REBLOZYL POWDER FOR SOLUTION FOR INJECTION 25 MG/VIAL [SIN16470P]
Active ingredients: REBLOZYL POWDER FOR SOLUTION FOR INJECTION 25 MG/VIAL
Product Info
REBLOZYL POWDER FOR SOLUTION FOR INJECTION 25 MG/VIAL
[SIN16470P]
Product information
Active Ingredient and Strength | LUSPATERCEPT - 25 MG/VIAL |
Dosage Form | INJECTION, POWDER, FOR SOLUTION |
Manufacturer and Country | PATHEON ITALIA S.P.A. - ITALY |
Registration Number | SIN16470P |
Licence Holder | BRISTOL-MYERS SQUIBB (SINGAPORE) PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | B03XA06 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Reblozyl is indicated in adults for the treatment of transfusion-dependent anaemia due to very low, low and intermediate-risk myelodysplastic syndromes (MDS) (see section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Reblozyl is indicated in adults for the treatment of anaemia associated with transfusion-dependent and non-transfusion-dependent beta-thalassaemia (see section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Limitation of use
Reblozyl is not indicated for use as a substitute for red blood cell (RBC) transfusions in patients who require immediate correction of anaemia.
Dosing
4.2 Posology and method of administration
Reblozyl treatment should be initiated by a physician experienced in treatment of haematological diseases.
Posology
Prior to each Reblozyl administration, the haemoglobin (Hb) level of patients should be assessed. In case of a red blood cell (RBC) transfusion occurring prior to dosing, the pre-transfusion Hb level must be considered for dosing purposes.
The recommended starting dose of Reblozyl is 1.0 mg/kg administered once every 3 weeks.
Myelodysplastic syndromes
The recommended desired Hb concentration range is between 10 g/dL and 12 g/dL. Dose increase for insufficient response is provided below.
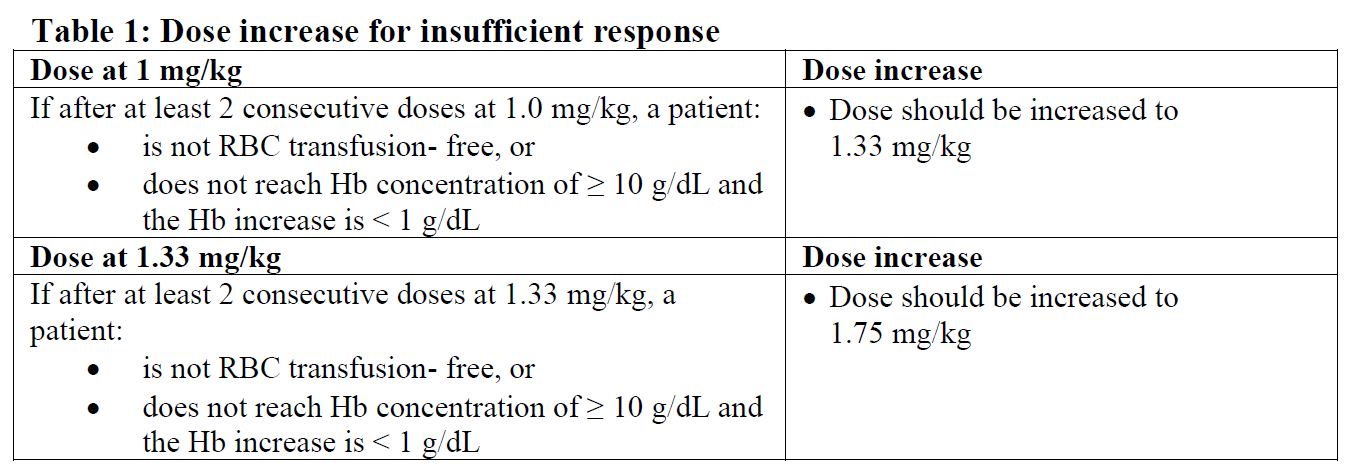
The dose increase should not occur more frequently than every 6 weeks (2 administrations) and should not exceed the maximum dose of 1.75 mg/kg every 3 weeks. The dose should not be increased immediately after a dose delay.
For patients with a pre-dose Hb level of > 9 g/dL and who have not yet achieved transfusion independence, a dose increase may be required at the physician’s discretion; the risk of Hb increasing above the target threshold with concomitant transfusion cannot be excluded.
If a patient loses response (i.e. transfusion independence), the dose should be increased by one dose level (see Table 2).
