KYPROLIS POWDER FOR SOLUTION FOR INFUSION 60MG/VIAL [SIN15184P]
Active ingredients: KYPROLIS POWDER FOR SOLUTION FOR INFUSION 60MG/VIAL
Product Info
KYPROLIS POWDER FOR SOLUTION FOR INFUSION 60MG/VIAL
[SIN15184P]
Product information
Active Ingredient and Strength | CARFILZOMIB - 60 MG/VIAL |
Dosage Form | INJECTION, POWDER, LYOPHILIZED, FOR SOLUTION |
Manufacturer and Country | PATHEON MANUFACTURING SERVICES LLC (BULK PRODUCTION AND PRIMARY PACKAGING) - UNITED STATES |
Registration Number | SIN15184P |
Licence Holder | AMGEN BIOTECHNOLOGY SINGAPORE PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01XG02 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
1 INDICATIONS AND USAGE
Relapsed or Refractory Multiple Myeloma
Kyprolis is indicated for the treatment of adult patients with relapsed or refractory multiple myeloma who have received one to three lines of therapy in combination with:
Lenalidomide and dexamethasone; or
Dexamethasone; or
Daratumumab and dexamethasone.
Dosing
2 DOSAGE AND ADMINISTRATION
2.1 Administration Precautions
Hydration
Adequate hydration is required prior to dosing in Cycle 1, especially in patients at high-risk of tumor lysis syndrome (TLS) or renal toxicity. Consider hydration with both oral fluids (30 mL per kg at least 48 hours before Cycle 1, Day 1) and intravenous fluids (250 mL to 500 mL of appropriate intravenous fluid prior to each dose in Cycle 1). If needed, give an additional 250 mL to 500 mL of intravenous fluids following Kyprolis administration. Continue oral and/or intravenous hydration, as needed, in subsequent cycles.
Monitor patients for evidence of volume overload and adjust hydration to individual patient needs, especially in patients with or at risk for cardiac failure [see Warnings and Precautions (5.1, 5.3) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
Electrolyte Monitoring
Monitor serum potassium levels regularly during treatment with Kyprolis [see Adverse Reactions (6.1) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
Premedications and Concomitant Medications
Premedicate with dexamethasone administered as part of the combination therapy [see Dosage and Administration (2.2)]. Administer dexamethasone orally or intravenously at least 30 minutes but no more than 4 hours prior to all doses of Kyprolis during Cycle 1 to reduce the incidence and severity of infusion-related reactions [see Warnings and Precautions (5.9) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information]. Reinstate dexamethasone premedication if these symptoms occur during subsequent cycles.
Provide thromboprophylaxis for patients being treated with Kyprolis in combination with other therapies [see Warnings and Precautions (5.8) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
Consider antiviral prophylaxis to decrease the risk of herpes zoster reactivation [see Adverse Reactions (6.1) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
Dose Calculation
For patients with body surface area (BSA) of 2.2 m2 or less, calculate the Kyprolis dose using actual BSA. Dose adjustments do not need to be made for weight changes of 20% or less.
For patients with a BSA greater than 2.2 m2, calculate the Kyprolis dose using a BSA of 2.2 m2.
