HYQVIA SOLUTION FOR INFUSION 100MG/ML [SIN17253P]
Active ingredients: HYQVIA SOLUTION FOR INFUSION 100MG/ML
Product Info
HYQVIA SOLUTION FOR INFUSION 100MG/ML
[SIN17253P]
Product information
Active Ingredient and Strength | (IG 10%) HUMAN NORMAL IMMUNOGLOBULIN - 100 MG/ML |
Dosage Form | INFUSION, SOLUTION |
Manufacturer and Country | BAXALTA BELGIUM MANUFACTURING SA (IGI 10% VIAL) - BELGIUM |
Registration Number | SIN17253P |
Licence Holder | TAKEDA PHARMACEUTICALS (ASIA PACIFIC) PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | J06BA01 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Replacement therapy in adults, children and adolescents (0 to 18 years) in:
Primary immunodeficiency syndromes (PID) with impaired antibody production (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Secondary immunodeficiencies (SID) in patients who suffer from severe or recurrent infections, ineffective antimicrobial treatment and either proven specific antibody failure (PSAF)* or serum IgG level of < 4 g/L.
*PSAF = failure to mount at least a 2-fold rise in IgG antibody titre to pneumococcal polysaccharide and polypeptide antigen vaccines.
Immunomodulatory therapy in adults, children and adolescents (0 to 18 years) in:
Chronic inflammatory demyelinating polyneuropathy (CIDP) as maintenance therapy after stabilization with IVIg.
Dosing
4.2 Posology and Method of Administration
Therapy should be initiated and monitored under the supervision of a physician experienced in the treatment of immunodeficiency/CIDP.
The medicinal product should be administered via the subcutaneous (SC) route. The dose and dose regimens are dependent on the indication.
The dose may need to be individualized for each patient dependent on the pharmacokinetic (PK) and clinical response. Dose based on bodyweight may require adjustment in underweight or overweight patients. The following dose regimens are given as a guideline.
Posology
Replacement therapy in PID
Patients naïve to immunoglobulin therapy
The dose required to achieve a trough level of 6 g/L is of the order of 0.4 to 0.8 g/kg body weight per month. The dose interval to maintain steady-state levels varies from 2-to-4 weeks.
IgG trough levels should be measured and assessed in conjunction with the incidence of infection. To reduce the rate of infection, it may be necessary to increase the dose and aim for higher IgG trough levels (> 6 g/L).
At the initiation of therapy, it is recommended that the treatment intervals for the first infusions be gradually prolonged from a 1-week dose to up to a 3- or 4-week dose. The cumulative monthly dose of IG 10% should be divided into 1-week, 2-week etc. doses according to the planned treatment intervals with HyQvia.
Patients previously treated with Intravenous immunoglobulin (IVIg)
For patients switching directly from IVIg, or who have a previous IVIg dose that can be referenced, the medicinal product should be administered at the same dose and at the same frequency as their previous IVIg treatment. If patients were previously on a 3-week dosing regimen, increasing the interval to 4-weeks can be accomplished by administering the same weekly equivalents.
Patients previously treated with subcutaneous immunoglobulin (SCIg)
The initial dose of the medicinal product is the same as for SCIg treatment but may be adjusted to 3- or 4-weeks interval. The first infusion should be given one week after the last treatment with the previous immunoglobulin.
Replacement therapy in SID
The recommended dose is 0.2 to 0.4 g/kg every 3 -to -4 weeks.
IgG trough levels should be measured and assessed in conjunction with the incidence of infection. Dose should be adjusted as necessary to achieve optimal protection against infections, an increase may be necessary in patients with persisting infection; a dose decrease can be considered when the patient remains infection free.
Immunomodulatory therapy in CIDP
Before initiating therapy, the weekly equivalent dose should be calculated by dividing the planned dose by the planned dose interval in weeks. The typical dosing interval range for HyQvia is 3 -to 4 - weeks. The recommended subcutaneous dose is 0.3 to 2.4 g/kg body weight per month, administered in 1-or 2- sessions over 1-or 2-days.