Transfusion-dependent β-thalassaemia
In patients who do not achieve a response after ≥ 2 consecutive doses (6 weeks), at the 1.0 mg/kg starting dose, the dose should be increased to 1.25 mg/kg. Minimum response is defined as a reduction in RBC transfusion burden by at least a third after ≥ 2 consecutive doses (6 weeks), at the 1.0 mg/kg starting dose. The dose should not be increased beyond the maximum dose of 1.25 mg/kg every 3 weeks.
If a patient loses response (if the RBC transfusion burden increases again after an initial response) the dose should be increased by one dose level (see Table 3).
Non-transfusion-dependent β-thalassaemia
In patients who do not achieve or maintain a response, defined as an increase from baseline in pre-dose Hb of ≥1 g/dL, after ≥ 2 consecutive doses (6 weeks) at the same dose level (in absence of transfusions, i.e., at least 3 weeks after the last transfusion), the dose should be increased by one dose level (see Table 2). The dose should not exceed the maximum dose of 1.25 mg/kg every 3 weeks.
Increase to next dose level
Increase to next dose level based on current dose is provided below.


Dose reduction and dose delay
In case of Hb increase > 2 g/dL within 3 weeks in absence of transfusion, compared with the Hb value at previous dose, Reblozyl dose should be reduced by one dose level.
If the Hb is ≥ 11.5 g/dL in the absence of transfusion for at least 3 weeks, the dose should be delayed until the Hb is ≤ 11.0 g/dL. If there is also a concomitant rapid increase in Hb from the Hb value at previous dose (> 2 g/dL within 3 weeks in absence of transfusion), a dose reduction to one step down should be considered after the dose delay.
Dose should not be reduced below 0.8 mg/kg (for MDS or transfusion-dependent β-thalassaemia) and below 0.6 mg/kg (for non-transfusion-dependent β-thalassaemia).
Reduced dose during treatment with luspatercept are provided below.


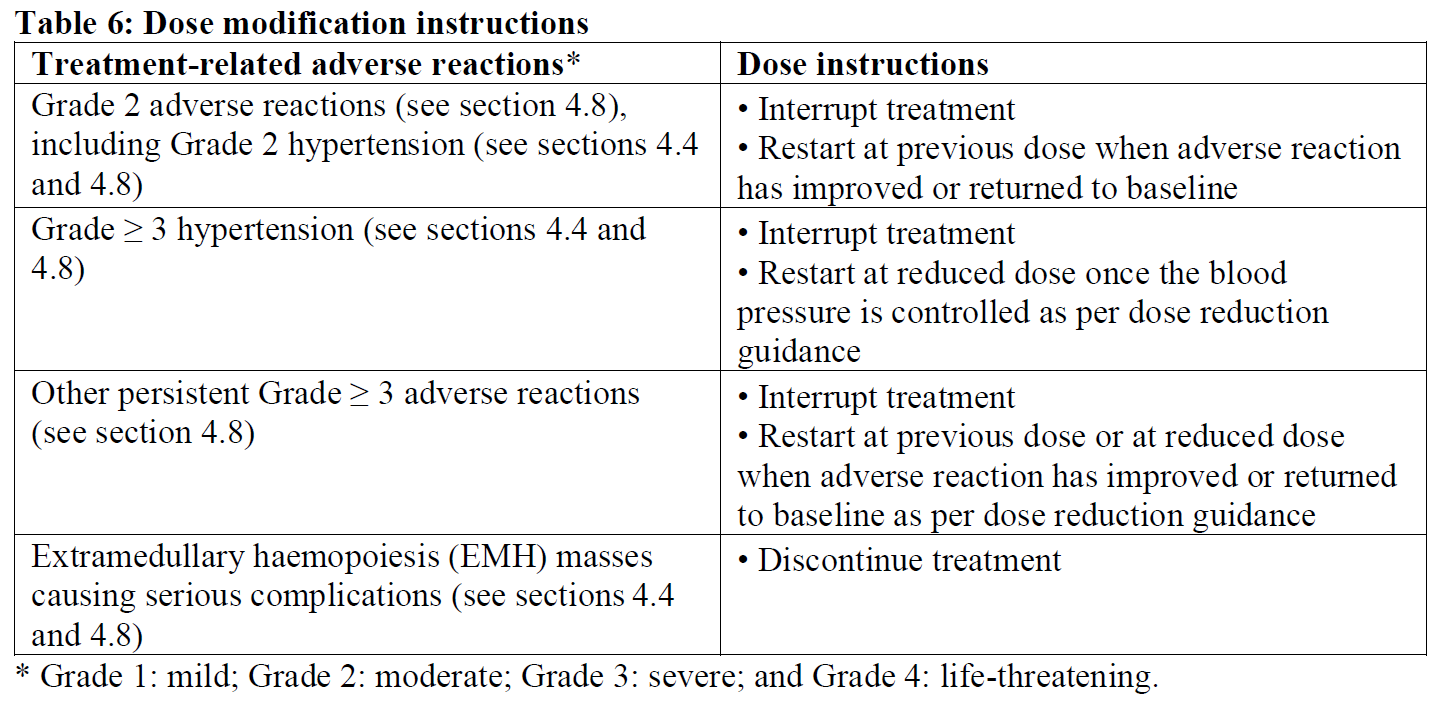
Dose modification due to adverse reactions
Instructions on dose interruptions or reductions for luspatercept treatment-related adverse reactions are outlined in Table 6.