2.2 Recommended Dosage
Kyprolis in Combination with Lenalidomide and Dexamethasone
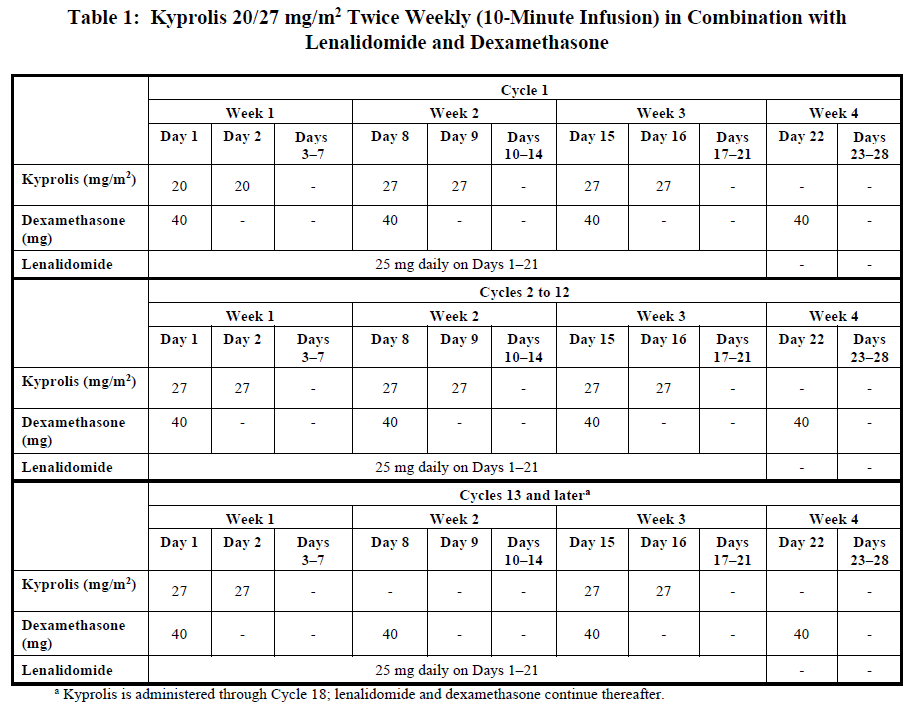
Administer Kyprolis intravenously as a 10-minute infusion on Days 1, 2, 8, 9, 15, and 16 of each 28-day cycle in combination with lenalidomide and dexamethasone until Cycle 12 as shown in Table 1 [see Clinical Studies (12.1) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information]. The recommended starting dose of Kyprolis is 20 mg/m2 on Cycle 1, Days 1 and 2. If tolerated, escalate the dose to 27 mg/m2 on Cycle 1, Day 8. From Cycle 13, administer Kyprolis on Days 1, 2, 15, 16 until Cycle 18. Discontinue Kyprolis after Cycle 18. Continue lenalidomide and dexamethasone until disease progression or unacceptable toxicity occurs. Refer to the Prescribing Information for lenalidomide and dexamethasone for additional dosage information.
Kyprolis in Combination with Dexamethasone
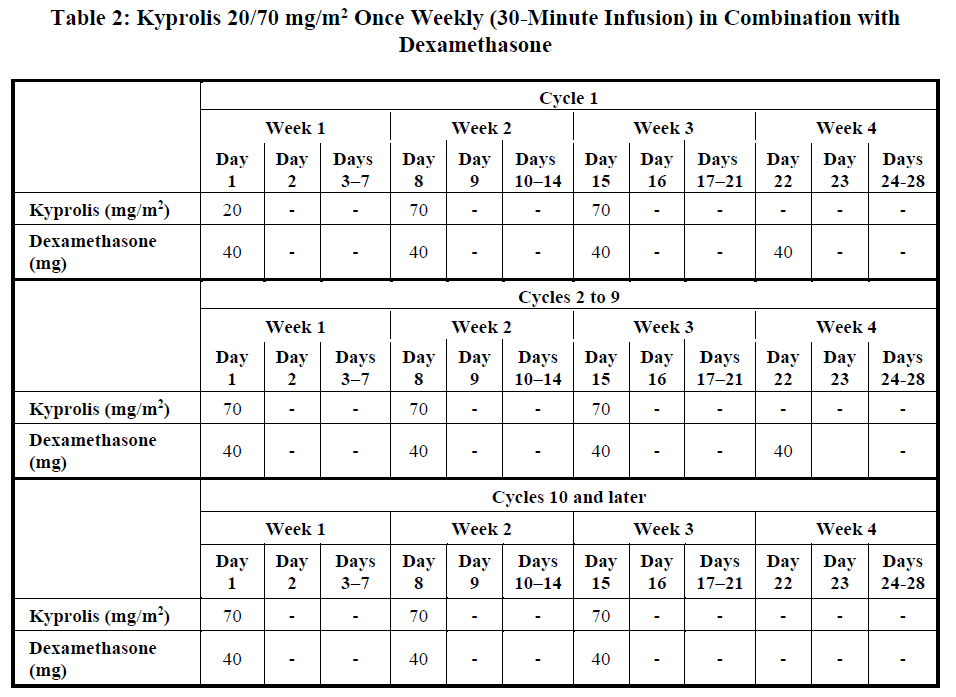
Once weekly 20/70 mg/m2 regimen by 30-minute infusion
Administer Kyprolis intravenously as a 30-minute infusion on Days 1, 8, and 15 of each 28-day cycle in combination with dexamethasone until disease progression or unacceptable toxicity as shown in Table 2 [see Clinical Studies (12.2) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information]. The recommended starting dose of Kyprolis is 20 mg/m2 on Cycle 1, Day 1. If tolerated, escalate the dose to 70 mg/m2 on Cycle 1, Day 8. Administer dexamethasone 30 minutes to 4 hours before Kyprolis. Refer to Prescribing Information for dexamethasone for additional dosage information.
