SOGROYA SOLUTION FOR INJECTION IN PRE-FILLED PEN 15MG/1.5ML [SIN17218P]
Active ingredients: SOGROYA SOLUTION FOR INJECTION IN PRE-FILLED PEN 15MG/1.5ML
Product Info
SOGROYA SOLUTION FOR INJECTION IN PRE-FILLED PEN 15MG/1.5ML
[SIN17218P]
Product information
Active Ingredient and Strength | SOMAPACITAN - 15 MG/1.5 ML |
Dosage Form | INJECTION, SOLUTION |
Manufacturer and Country | NOVO NORDISK A/S (FORMULATION, FILLING AND PRIMARY PACKAGER) - DENMARK |
Registration Number | SIN17218P |
Licence Holder | NOVO NORDISK PHARMA (SINGAPORE) PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | H01AC07 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Sogroya® is indicated for the replacement of endogenous growth hormone (GH) in children aged 3 years and above, and adolescents with growth failure due to growth hormone deficiency (paediatric GHD), and in adults with growth hormone deficiency (adult GHD).
Dosing
4.2 Posology and method of administration
Somapacitan should be initiated and monitored by physicians who are appropriately qualified and experienced in the diagnosis and management of patients with growth hormone deficiency (e.g. endocrinologists).
Posology
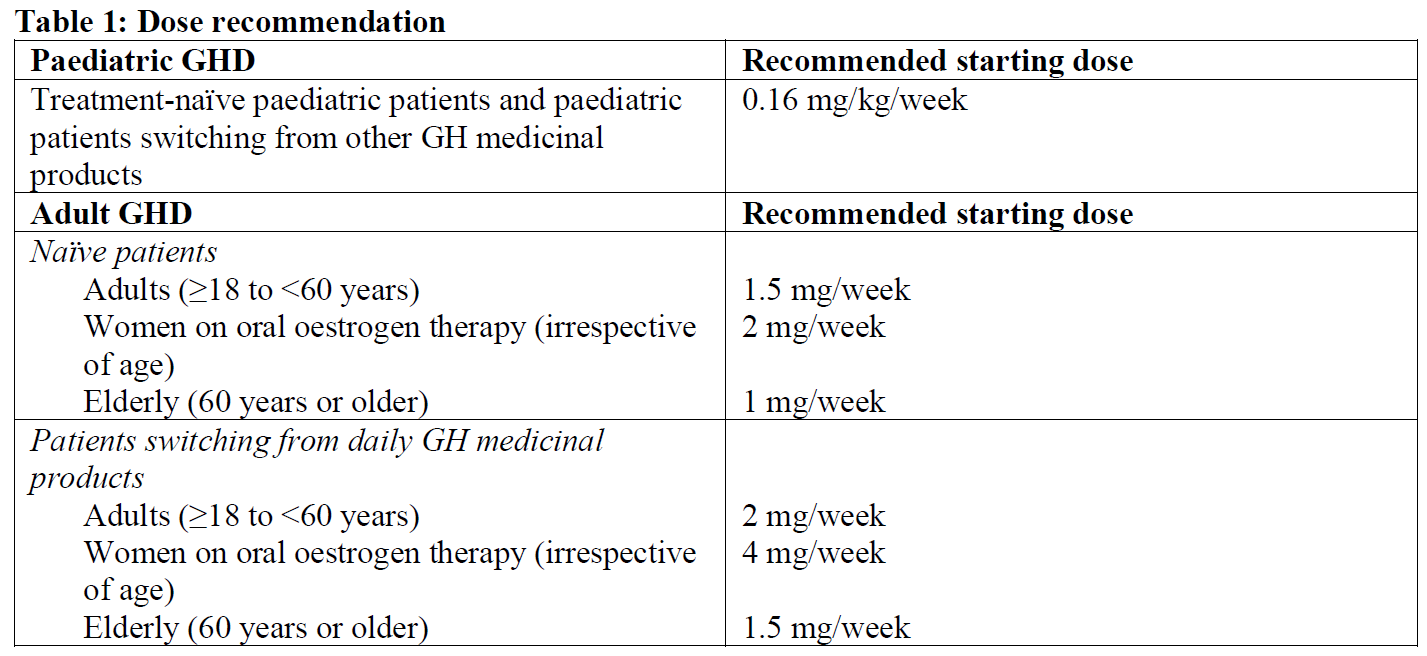
Paediatric GHD
Dose titration
Somapacitan dose may be individualised and adjusted based on growth velocity, adverse reactions, body weight and serum insulin-like growth factor I (IGF-I) concentrations.
Average IGF-I standard deviation score (SDS) levels (drawn 4 days after dosing) can guide dose titration. Dose adjustments should be targeted to achieve average IGF-I SDS levels in the normal range, i.e. between −2 and +2 (preferably close to 0 SDS).
If the IGF-I (SDS) is > 2, it should be reassessed after a subsequent somapacitan administration. If the value remains > 2, reducing the dose by 0.04 mg/kg/week is recommended. More than one dose reduction may be required in some patients.
In patients who have had the dose reduced but are not growing well, the dose may be gradually increased as tolerated up to a maximum dose of 0.16 mg/kg/week. Dose increments should not exceed 0.02 mg/kg per week. In paediatric GHD doses above 0.16mg/kg/week have not been studied and are not recommended.
Treatment evaluation
Evaluation of efficacy and safety should be considered at approximately 6- to 12-month intervals and may be assessed by evaluating auxological parameters, biochemistry (IGF-I, hormones, glucose, and lipid levels) and pubertal status. More frequent evaluations should be considered during puberty.
