patient's serum calcium level is not low (9.0 mg/dL (2.3mmol/L) or more) prior to administration.
2. The serum calcium level should be determined once a week at the start of administration and during the dose adjustment period, and at least once every two weeks during the maintenance period. If serum calcium level decreases to 8.4 mg/dL (2.1mmol/L) or less, the following measures should be taken.
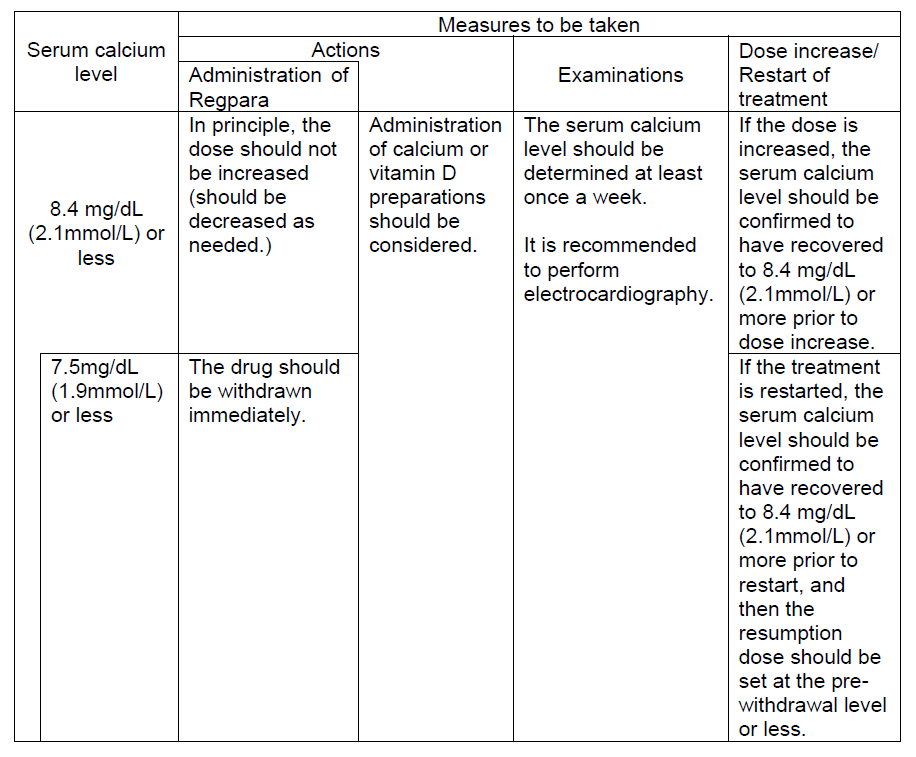
The serum calcium level should be determined before administration of Regpara so that the effect and safety of the drug may be properly evaluated. Furthermore, it is recommended that corrected serum calcium valuesNote) should be used as a guide in patients with hypoalbuminemia (serum albumin: <4.0 g/dL).
3. The serum PTH level should be periodically determined so that it may be maintained at the target level for management. It is recommended that the serum PTH level should be determined twice a month at the start of administration and during the dose adjustment period (about 3 months after the start of administration), and at least once a month after the serum PTH level is confirmed to be almost stable. The serum PTH level should be determined before administration of Regpara so that the effect and safety of the drug may be properly evaluated.
Note) Corrected serum calcium is calculated as follows:
Corrected serum calcium (mg/dL) = Serum calcium (mg/dL) − Serum albumin (g/dL) + 4.0
Contraindications
Table caption
CONTRAINDICATIONS (Regpara is contraindicated in the following patients.) |
