FOSTER NEXTHALER INHALATION POWDER 200 MCG/6 MCG PER DOSE [SIN17099P]
Active ingredients: FOSTER NEXTHALER INHALATION POWDER 200 MCG/6 MCG PER DOSE
Product Info
FOSTER NEXTHALER INHALATION POWDER 200 MCG/6 MCG PER DOSE
[SIN17099P]
Product information
Active Ingredient and Strength | BECLOMETASONE DIPROPIONATE - 0.2 MG/DOSE |
Dosage Form | AEROSOL, POWDER |
Manufacturer and Country | CHIESI FARMACEUTICI SPA - ITALY |
Registration Number | SIN17099P |
Licence Holder | ORIENT EUROPHARMA PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | R03AK08 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Foster Nexthaler is indicated in the regular treatment of asthma where use of a combination product (inhaled corticosteroid and long-acting beta2-agonist) is appropriate:
patients not adequately controlled with inhaled corticosteroids and 'as needed' inhaled short-acting beta2-agonist or
patients already adequately controlled on both inhaled corticosteroids and long-acting beta2-agonists.
Foster Nexthaler is indicated in adults.
Note: there are no relevant clinical data on the use of Foster Nexthaler for the treatment of acute asthma attacks.
Dosing
4.2 Posology and method of administration
Posology
Foster Nexthaler is not intended for the initial management of asthma. The dosage of Foster Nexthaler is individual and should be adjusted to the severity of the disease. This should be considered not only when treatment with combination products is initiated but also when the dose is adjusted. If an individual patient should require a combination of doses other than those available in the combination inhaler, appropriate doses of beta2-agonists and/or corticosteroids by individual inhalers should be prescribed.
Because of its extrafine particle size distribution, dose adjustment is required when patients are transferred to Foster Nexthaler inhalation powder from a formulation with a non-extrafine particle size distribution. When switching patients from previous treatments, it should be considered that the recommended total daily dose of beclometasone dipropionate for Foster Nexthaler is lower than that for current beclometasone dipropionate-containing non-extrafine products and should be adjusted to the needs of the individual patient.
Dose recommendations for adults 18 years and above
Two inhalations twice daily.
The maximum daily dose is 4 inhalations daily.
Patients should be regularly reassessed by a doctor, so that the dosage of Foster Nexthaler remains optimal and is only changed on medical advice. The dose should be titrated to the lowest dose at which effective control of symptoms is maintained. When control of symptoms is maintained with the lowest recommended dosage, then the next step-down could include the inhaled corticosteroid alone.
A lower strength of the beclometasone dipropionate component in the same Nexthaler device is available for step-down treatment (Foster Nexthaler 100/6 micrograms).
Patients should be advised to take Foster Nexthaler every day even when asymptomatic.
Special populations
There is no need to adjust the dose in elderly patients.
There are no data available for use of Foster Nexthaler in patients with hepatic or renal impairment (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Paediatric population
Foster Nexthaler 200/6 micrograms should not be used in children and adolescents below 18 years.
Method of administration
Foster Nexthaler is for inhalation use.
Nexthaler is a breath-operated inhaler. Moderate and severe asthmatic patients were shown to be able to produce sufficient inspiratory flow to trigger inhalation release from Nexthaler (see section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
The delivery of Foster Nexthaler with Nexthaler is flow-independent in the range of inspiratory flow that this patient population can achieve through the inhaler.
Correct use of the Nexthaler inhaler is essential in order for the treatment to be successful. The patient should be advised to read the Package Insert carefully and follow the instructions for use as given in the leaflet. For instructions for use, see below.
The number of inhalations shown in the window on the shell does not decrease on closing the cover if the patient has not inhaled through the inhaler.
The patient should be instructed to only open the inhaler’s cover when needed. In the event that the patient has opened the inhaler but not inhaled, and the cover is closed, the metered dose is moved back to the powder reservoir within the inhaler; the following metered dose can be safely inhaled.
Patients should rinse their mouth or gargle with water or brush their teeth after inhaling (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
INSTRUCTIONS FOR USE OF NEXTHALER INHALER
A. Contents of the Package
For information on the contents of the pack, see section 6.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
If the package contents are not the same as described in section 6.5, return your inhaler to the person who supplied it and get a new one.
B. General Warnings & Precautions
Do not remove the inhaler from the sachet if you do not intend to use it immediately.
Only use your inhaler as indicated.
Keep the cover closed until you need to take an inhalation from your inhaler.
When you are not using your inhaler keep it in a clean and dry place.
Do not attempt to take your Nexthaler inhaler apart for any reason.
