METZEN PROLONGED RELEASE TABLET 1000MG [SIN17290P]
Active ingredients: METZEN PROLONGED RELEASE TABLET 1000MG
Product Info
METZEN PROLONGED RELEASE TABLET 1000MG
[SIN17290P]
Product information
Active Ingredient and Strength | METFORMIN HYDROCHLORIDE - 1,000 MG |
Dosage Form | TABLET, EXTENDED RELEASE |
Manufacturer and Country | AMN LIFE SCIENCE PRIVATE LIMITED - INDIA |
Registration Number | SIN17290P |
Licence Holder | PHARMAZEN MEDICALS PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | A10BA02 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
Indication
Reduction in the risk or delay of the onset of type 2 diabetes mellitus in adult, overweight patients with IGT* and/or IFG* who are:
at high risk for developing overt type 2 diabetes mellitus (refer to section Pharmacodynamic properties) and
not suitable for intensive lifestyle modifications.
Treatment with Metzen prolonged release tablets must be based on a risk score incorporating appropriate measures of glycaemic control and including evidence of high cardiovascular risk (refer to section Pharmacodynamic properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
*IGT: Impaired Glucose Tolerance; IFG: Impaired Fasting Glucose.
Treatment of type 2 diabetes mellitus in adults, particularly in overweight patients, when dietary management and exercise alone does not result in adequate glycaemic control.
Metzen prolonged release tablets may be used as monotherapy or in combination with other oral antidiabetic agents, or with insulin.
Dosing
Recommended Dosage
Adults with normal renal function (GFR ≥90 mL/min)
Reduction in the risk or delay of the onset of type 2 diabetes
Metformin should only be considered where intensive lifestyle modifications are not feasible.
The therapy should be initiated with one tablet Metzen 500 mg prolonged release tablet once daily with the evening meal.
After 10 to 15 days dose adjustment on the basis of blood glucose measurements is recommended. A slow increase of dose may improve gastro-intestinal tolerability. The maximum recommended dose is 2000 mg once daily with the evening meal.
It is recommended to regularly monitor (every 3–6 months) the glycaemic status (OGTT and/or FPG and/or HbA1c value) as well as the risk factors to evaluate whether treatment needs to be continued, modified or discontinued.
A decision to re-evaluate therapy is also required if the patient subsequently implements improvements to diet and/or exercise, or if changes to the medical condition will allow increased lifestyle interventions to be possible.
Monotherapy and combination with other oral antidiabetic agents in Type 2 diabetes mellitus:
Metzen 500 mg:
The usual starting dose is one tablet of Metzen 500 mg prolonged release tablet once daily with evening meal.
Metzen prolonged release tablets must be swallowed whole and never crushed or chewed.
After 10 to 15 days the dose should be adjusted on the basis of blood glucose measurements.
To improve gastrointestinal tolerability, dosage increase is recommended in increments of 500 mg every 10 to 15 days, up to 2000 mg once daily with the evening meal. If glycaemic control is not achieved on Metzen prolonged release tablets 2000 mg once daily, Metzen prolonged release tablets 1000 mg twice daily should be considered, with both doses being given with food. If glycaemic control is still not achieved, patients may be switched to standard metformin tablets to a maximum dose of 3000 mg daily.
In patients already treated with metformin tablets, the starting dose of Metzen prolonged release tablets should be equivalent to the daily dose of metformin immediate release tablets. In patients treated with metformin at a dose above 2000 mg daily, switching to Metzen prolonged release tablet is not recommended.
Metzen 750 mg:
Metzen 750 mg prolonged release tablet is intended for patients who are already treated with metformin tablets (extended or immediate release). The dose of Metzen 750 mg prolonged release tablet should be equivalent to the daily dose of metformin tablets (extended or immediate release), up to a maximum dose of 1500 mg given with the evening meal.
After 10 to 15 days dose adjustment on the basis of blood glucose measurements is recommended. A slow increase of dose may improve gastrointestinal tolerability. The recommended dose of Metzen 750 mg prolonged release tablet is 2 tablets once daily. If glycaemic control is not achieved on Metzen prolonged release tablets 1500 mg once daily, the dose may be increased to a maximum dose of 2000 mg once daily. If glycaemic control is not achieved on Metzen prolonged release tablets 2000 mg once daily, Metzen prolonged release tablets 1000 mg twice daily should be considered, with both doses being given with food. If glycaemic control is still not achieved, patients may be switched to standard metformin tablets to a maximum dose of 3000 mg daily.
