CALQUENCE HARD CAPSULES 100MG [SIN15753P]
Active ingredients: CALQUENCE HARD CAPSULES 100MG
Product Info
CALQUENCE HARD CAPSULES 100MG
[SIN15753P]
Product information
Active Ingredient and Strength | ACALABRUTINIB - 100 MG |
Dosage Form | CAPSULE |
Manufacturer and Country | ASTRAZENECA AB - SWEDEN |
Registration Number | SIN15753P |
Licence Holder | ASTRAZENECA SINGAPORE PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01EL02 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Mantle Cell Lymphoma (MCL)
CALQUENCE as monotherapy is indicated for the treatment of patients with mantle cell lymphoma (MCL) who have received at least one prior therapy.
CALQUENCE in combination with bendamustine and rituximab (BR) is indicated for the treatment of adult patients with previously untreated MCL who are ineligible for autologous stem cell transplant (ASCT).
Chronic Lymphocytic Leukaemia (CLL) / Small Lymphocytic Lymphoma (SLL)
CALQUENCE is indicated:
in combination with obinutuzumab or as monotherapy for the treatment of patients with previously untreated chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL).
in combination with venetoclax with or without obinutuzumab for the treatment of patients with previously untreated CLL/SLL.
as monotherapy for the treatment of patients with CLL/SLL who have received at least one prior therapy.
Dosing
4.2 Posology and method of administration
Treatment with CALQUENCE should be initiated and supervised by a physician experienced in the use of anticancer therapies.
Posology
MCL
The recommended dose of CALQUENCE in monotherapy or in combination with other medicinal products is 100 mg (1 capsule) twice daily. For the combination regimens, refer to the prescribing information of each of the medicinal product for dosing information. For details of the combination regimens, see section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
CALQUENCE in combination with bendamustine and rituximab
CALQUENCE should be administered on Day 1 on Cycle 1 (each cycle is 28 days) until disease progression or unacceptable toxicity. Bendamustine should be administered at 90 mg/m2 on Days 1 and 2 of each cycle for a total of 6 cycles. Rituximab should be administered at 375 mg/m2 on Day 1 each cycle for a total of 6 cycles. Patients achieving a response (partial response [PR] or complete response [CR]) after the first 6 cycles, may receive maintenance rituximab at 375 mg/m2 on Day 1 of every other cycle for a maximum of 12 additional doses, starting on Cycle 8 up to Cycle 30.
CLL/SLL
The recommended dose of CALQUENCE for the treatment of CLL/SLL is 100 mg (1 capsule) twice daily, either as monotherapy or in combination. Refer to the prescribing information of each combination medicinal product for recommended dosing information (for details of the combination regimen, see section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
CALQUENCE doses should be separated by approximately 12 hours.
Treatment with CALQUENCE in monotherapy or in combination with obinutuzumab should continue until disease progression or unacceptable toxicity.
Treatment with CALQUENCE in combination with venetoclax with or without obinutuzumab should continue until disease progression, unacceptable toxicity or completion of 14 cycles of treatment (each cycle is 28 days).
Missed Dose
If a patient misses a dose of CALQUENCE by more than 3 hours, instruct the patient to take the next dose at its regularly scheduled time. Extra capsules of CALQUENCE should not be taken to make up for a missed dose.
Dose Adjustments
Adverse Reactions
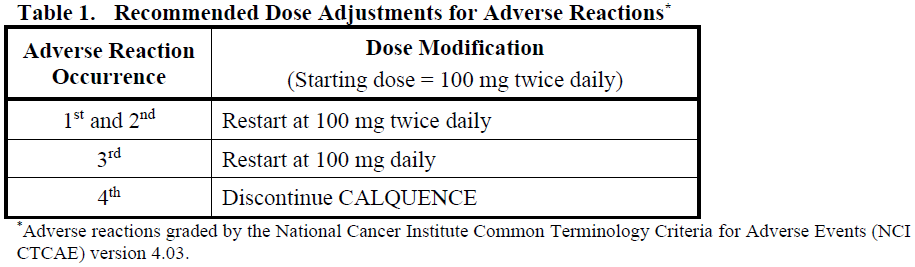
Recommended dose modifications of CALQUENCE for Grade ≥ 3 adverse reactions in patients receiving CALQUENCE monotherapy and CALQUENCE in combination with obinutuzumab or in combination with venetoclax with or without obinutuzumab are provided in Table 1.
Temporarily interrupt CALQUENCE to manage a Grade ≥ 3 non-haematological treatment-related adverse reaction, Grade 3 thrombocytopenia with significant bleeding, Grade 4 thrombocytopenia, or Grade 4 neutropenia lasting longer than 7 days. Upon resolution of the adverse reaction to Grade 1 or baseline (recovery), restart CALQUENCE as recommended in Table 1.
