PFIZER BORTEZOMIB POWDER FOR INJECTION 3.5MG [SIN15900P]
Active ingredients: PFIZER BORTEZOMIB POWDER FOR INJECTION 3.5MG
Product Info
PFIZER BORTEZOMIB POWDER FOR INJECTION 3.5MG
[SIN15900P]
Product information
Active Ingredient and Strength | BORTEZOMIB (AS A MANNITOL BORONIC ESTER) - 3.5 MG |
Dosage Form | INJECTION, POWDER, FOR SOLUTION |
Manufacturer and Country | GLAND PHARMA LIMITED - INDIA |
Registration Number | SIN15900P |
Licence Holder | PFIZER PRIVATE LIMITED |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01XG01 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Pfizer Bortezomib Powder for Injection is indicated as part of combination therapy for the treatment of patients with previously untreated multiple myeloma.
Pfizer Bortezomib Powder for Injection is indicated as monotherapy for the treatment of patients with multiple myeloma who have received at least 1 prior therapy.
Pfizer Bortezomib Powder for Injection is indicated as monotherapy for the treatment of patients with mantle cell lymphoma who have received at least 1 prior therapy.
Pfizer Bortezomib Powder for Injection in combination with rituximab, cyclophosphamide, doxorubicin and prednisone is indicated for the treatment of adult patients with previously untreated mantle cell lymphoma who are unsuitable for haematopoietic stem cell transplantation.
Dosing
4.2 Posology and method of administration
Pfizer Bortezomib Powder for Injection may be administered:
Intravenously (at a concentration of 1 mg/mL) as a 3–5 second bolus injection or
Subcutaneously (at a concentration of 2.5 mg/mL)
Because each route of administration has a different reconstituted concentration, caution should be used when calculating the volume to be administered.
At least 72 hours should elapse between consecutive doses of Pfizer Bortezomib Powder for Injection.
PFIZER BORTEZOMIB POWDER FOR INJECTION IS FOR INTRAVENOUS OR SUBCUTANEOUS USE ONLY. Intrathecal administration has resulted in death.
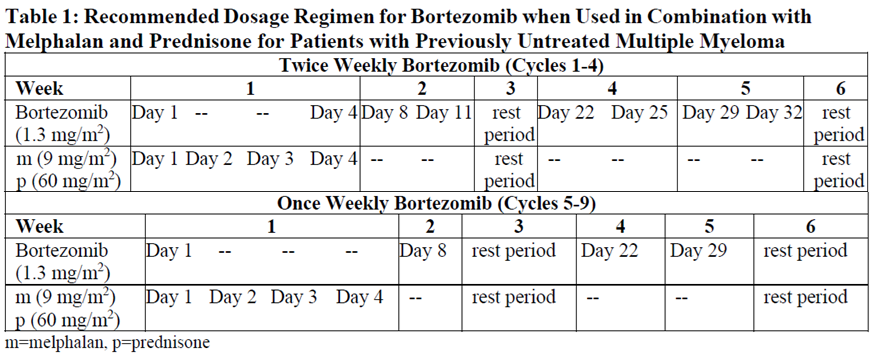
Recommended Dosage
Previously Untreated Multiple Myeloma – Non-Transplant Eligible
Pfizer Bortezomib Powder for Injection is administered in combination with oral melphalan and oral prednisone for nine 6-week treatment cycles as shown in Table 1. In Cycles 1–4, bortezomib is administered twice weekly (Days 1, 4, 8, 11, 22, 25, 29 and 32). In Cycles 5–9, bortezomib is administered once weekly (Days 1, 8, 22 and 29).
Dose Management Guidelines
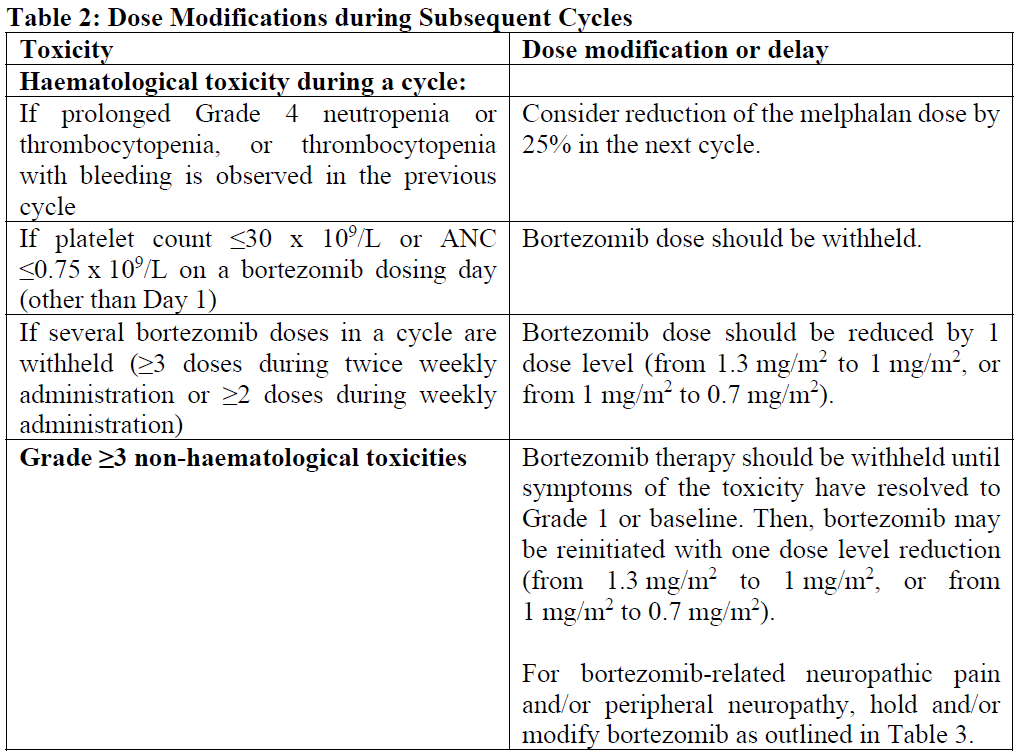
Dose modification and reinitiation of therapy when bortezomib is administered in combination with melphalan and prednisone
Prior to initiating a new cycle of therapy:
Platelet count should be ≥70 x 109/L and the absolute neutrophils count (ANC) should be ≥1.0 x 109/L
Non-haematological toxicities should have resolved to Grade 1 or baseline
For additional information concerning melphalan and prednisone, see manufacturer’s prescribing information.