The patient’s clinical response should be the primary consideration in dose adjustment. The dose may need to be adapted to achieve the desired clinical response. In clinical deterioration, the dose may be increased to the recommended maximum of 2.4 g/kg monthly. If the patient is clinically stable, periodic dose reductions may be needed to observe whether the patient still needs IG therapy.
A titration schedule that permits gradual dose increase over time (ramp-up) is recommended to ensure the patient’s tolerability until the full dose is reached. During the titration schedule, the calculated HyQvia dose and recommended dose intervals must be followed for the first and second infusions. Depending on the treating physician's discretion, in patients who tolerate the first 2 infusions well, subsequent infusions may be administered by gradually increasing doses and dose intervals, considering the volume and total infusion time. An accelerated titration schedule may be considered if the patient tolerates the SC infusion volumes and the first 2 infusions. Doses less than or equal to 0.4 g/kg may be administered without a titration schedule, provided acceptable patient tolerance.
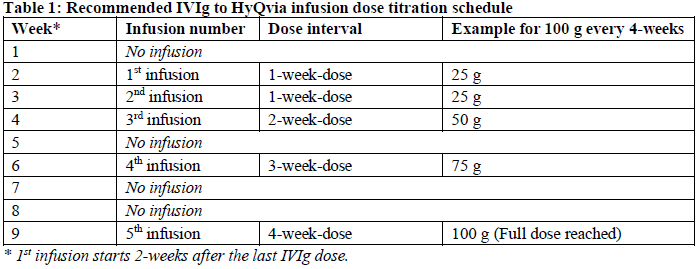
Patients must be on stable doses* of IVIg. Before initiating therapy with the medicinal product, the weekly equivalent dose should be calculated by dividing the last IVIg dose by the IVIg dose interval in weeks. The starting dose and dosing frequency are the same as the patient’s previous IVIg treatment. The typical dosing interval for HyQvia is 4-weeks. For patients with less frequent IVIg dosing (greater than 4-weeks), the dosing interval can be converted to 4-weeks while maintaining the same monthly equivalent IgG dose.
As shown in the table below, the calculated one-week dose (1st infusion) should be administered 2 - weeks after the last IVIg infusion. One week after the first dose, the next weekly equivalent dose (2nd infusion) should be administered. A titration schedule can take up to 9-weeks (Table 1), depending on the dosing interval and tolerability.
*(Variations in the dosing interval of up to ±7 days or monthly equivalent dose amount of up to ±20% between the subject’s IgG infusions are considered a stable dose.)
On a given infusion day, the maximum infusion volume should not exceed 1200 mL for patients weighing ≥ 40 kg or 600 mL for <40 kg. Suppose the maximum daily dose limit is exceeded or the patient cannot tolerate the infusion volume. In that case, the dose may be administered over multiple days in divided doses with 48-to 72-hours between doses to allow absorption of infusion fluid at the infusion site(s). The dose can be administered up to 3 infusion sites with a maximum infusion volume of 600 mL per site (or as tolerated). If using three sites, the maximum is 400 mL per site.
Paediatric population
Replacement therapy
The dosing schedule for children and adolescents (0 to 18 years) is the same as for adults. The dosing is based on body weight and adjusted to the clinical outcome. Currently, available data are described in sections 4.8, 5.1 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Immunomodulatory therapy
The dosing schedule for children and adolescents (0 to 18 years) is the same as for adults. The dosing is based on the calculated weekly equivalent dose and adjusted to the clinical outcome. Currently, available data are described in sections 4.8, 5.1 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Method of administration
The medicinal product is for subcutaneous use only, do not administer intravenously.
Each vial of IG 10% is supplied with the appropriate corresponding quantity of rHuPH20 (see section 6.5 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). The full contents of the rHuPH20 vial should be administered regardless of whether the full content of the IG 10% vial is administered.
The 2 components of the medicinal product must be administered sequentially through the same subcutaneous needle beginning with the rHuPH20 followed by IG 10%.
