ESTAELTO FILM COATED TABLETS 15MG [SIN17248P]
Active ingredients: ESTAELTO FILM COATED TABLETS 15MG
Product Info
ESTAELTO FILM COATED TABLETS 15MG
[SIN17248P]
Product information
Active Ingredient and Strength | RIVAROXABAN - 15 MG |
Dosage Form | TABLET, FILM COATED |
Manufacturer and Country | ZENTIVA PRIVATE LIMITED - INDIA |
Registration Number | SIN17248P |
Licence Holder | WELLESTA HEALTHCARE (SINGAPORE) PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | B01AF01 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Adults
Prevention of stroke and systemic embolism in adult patients with non-valvular atrial fibrillation. There are limited data on the relative effectiveness of rivaroxaban and warfarin in reducing the risk of stroke and systemic embolism when warfarin therapy is well-controlled (see section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE), and prevention of recurrent DVT and PE in adults. (See section 4.4 for haemodynamically unstable PE patients – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.)
Paediatric population
Estaelto 15 mg
Treatment of venous thromboembolism (VTE) and prevention of VTE recurrence in children and adolescents aged less than 18 years and weighing from 30 kg to 50 kg after at least 5 days of initial parenteral anticoagulation treatment.
Estaelto 20 mg
Treatment of venous thromboembolism (VTE) and prevention of VTE recurrence in children and adolescents aged less than 18 years and weighing more than 50 kg after at least 5 days of initial parenteral anticoagulation treatment.
Dosing
4.2 Posology and method of administration
Posology
Prevention of stroke and systemic embolism in adults
The recommended dose is 20 mg once daily, which is also the recommended maximum dose.
Therapy with Estaelto should be continued long term provided the benefit of prevention of stroke and systemic embolism outweighs the risk of bleeding (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
If a dose is missed the patient should take Estaelto immediately and continue on the following day with the once daily intake as recommended. The dose should not be doubled within the same day to make up for a missed dose.
Treatment of DVT, treatment of PE and prevention of recurrent DVT and PE in adults
The recommended dose for the initial treatment of acute DVT or PE is 15 mg twice daily for the first three weeks followed by 20 mg once daily for the continued treatment and prevention of recurrent DVT and PE.
Short duration of therapy (at least 3 months) should be considered in patients with DVT or PE provoked by major transient risk factors (i.e. recent major surgery or trauma). Longer duration of therapy should be considered in patients with provoked DVT or PE not related to major transient risk factors, unprovoked DVT or PE, or a history of recurrent DVT or PE.
When extended prevention of recurrent DVT and PE is indicated (following completion of at least 6 months therapy for DVT or PE), the recommended dose is 10 mg once daily. In patients in whom the risk of recurrent DVT or PE is considered high, such as those with complicated comorbidities, or who have developed recurrent DVT or PE on extended prevention with Estaelto 10 mg once daily, a dose of Estaelto 20 mg once daily should be considered.