Relapsed/Refractory Multiple Myeloma and Relapsed Mantle Cell Lymphoma
Recommended Dose
The recommended dose of bortezomib is 1.3 mg/m2/dose administered twice weekly for two weeks (Days 1, 4, 8, and 11) followed by a 10-day rest period (Days 12–21). This 3-week period is considered a treatment cycle. At least 72 hours should elapse between consecutive doses of bortezomib.
It is recommended that patients with a confirmed complete response receive 2 additional cycles of bortezomib beyond a confirmation. It is also recommended that responding patients who do not achieve a complete remission receive a total of 8 cycles of bortezomib therapy.
For extended therapy of more than 8 cycles, bortezomib may be administered on the standard schedule or, for relapsed multiple myeloma, on a maintenance schedule of once weekly for 4 weeks (Days 1, 8, 15, and 22) followed by a 13-day rest period (Days 23 to 35) (see section 5.1 for a summary of dose administration during clinical trials – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Dose Modification and Reinitiation of Therapy
Bortezomib therapy should be withheld at the onset of any Grade 3 non-haematological or Grade 4 haematological toxicities excluding neuropathy as discussed above (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). Once the symptoms of the toxicity have resolved, bortezomib therapy may be reinitiated at a 25% reduced dose (1.3 mg/m2/dose reduced to 1.0 mg/m2/dose; 1.0 mg/m2/dose reduced to 0.7 mg/m2/dose).
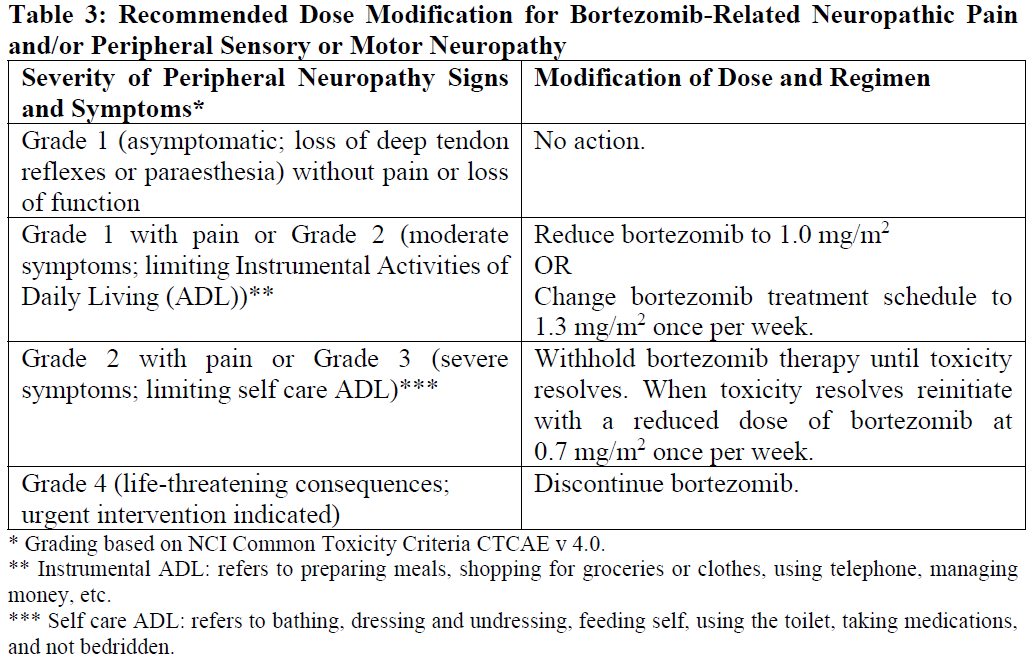
Table 3 contains the recommended dose modification for the management of patients who experience bortezomib-related neuropathic pain and/or peripheral sensory neuropathy. Severe autonomic neuropathy resulting in treatment interruption or discontinuation has been reported. Patients with pre-existing severe neuropathy should be treated with bortezomib only after careful risk/benefit assessment.