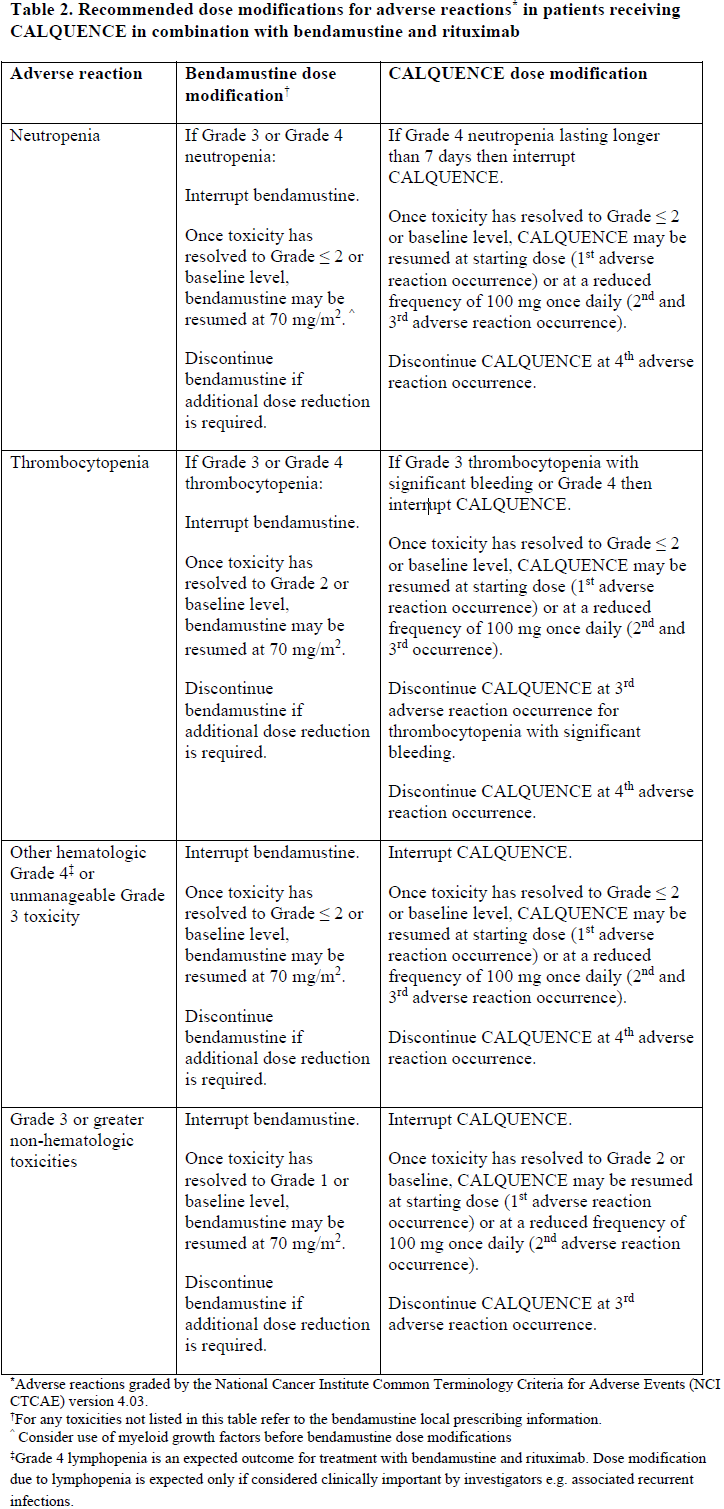
Dose modifications for adverse reactions in patients receiving CALQUENCE in combination with bendamustine and rituximab are listed in Table 2.
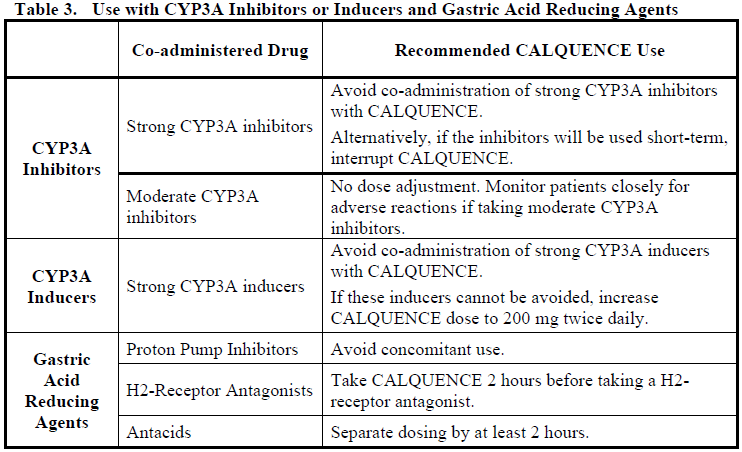
Refer to the prescribing information of each of the medicinal products used in combination with CALQUENCE for additional information for management of toxicities.
Method of Administration
CALQUENCE should be swallowed whole with water at approximately the same time each day. CALQUENCE can be taken with or without food. The capsule should not be chewed, dissolved, or opened.
Special patient populations
Elderly (≥ 65 years)
No dose adjustment is necessary based on age (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal Impairment
No dose adjustment is recommended in patients with mild to moderate renal impairment (eGFR greater than or equal to 30 mL/min/1.73m2 as estimated by MDRD (modification of diet in renal disease equation)). The pharmacokinetics and safety of CALQUENCE in patients with severe renal impairment (eGFR less than 29 mL/min/1.73m2) or end-stage renal disease have not been studied (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Hepatic Impairment
No dose adjustment is recommended in patients with mild or moderate hepatic impairment (Child-Pugh A, Child-Pugh B, or total bilirubin between 1.5–3 times the upper limit of normal [ULN] and any AST). Avoid administration of CALQUENCE in patients with severe hepatic impairment (Child-Pugh C or total bilirubin > 3 times ULN and any AST) (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Severe Cardiac Disease
Patients with severe cardiovascular disease were excluded from CALQUENCE clinical studies.
Paediatric and adolescents
The safety and efficacy of CALQUENCE in children and adolescents aged less than 18 years have not been established.
Contraindications
4.3 Contraindications
Hypersensitivity to acalabrutinib or to any of the excipients in the formulation.