Missed doses
In case of a missed or delayed scheduled treatment administration, the patient should be administered Reblozyl as soon as possible and dosing continued as prescribed with at least 3 weeks between doses.
Patients experiencing a loss of response
If patients experience a loss of response to Reblozyl, causative factors (e.g. a bleeding event) should be assessed. If typical causes for a loss of haematological response are excluded, dose increase should be considered as described above for the respective indication being treated (see Table 2 and Table 3).
Discontinuation
Reblozyl should be discontinued if patients do not experience a reduction in transfusion burden (for transfusion-dependent β-thalassaemia patients), or an increase from baseline Hb in the absence of transfusions (for non-transfusion-dependent β-thalassaemia patients), or a decrease in transfusion burden including no increase from baseline Hb (for MDS patients) after 9 weeks of treatment (3 doses) at the maximum dose level if no alternative explanations for response failure are found (e.g. bleeding, surgery, other concomitant illnesses) or if unacceptable toxicity occurs at any time.
Special populations
Elderly
No starting dose adjustment is required for Reblozyl (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). Limited data are available in β-thalassaemia patients ≥ 60 years of age.
Hepatic impairment
No starting dose adjustment is required for patients with total bilirubin (BIL) > upper limit of normal (ULN) and/or alanine aminotransferase (ALT) or aspartate aminotransferase (AST) < 3 x ULN (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). No specific dose recommendation can be made for patients with ALT or AST ≥ 3 x ULN or liver injury CTCAE Grade ≥ 3 due to lack of data (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal impairment
No starting dose adjustment is required for patients with mild or moderate renal impairment (individual estimated glomerular filtration rate [eGFR] 30 to 89 mL/min). Only limited data are available for patients with moderate renal impairment.
No specific dose recommendation can be made for patients with severe renal impairment (individual eGFR < 30 mL/min) due to lack of clinical data (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). Patients with renal impairment at baseline have been observed to have higher exposure (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). Consequently, these patients should be closely monitored for adverse reactions and dose adjustment should be managed accordingly (see Table 6).
Paediatric population
There is no relevant use of Reblozyl in the paediatric population for the indication of myelodysplastic syndromes, or in paediatric patients less than 6 months of age in β-thalassaemia.
The safety and efficacy of Reblozyl in the paediatric patients aged from 6 months to less than 18 years have not yet been established in β-thalassaemia. For non-clinical data, see section 5.3 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Method of administration
For subcutaneous use.
After reconstitution, Reblozyl solution should be injected subcutaneously into the upper arm, thigh or abdomen. The exact total dosing volume of the reconstituted solution required for the patient should be calculated and slowly withdrawn from the single-dose vial(s) into a syringe.
The recommended maximum volume of medicinal product per injection site is 1.2 mL. If more than 1.2 mL is required, the total volume should be divided into separate similar volume injections and administered across separate sites using the same anatomical location but on opposite sides of the body.
If multiple injections are required, a new syringe and needle must be used for each subcutaneous injection. No more than one dose from a vial should be administered.
If the Reblozyl solution has been refrigerated after reconstitution, it should be removed from the refrigerator 15–30 minutes prior to injection to allow it to reach room temperature. This will allow for a more comfortable injection.
For instructions on reconstitution of the medicinal product before administration, see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Contraindications
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Pregnancy (see section 4.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