The duration of therapy and dose selection should be individualised after careful assessment of the treatment benefit against the risk for bleeding (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
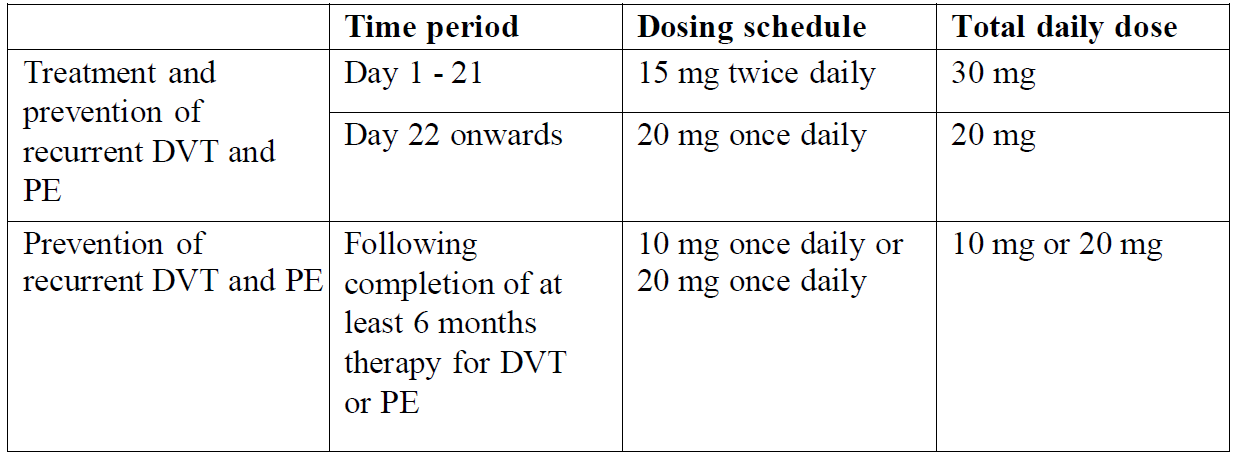
If a dose is missed during the 15 mg twice daily treatment phase (day 1 – 21), the patient should take Estaelto immediately to ensure intake of 30 mg Estaelto per day. In this case two 15 mg tablets may be taken at once. The patient should continue with the regular 15 mg twice daily intake as recommended on the following day.
If a dose is missed during the once daily treatment phase, the patient should take Estaelto immediately, and continue on the following day with the once daily intake as recommended. The dose should not be doubled within the same day to make up for a missed dose.
Treatment of VTE and prevention of VTE recurrence in children and adolescents
Estaelto treatment in children and adolescents aged less than 18 years should be initiated following at least 5 days of initial parenteral anticoagulation treatment (see section 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
The dose for children and adolescent is calculated based on body weight.
Body weight from 30 to 50 kg:
a once daily dose of 15 mg rivaroxaban is recommended. This is the maximum daily dose.Body weight of 50 kg or more:
a once daily dose of 20 mg rivaroxaban is recommended. This is the maximum daily dose.For patients with body weight less than 30 kg refer to the PI of rivaroxaban containing products in the form of granules for oral suspension.
The weight of a child should be monitored and the dose reviewed regularly. This is to ensure a therapeutic dose is maintained. Dose adjustments should be made based on changes in body weight only.
Treatment should be continued for at least 3 months in children and adolescents. Treatment can be extended up to 12 months when clinically necessary. There is no data available in children to support a dose reduction after 6 months treatment. The benefit-risk of continued therapy after 3 months should be assessed on an individual basis taking into account the risk for recurrent thrombosis versus the potential bleeding risk.
If a dose is missed, the missed dose should be taken as soon as possible after it is noticed, but only on the same day. If this is not possible, the patient should skip the dose and continue with the next dose as prescribed. The patient should not take two doses to make up for a missed dose.
Converting from Vitamin K Antagonists (VKA) to Estaelto
Prevention of stroke and systemic embolism:
VKA treatment should be stopped and Estaelto therapy should be initiated when the International Normalised Ratio (INR) is ≤ 3.0.Treatment of DVT, PE and prevention of recurrence in adults and treatment of VTE and prevention of recurrence in paediatric patients:
VKA treatment should be stopped and Estaelto therapy should be initiated once the INR is ≤ 2.5.
When converting patients from VKAs to Estaelto, INR values will be falsely elevated after the intake of Estaelto. The INR is not valid to measure the anticoagulant activity of Estaelto, and therefore should not be used (see section 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Converting from Estaelto to Vitamin K antagonists (VKA)
There is a potential for inadequate anticoagulation during the transition from Estaelto to VKA. Continuous adequate anticoagulation should be ensured during any transition to an alternate anticoagulant. It should be noted that Estaelto can contribute to an elevated INR.
