AKYNZEO CAPSULES 300MG/0.5MG [SIN15031P]
Active ingredients: AKYNZEO CAPSULES 300MG/0.5MG
Product Info
AKYNZEO CAPSULES 300MG/0.5MG
[SIN15031P]
Product information
Active Ingredient and Strength | NETUPITANT - 300 MG |
Dosage Form | CAPSULE, GELATIN COATED |
Manufacturer and Country | HELSINN BIREX PHARMACEUTICALS LTD. (INTERMEDIATE NETUPITANT TABLETS AND FINAL DRUG PRODUCT) - IRELAND |
Registration Number | SIN15031P |
Licence Holder | JUNIPER HEALTHCARE PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | A04AA55 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
1 INDICATIONS AND USAGE
AKYNZEO is indicated in adults for the:
Prevention of acute and delayed nausea and vomiting associated with highly emetogenic chemotherapy, including cisplatin.
Prevention of acute and delayed nausea and vomiting associated with moderately emetogenic cancer chemotherapy.
Dosing
2 DOSAGE AND ADMINISTRATION
Administration
One AKYNZEO capsule administered approximately 1 hour prior to the start of chemotherapy.
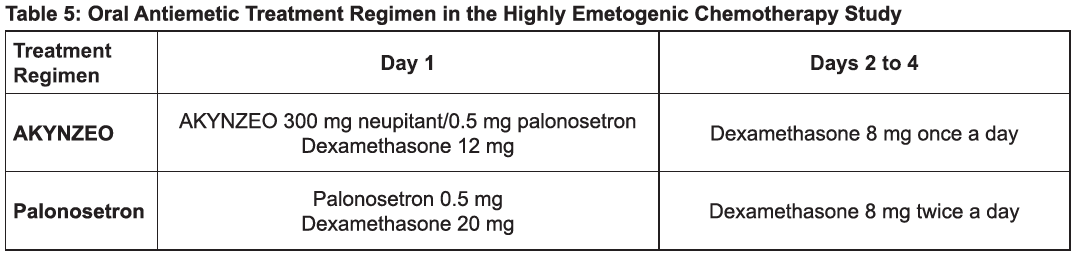
Highly Emetogenic Chemotherapy, including Cisplatin Based Chemotherapy
The recommended dosage in adults is one capsule of AKYNZEO administered approximately 1 hour prior to the start of chemotherapy with dexamethasone 12 mg administered orally 30 minutes prior to chemotherapy on day 1 and 8 mg orally once daily on days 2 to 4 [see Clinical Studies (14), Table 5)].
[From 14 CLINICAL STUDIES]
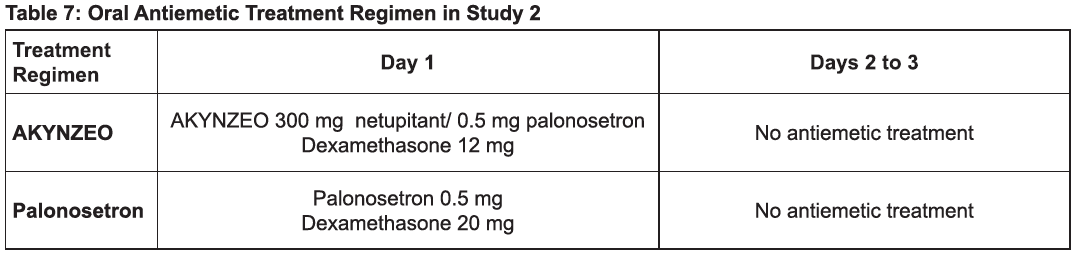
Moderately Emetogenic Chemotherapy / Anthracyclines and Cyclophosphamide-based Chemotherapy
The recommended dosage in adults is one capsule of AKYNZEO approximately 1 hour prior to the start of chemotherapy with dexamethasone 12 mg administered orally 30 minutes prior to chemotherapy on day 1. Administration of dexamethasone on days 2 to 4 is not necessary [see Clinical Studies (14), Table 7].
[From 14 CLINICAL STUDIES]
AKYNZEO can be taken with or without food.
Contraindications
4 CONTRAINDICATIONS
Pregnancy