Example: Patient is prescribed 110 grams (g) of HyQvia: This will require 3 vials of 30 g and 1 vial of 20 g for the total dose of 110 g/1100 mL of the IG 10% component of HyQvia. The volume of rHuPH20 will be (3 x 15 mL + 1 x10 mL) = 55 mL. If the dose is greater than 120 g, HyQvia may be administered over multiple days in divided doses with 48-to 72-hours between doses to allow absorption of infusion fluid at the infusion site(s).
Infusion site leakage can occur during or after subcutaneous administration of immunoglobulin, including HyQvia. Consider using longer needles (12 mm or 14 mm) and/or more than one infusion site. Any change of needle size would have to be supervised by the treating physician.
Home-treatment
In case subcutaneous infusion of HyQvia is used for home treatment, therapy should be initiated and monitored by a physician experienced in the guidance of patients for home treatment. The patient or a caregiver must be instructed in infusion techniques, the use of an infusion pump or syringe driver, the keeping of a treatment diary, recognition of possible severe adverse reactions and measures to be taken in case these occur.
HyQvia can be administered in a full therapeutic dose at up to 3 infusion sites up to every 4-weeks. Adjust the frequency and number of infusion sites, considering volume, total infusion time, and tolerability so that the patient receives the same weekly equivalent dose. If a patient misses a dose, administer the missed dose as soon as possible and then resume scheduled treatments as applicable.
Device-assisted infusion
The IG 10% component should be infused using a pump. The rHuPH20 may be hand-pushed or infused by a pump. A 24-gauge needle may be required to allow patients to infuse at flow rates of 300 mL/hr/infusion site. However, needles with smaller diameters may be used if slower flow rates are acceptable. For the 1.25 mL rHuPH20 vial size use an 18-to 22-gauge needle to withdraw the contents of the vial to prevent stopper push through or coring; for all other vial sizes a needle or needleless device may be used to withdraw the contents of the vial.
Infusion site
The suggested site(s) for the infusion of the medicinal product are the middle to upper abdomen and thighs. If 2 sites are used, the 2 infusion sites should be on contra lateral sides of the body. If using three infusion sites, the sites should be at least 10 cm apart. Avoid bony prominences, or scarred areas. The product should not be infused at or around an infected or acutely inflamed area due to the potential risk of spreading a localized infection. Avoid at least 5 cm away from the umbilicus.
Infusion rate
It is recommended that the rHuPH20 component be administered at a constant rate and that the rate of administration of the IG 10% should not be increased above the recommended rates, particularly when the patient has just started with HyQvia therapy.
First, the full dose of rHuPH20 solution is infused at a rate of 1 to 2 mL/minute (or 60 mL/hr to 120 mL/hr) per infusion site or as tolerated. Within 10 minutes of the rHuPH20, start the infusion of the full dose per site of IG 10% through the same subcutaneous needle set.
The following infusion rates of the IG 10% are recommended per infusion site.
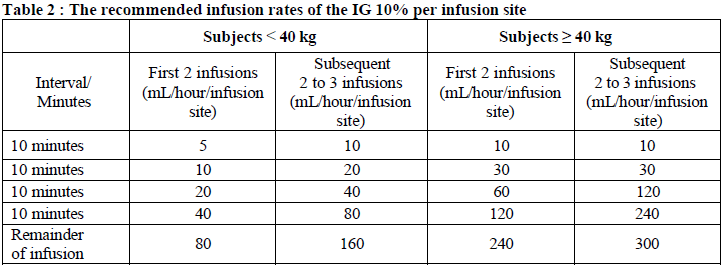
If the patient tolerates the initial infusions at the full dose per site and maximum rate, an increase in the rate of successive infusions may be considered at the discretion of the physician and the patient.
For instructions on the handling and preparation of the medicinal product before administration, see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Contraindications
4.3 Contraindications
HyQvia must not be given intravenously or intramuscularly.
Hypersensitivity to the active substance (IgG) or to any of the excipients listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Hypersensitivity to human immunoglobulins, especially in very rare cases of IgA deficiency when the patient has antibodies against IgA.
Known systemic hypersensitivity to hyaluronidase or rHuPH20.