No clinical trial data are available to guide converting patients from Estaelto to warfarin. Estaelto affects INR, so INR measurements made during co-administration with warfarin may not be useful for determining the appropriate dose of warfarin.
One approach is to discontinue Estaelto and begin both a parenteral anticoagulant and warfarin at the time the next dose of Estaelto would have been taken.
In another approach, in patients converting from Estaelto to VKA, VKA should be given concurrently until the INR is ≥ 2.0. For the first two days of the conversion period, standard initial dosing of VKA should be used followed by VKA dosing, as guided by INR testing. While patients are on both Estaelto and VKA the INR should not be tested earlier than 24 hours after the previous dose but prior to the next dose of Estaelto. Once Estaelto is discontinued INR testing may be done reliably at least 24 hours after the last dose (see sections 4.5 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). Patients are to be carefully monitored for signs of bleeding complications during the conversion period.
Paediatric patients:
Children who convert from Estaelto to VKA need to continue Estaelto for 48 hours after the first dose of VKA. After 2 days of co-administration an INR should be obtained prior to the next scheduled dose of Estaelto. Co-administration of Estaelto and VKA is advised to continue until the INR is ≥ 2.0. Once Estaelto is discontinued INR testing may be done reliably 24 hours after the last dose (see above and section 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Converting from parenteral anticoagulants to Estaelto
For adult and paediatric patients currently receiving a parenteral anticoagulant, discontinue the parenteral anticoagulant and start Estaelto 0 to 2 hours before the time that the next scheduled administration of the parenteral medicinal product (e.g. low molecular weight heparins) would be due or at the time of discontinuation of a continuously administered parenteral medicinal product (e.g. intravenous unfractionated heparin).
Converting from Estaelto to parenteral anticoagulants
Discontinue Estaelto and give the first dose of parenteral anticoagulant at the time the next Estaelto dose would be taken.
Special populations
Renal impairment
Adults:
Limited clinical data for patients with severe renal impairment (creatinine clearance 15 – 29 ml/min) indicate that rivaroxaban plasma concentrations are significantly increased. Therefore, Estaelto is to be used with caution in these patients. Use is not recommended in patients with creatinine clearance < 15 ml/min (see sections 4.4 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
In patients with moderate (creatinine clearance 30 – 49 ml/min) or severe (creatinine clearance 15 – 29 ml/min) renal impairment the following dose recommendations apply:
For the prevention of stroke and systemic embolism in patients with non-valvular atrial fibrillation, the recommended dose is 15 mg once daily (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
For the treatment of DVT, treatment of PE and prevention of recurrent DVT and PE: patients should be treated with 15 mg twice daily for the first 3 weeks.
Thereafter, when the recommended dose is 20 mg once daily, a reduction of the dose from 20 mg once daily to 15 mg once daily should be considered if the patient’s assessed risk for bleeding outweighs the risk for recurrent DVT and PE. The recommendation for the use of 15 mg is based on PK modelling and has not been studied in this clinical setting (see sections 4.4, 5.1 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
When the recommended dose is 10 mg once daily, no dose adjustment from the recommended dose is necessary.
No dose adjustment is necessary in patients with mild renal impairment (creatinine clearance 50 – 80 ml/min) (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Paediatric population:
Children and adolescents with mild renal impairment (glomerular filtration rate 50 – 80 ml/min/1.73 m2): no dose adjustment is required, based on data in adults and limited data in paediatric patients (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Children and adolescents with moderate or severe renal impairment (glomerular filtration rate < 50 ml/min/1.73 m2): Rivaroxaban is not recommended as no clinical data is available (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Hepatic impairment
Estaelto is contraindicated in patients with hepatic disease associated with coagulopathy and clinically relevant bleeding risk including cirrhotic patients with Child Pugh B and C (see sections 4.3 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). No clinical data is available in children with hepatic impairment.
Elderly population
No dose adjustment (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Body weight
No dose adjustment for adults (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
For paediatric patients the dose is determined based on body weight.