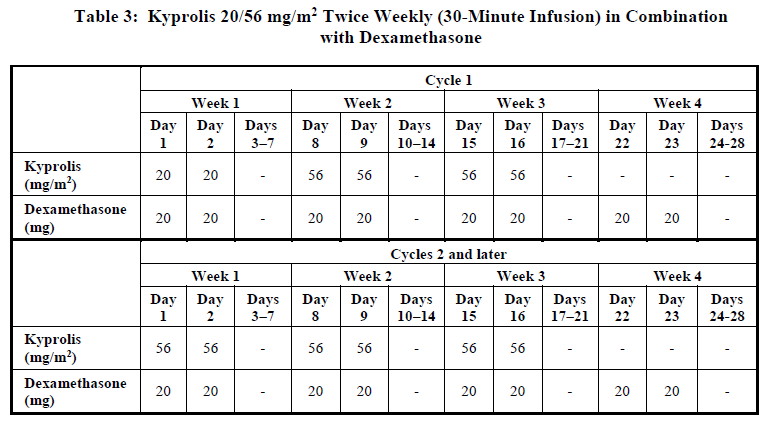
Twice weekly 20/56 mg/m2 regimen by 30-minute infusion
Administer Kyprolis intravenously as a 30-minute infusion on Days 1, 2, 8, 9, 15, and 16 of each 28-day cycle in combination with dexamethasone until disease progression or unacceptable toxicity as shown in Table 3 [see Clinical Studies (12.2) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information]. The recommended starting dose of Kyprolis is 20 mg/m2 on Cycle 1, Days 1 and 2. If tolerated, escalate the dose to 56 mg/m2 on Cycle 1, Day 8. Administer dexamethasone 30 minutes to 4 hours before Kyprolis. Refer to the Prescribing Information for dexamethasone for additional dosage information.
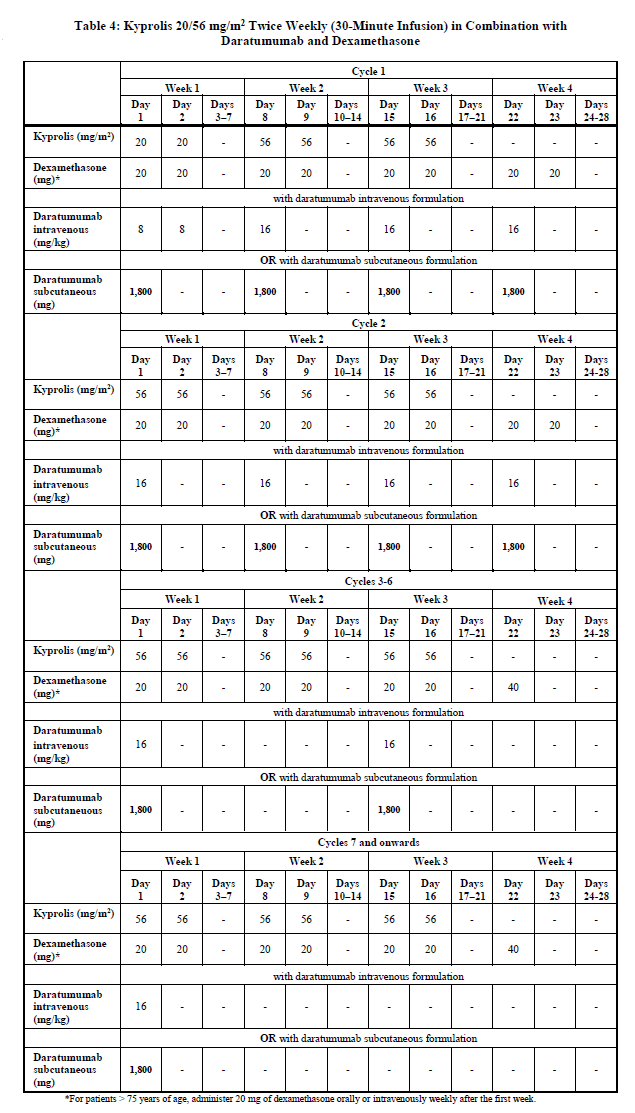
Kyprolis in Combination with Daratumumab and Dexamethasone
Twice weekly 20/56 mg/m2 regimen by 30-minute infusion
Administer Kyprolis intravenously as a 30-minute infusion on Days 1, 2, 8, 9, 15 and 16 of each 28-day cycle in combination with daratumumab and dexamethasone until disease progression or unacceptable toxicity as shown in Table 4 [see Clinical Studies (12.3) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information]. The recommended starting dose of Kyprolis is 20 mg/m2 on Cycle 1, Days 1 and 2. If tolerated, escalate the dose to 56 mg/m2 on Cycle 1, Day 8 and thereafter. Administer dexamethasone 30 minutes to 4 hours before Kyprolis and 1 to 3 hours before daratumumab. Refer to the Prescribing Information for daratumumab and dexamethasone for additional dosage information.
