ILUMYA SOLUTION FOR INJECTION IN PRE-FILLED SYRINGE 100MG/ML [SIN17076P]
Active ingredients: ILUMYA SOLUTION FOR INJECTION IN PRE-FILLED SYRINGE 100MG/ML
Product Info
ILUMYA SOLUTION FOR INJECTION IN PRE-FILLED SYRINGE 100MG/ML
[SIN17076P]
Product information
Active Ingredient and Strength | TILDRAKIZUMAB - 100 MG/ML |
Dosage Form | INJECTION, SOLUTION |
Manufacturer and Country | VETTER PHARMA-FERTIGUNG GMBH & CO. KG - GERMANY |
Registration Number | SIN17076P |
Licence Holder | RANBAXY (MALAYSIA) SDN. BHD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L04AC17 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
INDICATION
Tildrakizumab is an interleukin-23 antagonist indicated for the treatment of adults with moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
Dosing
DOSAGE AND ADMINISTRATION
Tildrakizumab is administered by subcutaneous injection. The recommended dose is 100 mg at Weeks 0, 4, and every twelve weeks thereafter.
In the Phase III clinical studies, the results showed that patients had a PASI 75 response of 61% – 64% and PASI 90 response of 35% – 39% after 12 weeks of treatment with tildrakizumab 100 mg [see Clinical Studies – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information]. It is recommended that patients should be monitored for the clinical response to treatment. Consideration should be given to discontinuing treatment in patients who have shown no response after 16 weeks of treatment. Some patients with initial partial response may subsequently improve with continued treatment beyond 16 weeks.
Tuberculosis Assessment Prior to Initiation of Tildrakizumab
Evaluate patients for Tuberculosis (TB) infection prior to initiating treatment with tildrakizumab [see Warnings and Precautions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
Method of Administration:
Tildrakizumab is administered by subcutaneous injection. Injection sites should be alternated. Tildrakizumab should not be injected into areas where the skin is affected by plaque psoriasis or is tender, bruised, red, hard, thick, or scaly. The pre-filled syringe must not be shaken. Each pre-filled syringe is for single use only.
Inject the full amount of tildrakizumab according to the instructions for use provided in the Section of Preparation and Administration.
After proper training in subcutaneous injection technique, patients may self-inject tildrakizumab if a physician determines that it is appropriate. However, the physician should ensure appropriate follow-up of patients. Patients should be instructed to inject the full amount of tildrakizumab.
Preparation and Administration:
Before injection, remove tildrakizumab carton from the refrigerator, and let the prefilled syringe (in the carton with the lid closed) sit at room temperature for 30 minutes.
Follow the instructions on the tildrakizumab carton to remove the prefilled syringe correctly, and remove only when ready to inject. Do not pull off the needle cover until you are ready to inject.
Inspect tildrakizumab visually for particulate matter and discoloration prior to administration. tildrakizumab is a clear to slightly opalescent, colorless to slightly yellow solution. Do not use if the liquid contains visible particles or the syringe is damaged. Air bubbles may be present; there is no need to remove them.
Choose an injection site with clear skin and easy access (such as abdomen, thighs, or upper arm). Do not administer 2 inches around the navel or where the skin is tender, bruised, erythematous, indurated, or affected by psoriasis. Also, do not inject into scars, stretch marks, or blood vessels.
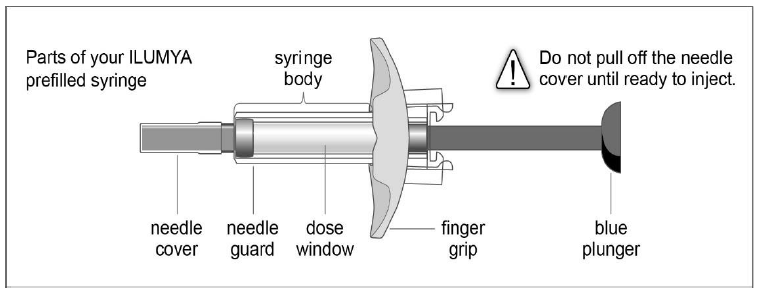
While holding the body of the syringe, pull the needle cover straight off (do not twist) and discard.
Inject tildrakizumab subcutaneously as recommended [see Dosage and Administration].
Press down the blue plunger until it can go no further. This activates the safety mechanism that will ensure full retraction of the needle after the injection is given.
Remove the needle from the skin entirely before letting go of the blue plunger. After the blue plunger is released, the safety lock will draw the needle inside the needle guard.
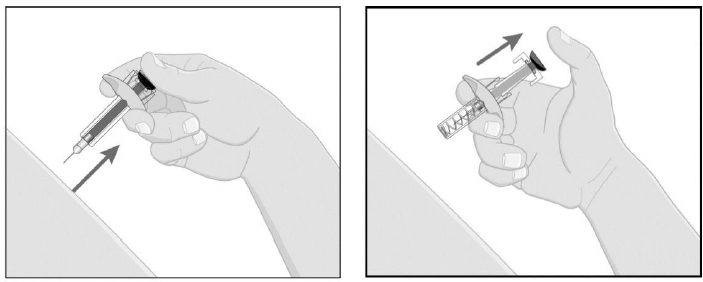
Discard any unused portion. Dispose of used syringe.
Contraindications
CONTRADICTIONS
Tildrakizumab is contraindicated in patients with a previous serious hypersensitivity reaction to tildrakizumab or to any of the excipients [see Warnings and Precautions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
