HIBRUKA TABLET 50MG [SIN16645P]
Active ingredients: HIBRUKA TABLET 50MG
Product Info
HIBRUKA TABLET 50MG
[SIN16645P]
Product information
Active Ingredient and Strength | ORELABRUTINIB - 50 MG |
Dosage Form | TABLET |
Manufacturer and Country | WUXI STA PHARMACEUTICAL CO., LTD. - CHINA |
Registration Number | SIN16645P |
Licence Holder | FORTREA SINGAPORE PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01EL04 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4 Indications
HIBRUKA is indicated for the treatment of adult patients with mantle cell lymphoma (MCL) who have received at least one prior therapy.
HIBRUKA is indicated for the treatment of adult patients with relapsed or refractory marginal zone lymphoma (MZL) who have received at least one prior anti-CD20-based therapy (see 15. Clinical Trials – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Dosing
6 Dosage & Administration
HIBRUKA should be administered under the direction of a physician specialized in the diagnosis and treatment of patients with the intended indications.
HIBRUKA tablets should be taken orally once daily at approximately the same time each day, either before or after meal. Swallow the whole tablet with water. Do not break, crush or chew the tablets.
The recommended dose of HIBRUKA is 150 mg (three 50 mg tablets) orally once daily until disease progression or unacceptable toxicity.
Missed Dose
If a dose is missed at the scheduled time, it should be taken as soon as possible only if it is at least 8 hours before the next dose and return to the normal schedule in the following day. Do not take extra tablets to make up for the missed dose.
Dosage Adjustments
Recommended dose modifications are provided in Table 1.
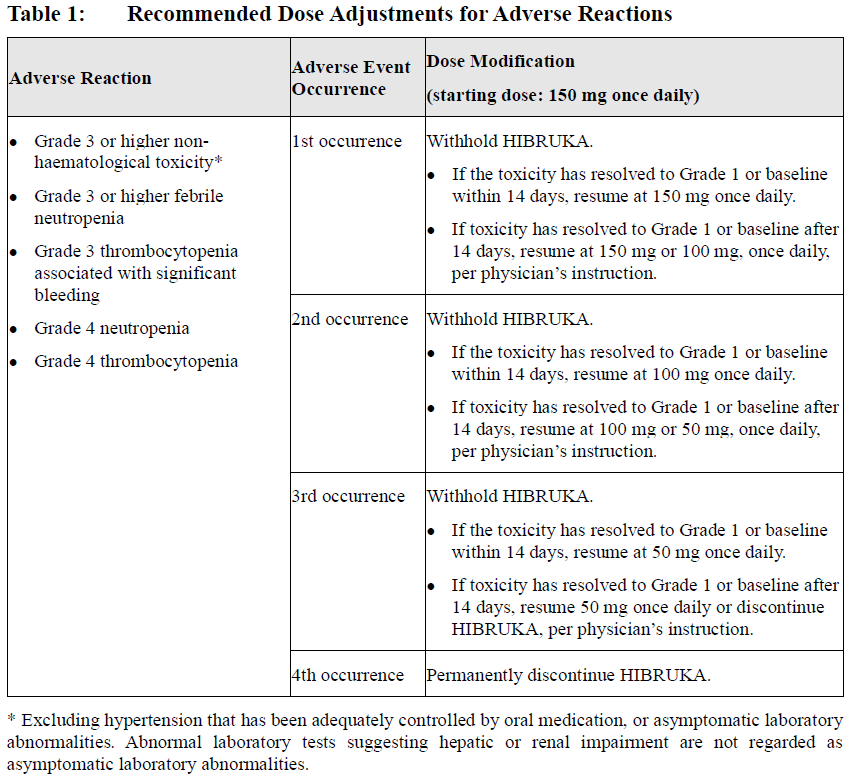
Asymptomatic lymphocytosis is usually not regarded as an adverse reaction. Patients who have experienced such event can continue HIBRUKA under the direction of the treating physician.
Co-administration with CYP3A4 Inhibitors or Inducers
Co-administration with strong or moderate CYP3A inducers should be avoided.
Co-administration with CYP3A inhibitors can increase the exposure of HIBRUKA. Co-administration with strong or moderate CYP3A inhibitors should be avoided (See 13. Drug Interactions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Use in Specific Populations
Hepatic Impairment
No dose modification is recommended in patients with mild hepatic impairment (total bilirubin ≤ ULN and AST > ULN or total bilirubin > 1 to 1.5 × ULN and any AST). Based on limited data, no dose recommendations can be made in patients with moderate hepatic impairment (total bilirubin > 1.5 to 3 × ULN and any AST). The use of HIBRUKA in patients with severe hepatic impairment (total bilirubin > 3 × ULN and any AST) should be avoided (See 17. Pharmacodynamics and Pharmacokinetics – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal Impairment
No dose modification is recommended in patients with mild and moderate renal impairment. Patients with severe renal impairment must use HIBRUKA with caution under the direction of a physician, and renal function should be closely monitored (See 9. Warnings and Precautions, 17. Pharmacodynamics and Pharmacokinetics – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Geriatric Use
No dose modification is required for elderly patients (See 12. Geriatric Use – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Paediatric Use
The safety and efficacy of HIBRUKA in paediatric patients have not been established.
Contraindications
8 Contraindications
HIBRUKA is contraindicated in patients with:
Severe hepatic impairment.
Hypersensitivity (manifested by symptoms such as anaphylactic or anaphylactoid reaction) to HIBRUKA or to any of the excipients (see 18. List of Excipients – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
