SALATAZ POWDER FOR SOLUTION FOR INJECTION/INFUSION 4.5G/VIAL [SIN15299P]
Active ingredients: SALATAZ POWDER FOR SOLUTION FOR INJECTION/INFUSION 4.5G/VIAL
Product Info
SALATAZ POWDER FOR SOLUTION FOR INJECTION/INFUSION 4.5G/VIAL
[SIN15299P]
Product information
Active Ingredient and Strength | PIPERACILLIN SODIUM 4.17 G EQV PIPERACILLIN - 4 G |
Dosage Form | INJECTION, POWDER, FOR SOLUTION |
Manufacturer and Country | SHANDONG ANXIN PHARMACEUTICAL CO., LTD. (INTERMEDIATE BULK DRUG PRODUCT MANUFACTURER) - CHINA |
Registration Number | SIN15299P |
Licence Holder | PAN-MALAYAN PHARMACEUTICALS PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J01CR05 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Piperacillin/tazobactam is indicated in the treatment of the following infections produced by sensitive micro-organisms (see section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information):
Serious infections of the lower airways.
Urinary tract infections (complicated or simple).
Intra-abdominal infections.
Infections of the skin and skin structure.
Bacterial septicaemia.
Initial empirical treatment in patients with episodes of Febrile neutropenia*.
Polymicrobial infections produced by aerobe and anaerobe micro-organisms.
National guidelines concerning bacterial resistances and the use and appropriate prescription of antibiotics must be taken into account.
*For treatment of episodes of Febrile neutropenia, piperacillin/tazobactam should be used in combination with another antibiotic of suitable spectrum, preferably an aminoglycoside. Treatment must be initiated empirically, on the appearance of fever in neutropenic patients.
Dosing
4.2 Posology and method of administration
Adults and children over 12 years of age
Total daily dose depends on the severity and location of the infection and may vary from 2/0.25 g to 4/0.5 g piperacillin/tazobactam administered every six or eight hours.
The usual dose with normal renal function is 4 g piperacillin/0.5 g tazobactam administered every 8 hours.
For patients with febrile neutropenia, recommended dose is 4 g piperacillin/0.5 g tazobactam administered every 6 hours in conjunction with an aminoglycoside.
Children under 12 years of age
*Intra-abdominal infections
The recommended doses for this indication are:
– For children between 2 and 12 years old, with weighing ≤ 40 kg, and with a normal renal function, the recommended dose is 100 mg of piperacillin/12.5 mg of tazobactam per kilogram weight every 8 hours.
– For children aged 2 and 12 years old, with weighing more than 40 kg and with normal renal function, the recommended dose is the same as for adults, that is, 4 g piperacillin/0.5 g tazobactam every 8 hours.
*Episodes of febrile neutropenia
In children weighing less than 50 kg, the dose should be adjusted to 80 mg/kg of piperacillin/10 mg/kg of tazobactam, administered every 6 hours in combination with an aminoglycoside.
In children weighing over 50kg, follow adult dosing guidance, including the aminoglycosides.
Until more experience is available, piperacillin/tazobactam should not be used in children who do not have neutropenia.
Duration of the treatment
The duration of the treatment will depend on the severity of the infection and the bacteriological and clinical response of the patient. The recommended duration is a minimum of 5 days and a maximum of 14 days, considering that administration should be stopped at least 48 hours after disappearance of the clinical signs and symptoms.
In general, treatment should continue for forty-eight hours after the symptoms and fever have disappeared.
Elderly patients
No dose adjustment is required on the basis of age alone. Nevertheless, in this population group the dose should be adjusted in the presence of impaired renal function.
Patients with impaired renal function
A dose adjustment is required in patients with impaired renal function.
– Adults and children over 12 years old weighing >50kg with renal insufficiency
Suggested daily doses are as follows:

The maximum daily dose for patients on haemodialysis is 8/1 g of piperacillin/tazobactam. Furthermore, as haemodialysis eliminates 30% – 50% of the piperacillin in 4 hours, an additional dose of 2/0.25 g of piperacillin/tazobactam should be administered after each dialysis session.
– Adults and children over 12 years old weighing <50kg with renal insufficiency

For children weighing <50kg on hemodialysis, the recommended doses is 45mg/kg every 8 hours.
The pharmacokinetics of piperacillin/tazobactam have not been evaluated in children between 2 and 12 years of age with renal impairment. Each patient must be monitored closely for signs of drug toxicity. Drug dose and interval dose should be adjusted accordingly. In patients with renal insufficiency or hemodialysis patients, intravenous dosages and administration intervals should be adjusted to the degree of renal function impairment.
There is no information on dose adjustment in neutropenic patients with impaired renal function.
Patients with impaired liver functions
No adjustment of the dose of piperacillin/tazobactam is required in patients with liver failure.
Duration of therapy
Therapy is recommended for a minimum of 5 days and maximum of 14 days, considering that dose administration should continue at least 48 hours after the resolution of clinical signs and symptoms or fever.
Method of administration
SALATAZ (Piperacillin/Tazobactam) 4/0.5 g can be administered by slow intravenous injection (3 – 5 minutes) or by slow intravenous infusion (20 – 30 minutes).
Whenever SALATAZ is used concurrently with another antibiotic, especially an aminoglycoside, the drug must not be mixed in intravenous solutions or administered concurrently due to physical incompatibility.
SALATAZ should not be mixed with other drugs in a syringe or infusion bottle since compatibility has not been established. In particular, whenever SALATAZ is used concurrently with another antibiotic, especially an aminoglycoside (with the exceptions shown below), the SALATAZ must not be mixed in intravenous solutions or administered concurrently due to physical incompatibility.
Co-administration of Piperacillin/Tazobactam with Aminoglycosides:
Due to the in vitro inactivation of the aminoglycoside by the β-lactam antibiotics, piperacillin/tazobactam and the aminoglycoside are recommended for separate administration.
Piperacillin/tazobactam and the aminoglycoside should be reconstituted and diluted separately when concomitant therapy with aminoglycosides is indicated.
The following compatibility information does not apply to the piperacillin/tazobactam formulation not containing EDTA.
In circumstances where co-administration is preferred, the reformulated piperacillin/tazobactam containing EDTA supplied in vials is compatible for simultaneous coadministration via Y-site infusion only with the following aminoglycosides under the following conditions:
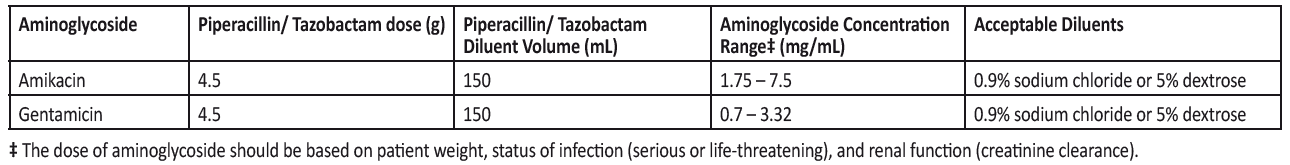
Compatibility of piperacillin/tazobactam with other aminoglycosides has not been established. Only the concentration and diluents for amikacin and gentamicin with the dosages of piperacillin/tazobactam listed in the table above have been established as compatible for co-administration via Y-site infusion. Simultaneous co-administration via Y-site infusion in any manner other than listed above may result in inactivation of the aminoglycoside by piperacillin/tazobactam.
Contraindications
4.3 Contraindications
Piperacillin/Tazobactam is contraindicated in patients with a history of hypersensitivity to any penicillin and/or cephalosporins or beta-lactamase inhibitors.