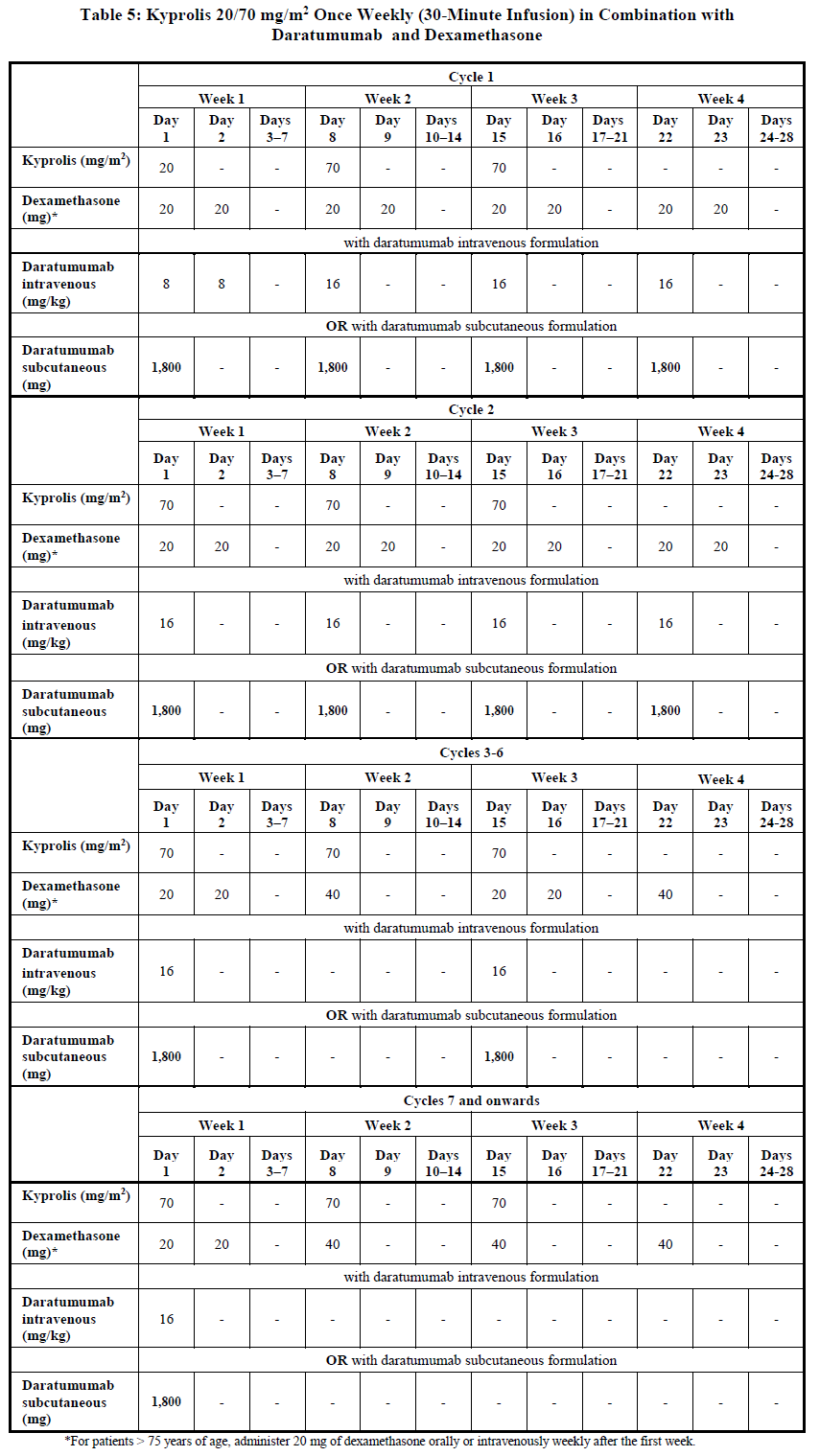
Once weekly 20/70 mg/m2 regimen by 30-minute infusion
Administer Kyprolis intravenously as a 30-minute infusion on Days 1, 8 and 15 of each 28-day cycle in combination with daratumumab and dexamethasone until disease progression or unacceptable toxicity as shown in Table 5 [see Clinical Studies (12.3) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information]. The recommended starting dose of Kyprolis is 20 mg/m2 on Cycle 1, Day 1. If tolerated, escalate the dose to 70 mg/m2 on Cycle 1, Day 8 and thereafter. Administer dexamethasone 30 minutes to 4 hours before Kyprolis and 1 to 3 hours before daratumumab. Refer to the Prescribing Information for daratumumab and dexamethasone for additional dosage information.
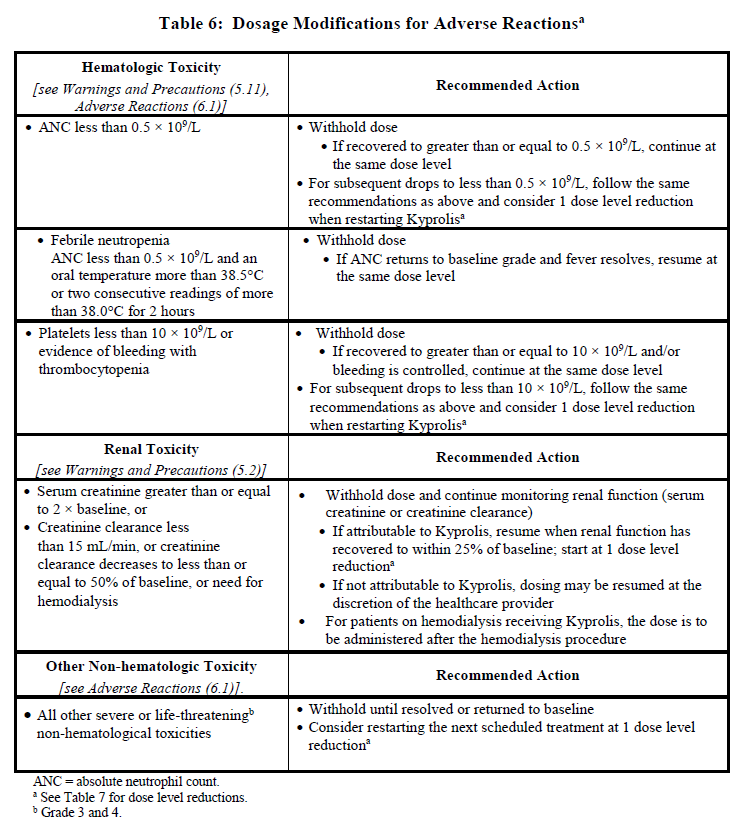
2.3 Dosage Modifications for Adverse Reactions
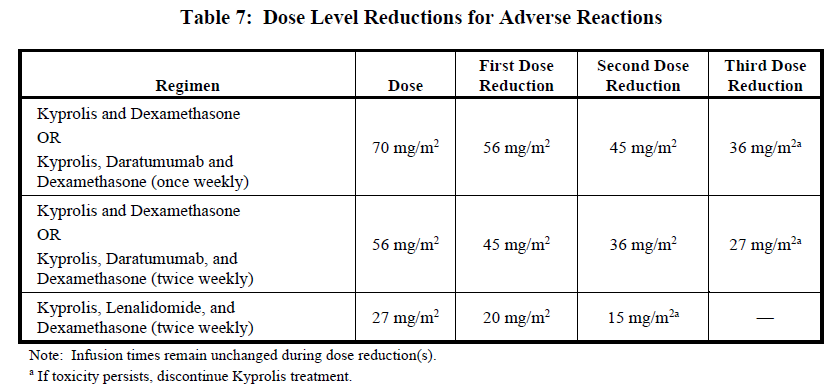
Recommended actions and dosage modifications for Kyprolis are presented in Table 6. Dose level reductions are presented in Table 7. See the lenalidomide, intravenous daratumumab, and dexamethasone Prescribing Information respectively for recommended dosage modifications associated with each product.
2.4 Dosage Modifications for Hepatic Impairment