Treatment should be discontinued in patients having achieved final height or near final height, i.e. an annualised height velocity < 2 cm/year and a bone age > 14 years in girls or > 16 years in boys which corresponds to the closure of the epiphyseal growth plates, see section 4.3. Once the epiphyses are fused, patients should be clinically re-evaluated for the need for growth hormone treatment.
When GHD persists after growth completion, growth hormone treatment should be continued to achieve full somatic adult development including lean body mass and bone mineral accrual (for guidance on dosing see recommended dose for adults (Table 1)).
Adult GHD
Dose titration
The somapacitan dose must be individually adjusted for each patient. It is recommended to increase the dose gradually with 2–4 weeks intervals in steps from 0.5 mg to 1.5 mg based on the patients’ clinical response and experience of adverse reactions up to a dose of 8 mg somapacitan per week. Serum insulin like growth factor-I (IGF-I) levels (drawn 3–4 days after dosing) can be used as guidance for the dose titration. The IGF-I standard deviation score (SDS) target should aim for the upper normal range not exceeding 2 SDS. IGF-I SDS levels in the target range are usually achieved within 8 weeks of dose titration. Longer dose titration may be necessary in some adult GHD patients (see below and section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Treatment evaluation
Using IGF-I SDS as a biomarker for dose titration, the aim is to reach IGF-I SDS levels within the age-adjusted upper reference range (IGF-I SDS upper reference range: 0 and +2) within 12 months of titration. If this target range cannot be achieved within this period, or the patient does not obtain the desired clinical response, other treatment options should be considered.
During somapacitan maintenance treatment, evaluation of efficacy and safety should be considered at approximately 6- to 12-month intervals and may be assessed by evaluating biochemistry (IGF-I-, glucose-, and lipid levels), body composition, and body mass index.
Paediatric and adult GHD
Switching from other growth hormone products
Patients switching from a weekly growth hormone to somapacitan are recommended to continue administration at their once weekly dosing day.
Patients switching from daily human growth hormone to once-weekly somapacitan should choose the preferred day for the weekly dose and inject the final dose of daily treatment the day before (or at least 8 hours before) injecting the first dose of once-weekly somapacitan. Patients should follow the instructions for the dose presented in Table 1.
Oral oestrogen therapy
Females on oral oestrogen-containing therapy may have reduced IGF-I levels and may require dose adjustment of growth hormone to achieve the treatment goal (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Missed dose
Patients who miss a dose are advised to inject once-weekly somapacitan upon discovery as soon as possible, within 3 days after the missed dose, and then resume their usual once-weekly dosing schedule. If more than 3 days have passed, the dose should be skipped and the next dose should be administered on the regularly scheduled day. If two or more doses have been missed, the dose should be resumed on the regularly scheduled day.
Changing the dosing day
The day of weekly injection can be changed as long as the time between two doses is at least 4 days. After selecting a new dosing day, the once weekly dosing should be continued.
Flexibility in dosing time
On occasions when injection at the scheduled dosing day is not possible, once-weekly somapacitan can be administered up to 2 days before or 3 days after the scheduled weekly dosing day as long as the time between two doses is at least 4 days (96 hours). Once-weekly dosing for the next dose could be resumed at the regularly scheduled dosing day.
Special populations
Elderly (60 years or older)
Generally, lower doses of somapacitan may be necessary in older patients. For further information, see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Paediatric population
Limited data on the clinical effects of somapacitan are available in paediatric GHD patients under 3 years of age. Currently available data are described in sections 5.1 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information, but no recommendation on a posology can be made.
Gender
Men show an increasing IGF-I sensitivity over time. This means that there is a risk that men are overtreated. Women, especially those on oral oestrogen, may require higher doses and a longer titration period than men, see sections 5.1 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information. In females using oral oestrogen, it should be considered to change the route of oestrogen administration (e.g. transdermal, vaginal) see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Renal impairment
No adjustment of the starting dose is required for adult patients with renal impairment. Patients with renal impairment may need lower doses of somapacitan, but since the dose of somapacitan is individually adjusted according to the need of each patient, no further dose adjustment is required, see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information. Somapacitan has not been studied in paediatric patients with renal impairment.
Hepatic impairment
No adjustment of the starting dose is required for adult patients with hepatic impairment. Patients with moderate hepatic impairment may need higher doses of somapacitan, but since the dose of somapacitan is individually adjusted according to the need of each patient, no further dose adjustment is required. No information regarding the use of somapacitan in patients with severe hepatic impairment is available. Caution should be exercised if treating these patients with somapacitan, see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information. Somapacitan has not been studied in paediatric patients with hepatic impairment.
Method of administration
Somapacitan is to be administered once-weekly at any time of the day.
Somapacitan is to be injected subcutaneously in the abdomen, thighs, buttocks or upper arms without dose adjustment.
The injection site should be rotated every week to prevent local lipoatrophy.
The Sogroya® 15 mg/1.5 mL (10 mg/mL) pen delivers doses from 0.10 mg (0.01 mL) to 8 mg (0.8 mL) in increments of 0.10 mg.
For instructions of the medicinal product before administration, see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Contraindications
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Somapacitan must not be used when there is any evidence of activity of a tumour. Intracranial tumours must be inactive and antitumour therapy must be completed prior to starting somapacitan therapy. Treatment should be discontinued if there is evidence of tumour growth, see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Somapacitan must not be used for longitudinal growth promotion in children with closed epiphyses, see section 4.2.
Patients with acute critical illness suffering from complications following open heart surgery, abdominal surgery, multiple accidental trauma, acute respiratory failure or similar conditions must not be treated with somapacitan (regarding patients undergoing substitution therapy, see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
