NOTRIXUM INJECTION 10MG/ML [SIN16945P]
Active ingredients: NOTRIXUM INJECTION 10MG/ML
Product Info
NOTRIXUM INJECTION 10MG/ML
[SIN16945P]
Product information
Active Ingredient and Strength | ATRACURIUM BESILATE - 10 MG/ML |
Dosage Form | INJECTION, SOLUTION |
Manufacturer and Country | PT. NOVELL PHARMACEUTICAL LABORATORIES - INDONESIA REP OF |
Registration Number | SIN16945P |
Licence Holder | GOLDPLUS UNIVERSAL PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | M03AC04 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Indications
NOTRIXUM is a highly selective, competitive or non-depolarising neuromuscular blocking agent which is used as an adjunct to general anaesthesia to enable tracheal intubation to be performed and to relax skeletal muscles during surgery or controlled ventilation during a wide range of medical procedures.
Dosing
Dosage and Administration
In common with all neuromuscular blocking agents monitoring of neuromuscular function is recommended during the use of NOTRIXUM in order to individualise dosage requirements.
Use by injection in adults
NOTRIXUM is administered by intravenous (i.v.) injection. The dosage range for adults is 0.3 to 0.6 mg/kg (depending on the duration of full block required) and will provide adequate relaxation for 15 to 35 minutes.
Endotracheal intubation can usually be accomplished within 90 seconds from the i.v. injection of 0.5 to 0.6 mg/kg.
Full block can be prolonged with supplementary doses of 0.1 to 0.2 mg/kg as required. Successive supplementary dosing does not give rise to accumulation of neuromuscular blocking effect.
Spontaneous recovery from the end of full block occurs in about 35 minutes as measured by the restoration of the tetanic response to 95% of normal neuromuscular function.
The neuromuscular block produced by NOTRIXUM can be rapidly reversed by standard doses of anticholinesterase agents, such as neostigmine and edrophonium, accompanied or preceded by atropine, with no evidence of recurarisation.Use as an infusion in adults
After an initial bolus dose of 0.3 to 0.6 mg/kg, NOTRIXUM can be used to maintain neuromuscular block during long surgical procedures by administration as a continuous infusion at rates of 0.3 to 0.6 mg/kg/h.
NOTRIXUM can be administered by infusion during cardiopulmonary bypass surgery at the recommended infusion rates. Induced hypothermia to a body temperature of 25°C to 26°C reduces the rate of inactivation of NOTRIXUM, therefore full neuromuscular block may be maintained by approximately half the original infusion rate at these low temperatures.
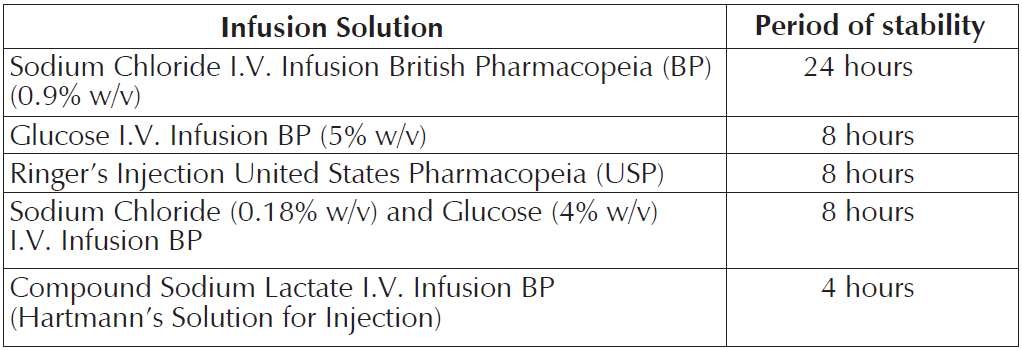
NOTRIXUM is compatible with the following infusion solutions for the times stated below:
Chemical and physical in-use stability has been demonstrated for the stated periods at temperature of up to 30°C. From a microbiological point of view, the product should be used immediately.
If not used immediately, in-use storage times and conditions prior to use are the responsibility of the user and would normally not be longer than 24 hours at 2 to 8°C, unless dilution has taken place in controlled and validated aseptic conditions.
Use in children
The dosage in children over the age of 1 month is the same as that in adults on a bodyweight basis.Use in the elderly
NOTRIXUM may be used at standard dosage in elderly patients.
It is recommended, however, that the initial dose be at the lower end of the range and that it be administered slowly.Use in patients with reduced renal and/or hepatic function
NOTRIXUM may be used at standard dosage at all levels of renal or hepatic function, including end-stage failure.Use in patients with cardiovascular disease
In patients with clinically significant cardiovascular disease, the initial dose of NOTRIXUM should be administered over a period of 60 seconds.
Contraindications
Contraindications
NOTRIXUM is contraindicated in patients known to be hypersensitive to atracurium, cisatracurium or benzenesulfonic acid.