For patients with mild (total bilirubin > 1 to 1.5 × ULN and any AST or total bilirubin ≤ ULN and AST > ULN) or moderate (total bilirubin > 1.5 to 3 × ULN and any AST) hepatic impairment, reduce the dose of Kyprolis by 25% [see Use in Specific Populations (7.6), Clinical Pharmacology (10.3) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
2.5 Recommended Dosage for End Stage Renal Disease
For patients with end stage renal disease who are on hemodialysis, administer Kyprolis after the hemodialysis procedure.
2.6 Preparation and Administration
Kyprolis vials contain no antimicrobial preservatives and are intended for single-dose only. The reconstituted solution contains carfilzomib at a concentration of 2 mg/mL.
Read the complete preparation instructions prior to reconstitution. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Reconstitution/Preparation Steps:
1. Remove vial from refrigerator just prior to use.
2. Calculate the dose (mg/m2) and number of vials of Kyprolis required using the patient’s BSA at baseline.
3. Aseptically reconstitute each Kyprolis vial only with Sterile Water for Injection, USP using the volumes described in Table 8. Use a 21-gauge or larger needle (0.8 mm or smaller external diameter needle) to reconstitute each vial by slowly injecting Sterile Water for Injection through the stopper and directing the Sterile Water for Injection onto the INSIDE WALL OF THE VIAL to minimize foaming. There is no data to support the use of closed system transfer devices with Kyprolis.