C. Key features of your Nexthaler inhaler
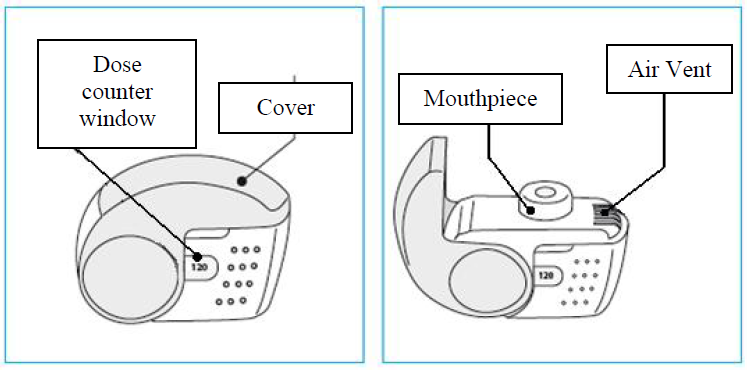
Taking a dose from your Nexthaler inhaler requires just three simple steps: Open, Inhale, Close.
D. Before using a new Nexthaler inhaler
1. Open the sachet and take out your inhaler.
Do not use your inhaler if the sachet is not sealed or it is damaged – return it to the person who supplied it and get a new one.
Use the label on the box to write down the date you open the sachet.
2. Inspect your inhaler.
If your inhaler looks broken or damaged, return it to the person who supplied it and get a new one.
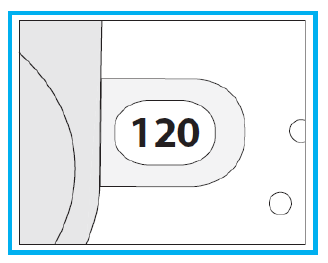
3. Check the Dose Counter Window. If your inhaler is brand new you will see “120” in the Dose Counter Window.
Do not use a new inhaler if the number shown is less than “120” – return it to the person who supplied it and get a new one.
E. How to use your Nexthaler inhaler
If you are not sure you are receiving your dose correctly contact your pharmacist or doctor.
If you are not sure the dose counter has gone down by one after inhalation, wait until your next scheduled dose and take this as normal. Do not take an extra dose.
E.1. Open
1. Hold your inhaler firmly in the upright position.
2. Check the number of inhalations left: any number between “1” and “120” shows that there are inhalations left.
If the Dose Counter Window shows “0” there are no inhalations left – dispose of your inhaler and get a new one.
3. Open the cover fully.
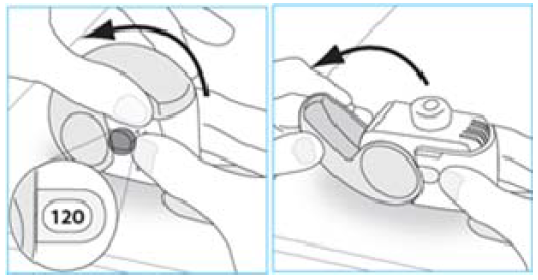
4. Before inhaling breathe out as far as is comfortable.
Do not breathe out through your inhaler.
E.2. Inhale
Whenever possible, stand or sit in an upright position when inhaling.
1. Lift your inhaler up, bring it to your mouth and place your lips around the mouthpiece.
Do not cover the air vent when holding your inhaler.
Do not inhale through the air vent.
2. Take a forceful and deep breath through your mouth.
You may notice a taste when you take your inhalation.
You may hear or feel a click when you take your inhalation.
Do not inhale through your nose.
Do not remove your inhaler from your lips during the inhalation.
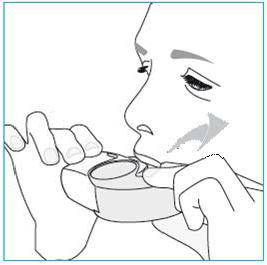
3. Remove your inhaler from your mouth.
4. Hold your breath for 5 to 10 seconds or as long as is comfortable.
5. Breathe out slowly.
Do not breathe out through your inhaler.
E.3. Close
1. Move your inhaler back to the upright position and close the cover fully.
2. Check that the dose counter has gone down by one.
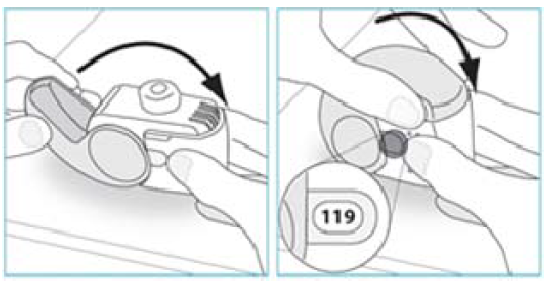
3. If you need to take another dose, repeat steps E.1 to E.3.
F. Cleaning
Normally, it is not necessary to clean your inhaler.
If necessary you may clean your inhaler after use with a dry cloth or tissue.
Do not clean your inhaler with water or other liquids. Keep it dry.
G. Storage and Disposal
For information on storage conditions and disposal instructions, see sections 6.4 and 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Contraindications
4.3 Contraindications
Hypersensitivity to the active substances or to any of the excipients listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