Metzen 1000 mg:
Metzen 1000 mg prolonged release tablets is intended as a maintenance therapy for patients treated with either 1000 mg or 2000 mg of metformin hydrochloride. On switch, the daily dose of Metzen prolonged release tablets should be equivalent to the current daily dose of metformin hydrochloride.
Metzen 1000 mg prolonged release tablets should be taken once daily with the evening meal at a maximum recommended dose of 2 tablets per day. If glycaemic control is not achieved on Metzen prolonged release tablets 2000 mg once daily, Metzen prolonged release tablets 1000 mg twice daily should be considered, with both doses being given with food. If glycaemic control is still not achieved, patients may be switched to standard metformin tablets to a maximum dose of 3000 mg daily.
Combining Metzen prolonged release tablets dosage strengths
The combined use of different strengths of Metzen prolonged release tablets 500, Metzen prolonged release tablets 750 or Metzen prolonged release tablets 1000 is not recommended. Only one strength (Metzen prolonged release tablets 500, Metzen prolonged release tablets 750 or Metzen prolonged release tablets 1000) should be used at a time in order to avoid accidentally exceeding the recommended upper daily dose limit of 2000mg.
Transfer from another oral antidiabetic agent
If transfer from another oral antidiabetic agent is intended, discontinue the other agent and initiate Metzen 500 mg prolonged release tablet at the dose indicated above, before switching to Metzen 750mg prolonged release tablet or Metzen 1000mg prolonged release tablet.
Combination with insulin:
Metformin and insulin may be used in combination therapy to achieve better blood glucose control. The usual starting dose of Metzen prolonged release tablet is one 500 mg tablet once daily, while insulin dosage is adjusted on the basis of blood glucose measurements.
Elderly:
Due to the potential for decreased renal function in elderly subjects, the metformin dosage should be adjusted based on renal function. Regular assessment of renal function is necessary (refer to section warnings and precautions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Benefit in the reduction of risk or delay of the onset of type 2 diabetes mellitus has not been established in patients 75 years and older (refer to section pharmacodynamics properties – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information) and metformin initiation is therefore not recommended in these patients (refer to section warnings and precautions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Renal impairment
A GFR should be assessed before initiation of treatment with metformin containing products and at least annually thereafter. In patients at an increased risk of further progression of renal impairment and in the elderly, renal function should be assessed more frequently, e.g. every 3–6 months.
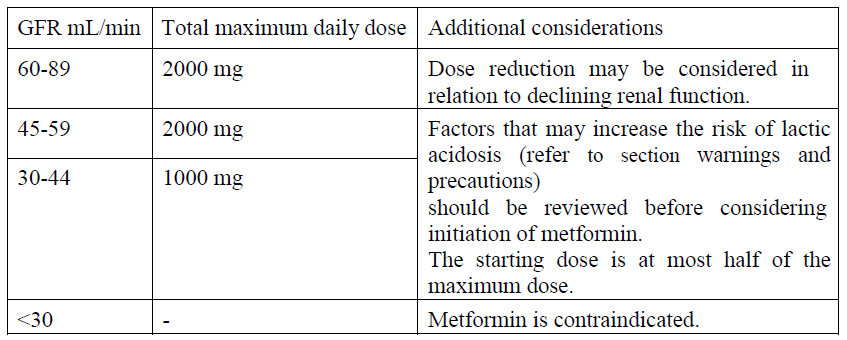
Paediatric population:
In the absence of available data, Metzen prolonged release tablets should not be used in children.
Contraindications
Contraindications
Hypersensitivity to Metformin Hydrochloride or to any of the excipients.
Any type of acute metabolic acidosis (such as lactic acidosis, diabetic keto-acidosis)
Diabetic pre-coma
Severe renal failure (GFR <30 ml/min)
Acute conditions with the potential to alter renal function such as:
Dehydration
Severe infection
Shock
Disease which may cause tissue hypoxia (especially acute disease, or worsening of chronic disease) such as: decompensated heart failure, respiratory failure, recent myocardial infarction, shock.
Hepatic insufficiency, acute alcohol intoxication, alcoholism.