4. Gently swirl and/or invert the vial slowly for about 1 minute, or until complete dissolution. DO NOT SHAKE to avoid foam generation. If foaming occurs, allow the solution to settle in the vial until foaming subsides (approximately 5 minutes) and the solution is clear.
5. Visually inspect for particulate matter and discoloration prior to administration. The reconstituted product should be a clear, colorless solution and should not be administered if any discoloration or particulate matter is observed.
6. Discard any unused portion left in the vial. DO NOT pool unused portions from the vials. DO NOT administer more than one dose from a vial.
7. Administer Kyprolis directly by intravenous infusion or in a 50 mL to 100 mL intravenous bag containing 5% Dextrose Injection. Do not administer as an intravenous push or bolus.
8. When administering in an intravenous bag, use a 21-gauge or larger gauge needle (0.8 mm or smaller external diameter needle) to withdraw the calculated dose from the vial and dilute into 50 mL or 100 mL intravenous bag containing only 5% Dextrose Injection (based on the calculated total dose and infusion time).
9. Flush the intravenous administration line with 0.9% Sodium Chloride Injection or 5% Dextrose Injection immediately before and after Kyprolis administration.
10. Do not mix Kyprolis with or administer as an infusion with other medicinal products.
The stabilities of reconstituted Kyprolis under various temperature and container conditions are shown in Table 9.

Contraindications
4 CONTRAINDICATIONS
None.