Gender
No dose adjustment (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Patients undergoing cardioversion
Estaelto can be initiated or continued in patients who may require cardioversion. For transesophageal echocardiogram (TEE) guided cardioversion in patients not previously treated with anticoagulants, Estaelto treatment should be started at least 4 hours before cardioversion to ensure adequate anticoagulation (see sections 5.1 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). For all patients, confirmation should be sought prior to cardioversion that the patient has taken Estaelto as prescribed. Decisions on initiation and duration of treatment should take established guideline recommendations for anticoagulant treatment in patients undergoing cardioversion into account.
Patients with non-valvular atrial fibrillation who undergo PCI (percutaneous coronary intervention) with stent placement
There is limited experience of a reduced dose of 15 mg Estaelto once daily (or 10 mg Estaelto once daily for patients with moderate renal impairment [creatinine clearance 30 – 49 ml/min]) in addition to a P2Y12 inhibitor for a maximum of 12 months in patients with non-valvular atrial fibrillation who require oral anticoagulation and undergo PCI with stent placement (see sections 4.4 and 5.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Paediatric population
The safety and efficacy of rivaroxaban in children aged 0 to < 18 years have not been established in the indication prevention of stroke and systemic embolism in patients with non-valvular atrial fibrillation. No data are available. Therefore, it is not recommended for use in children below 18 years of age in indications other than the treatment of VTE and prevention of VTE recurrence.
Method of administration
Adults
Estaelto is for oral use.
The tablets are to be taken with food (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Crushing of tablets
For patients who are unable to swallow whole tablets, Estaelto tablet may be crushed and mixed with water or apple puree immediately prior to use and administered orally. After the administration of crushed Estaelto 15mg and 20 mg film-coated tablets, the dose should be immediately followed by food.
The crushed tablet may also be given through gastric tubes (see sections 5.2 and 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Children and adolescents weighing 30 kg to 50 kg
Estaelto is for oral use.
The patient should be advised to swallow the tablet with liquid. It should also be taken with food (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). The tablets should be taken approximately 24 hours apart.
In case the patient immediately spits up the dose or vomits within 30 minutes after receiving the dose, a new dose should be given. However, if the patient vomits more than 30 minutes after the dose, the dose should not be re-administered and the next dose should be taken as scheduled.
The tablet must not be split in an attempt to provide a fraction of a tablet dose.
Crushing of tablets
For patients who are unable to swallow whole tablets, rivaroxaban containing products in the form of granules for oral suspension should be used.
If the oral suspension is not immediately available, when doses of 15mg or 20 mg rivaroxaban are prescribed, these could be provided by crushing the 15mg or 20 mg tablet and mixing it with water or apple puree immediately prior to use and administering orally.
The crushed tablet may be given through a nasogastric or gastric feeding tube (see sections 5.2 and 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Contraindications
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Active clinically significant bleeding.
Lesion or condition, if considered to be a significant risk for major bleeding. This may include current or recent gastrointestinal ulceration, presence of malignant neoplasms at high risk of bleeding, recent brain or spinal injury, recent brain, spinal or ophthalmic surgery, recent intracranial haemorrhage, known or suspected oesophageal varices, arteriovenous malformations, vascular aneurysms or major intraspinal or intracerebral vascular abnormalities.
Concomitant treatment with any other anticoagulants, e.g. unfractionated heparin (UFH), low molecular weight heparins (enoxaparin, dalteparin, etc.), heparin derivatives (fondaparinux, etc.), oral anticoagulants (warfarin, dabigatran etexilate, apixaban, etc.) except under specific circumstances of switching anticoagulant therapy (see section 4.2) or when UFH is given at doses necessary to maintain an open central venous or arterial catheter (see section 4.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Hepatic disease associated with coagulopathy and clinically relevant bleeding risk including cirrhotic patients with Child Pugh B and C (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Pregnancy and breast-feeding (see section 4.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