Previously Untreated Mantle Cell Lymphoma
Recommended Dosage in Combination with Rituximab, Cyclophosphamide, Doxorubicin and Prednisone
Pfizer Bortezomib Powder for Injection is administered at the recommended dose of 1.3 mg/m2 body surface area twice weekly for two weeks on Days 1, 4, 8, and 11, followed by a 10-day rest period on Days 12–21. This 3-week period is considered a treatment cycle. Six bortezomib cycles are recommended, although for patients with a response first documented at Cycle 6, two additional bortezomib cycles may be given. At least 72 hours should elapse between consecutive doses of bortezomib.
The following medicinal products are administered on Day 1 of each bortezomib 3-week treatment cycle as intravenous infusions: rituximab at 375 mg/m2, cyclophosphamide at 750 mg/m2, and doxorubicin at 50 mg/m2.
Prednisone is administered orally at 100 mg/m2 on Days 1, 2, 3, 4 and 5 of each treatment cycle.
Dose Adjustments during Treatment for Patients with Previously Untreated Mantle Cell Lymphoma
Prior to initiating a new cycle of therapy:
Platelet count should be ≥100 x 109/L and ANC should be ≥1.5 x 109/L
Haemoglobin should be ≥8 g/dL
Non-haematologic toxicity should have recovered to Grade 1 or baseline
Bortezomib treatment must be withheld at the onset of any ≥Grade 3 bortezomib-related nonhaematological toxicities (excluding neuropathy) or ≥Grade 3 haematological toxicities (see also section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). For dose adjustments, see Table 4 below. Colony stimulating factors may be administered for haematologic toxicity according to local standard practice. Platelet transfusion for the treatment of thrombocytopenia may be considered.

In addition, when bortezomib is given in combination with other chemotherapeutic medicinal products, appropriate dose reductions for these medicinal products should be considered in the event of toxicities, according to the recommendations in the respective Product Information documents.
Patients with Renal Impairment
Based on the data from a small study, the pharmacokinetics of bortezomib are not influenced by mild (CrCL=40–59 mL/min/1.73 m2, n=10) or moderate (CrCL=20–39 mL/min/1.73 m2, n=9) renal impairment. Therefore, dosing adjustments of bortezomib are not necessary for these patients. The effect of severe renal impairment (CrCL<20 mL/min/1.73 m2) has not been determined. Since dialysis may reduce bortezomib concentrations, the drug should be administered after the dialysis procedure (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
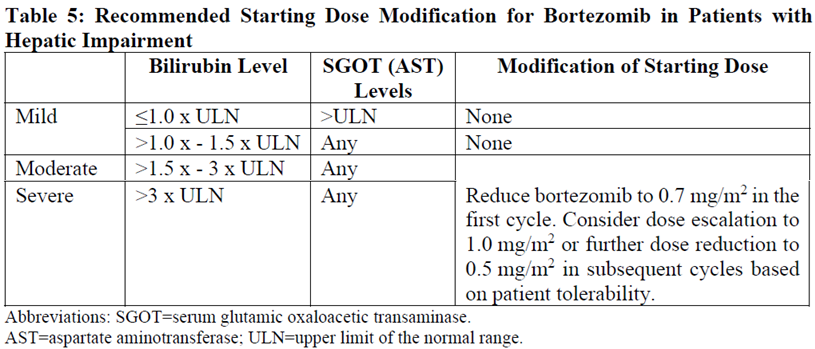
Patients with Hepatic Impairment
Patients with mild hepatic impairment do not require a starting dose adjustment and should be treated per the recommended bortezomib dose. Patients with moderate or severe hepatic impairment should be started on bortezomib at a reduced dose of 0.7 mg/m2 per injection during the first cycle, and a subsequent dose escalation to 1.0 mg/m2 or further dose reduction to 0.5 mg/m2 may be considered based on patient tolerance (see Table 5).
Paediatric Use
The safety and effectiveness of bortezomib in children has not been established.
Method of Administration
Intravenous injection (IV)
Bortezomib is administered as a 3–5 second bolus intravenous injection through a peripheral or central intravenous catheter followed by a flush with 0.9% sodium chloride solution for injection.
Subcutaneous injection (SC)
The reconstituted solution is injected into the thighs (right or left) or abdomen (right or left). Injection sites should be rotated for successive injections.
If local injection site reactions occur following bortezomib injection subcutaneously, a less concentrated bortezomib solution (1 mg/mL instead of 2.5 mg/mL) may be administered subcutaneously or change to IV injection.
When bortezomib is given in combination with other medicinal products, refer to the Product Information for these products for instructions for administration.
Contraindications
4.3 Contraindications
Pfizer Bortezomib Powder for Injection is contraindicated in patients with acute diffuse infiltrative pulmonary and pericardial disease and hypersensitivity to bortezomib, boron or mannitol.
