METALYSE FOR INJECTION 5,000 U/VIAL [SIN17293P]
Active ingredients: METALYSE FOR INJECTION 5,000 U/VIAL
Product Info
METALYSE FOR INJECTION 5,000 U/VIAL
[SIN17293P]
Product information
Active Ingredient and Strength | TENECTEPLASE - 5,000 UNITS (25 MG)/VIAL |
Dosage Form | INJECTION, POWDER, FOR SOLUTION |
Manufacturer and Country | BOEHRINGER INGELHEIM PHARMA GMBH & CO. KG - GERMANY |
Registration Number | SIN17293P |
Licence Holder | BOEHRINGER INGELHEIM SINGAPORE PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | B01AD11 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
METALYSE is indicated in adults for the thrombolytic treatment of acute ischaemic stroke (AIS) within 4.5 hours from last known well and after exclusion of intracranial haemorrhage.
Dosing
4.2 Posology and method of administration
Posology
METALYSE should be administered as early as possible and no later than 4.5 hours after last known well and after exclusion of intracranial haemorrhage by appropriate imaging techniques. The treatment effect is time-dependent; therefore, earlier treatment increases the probability of a favourable outcome.
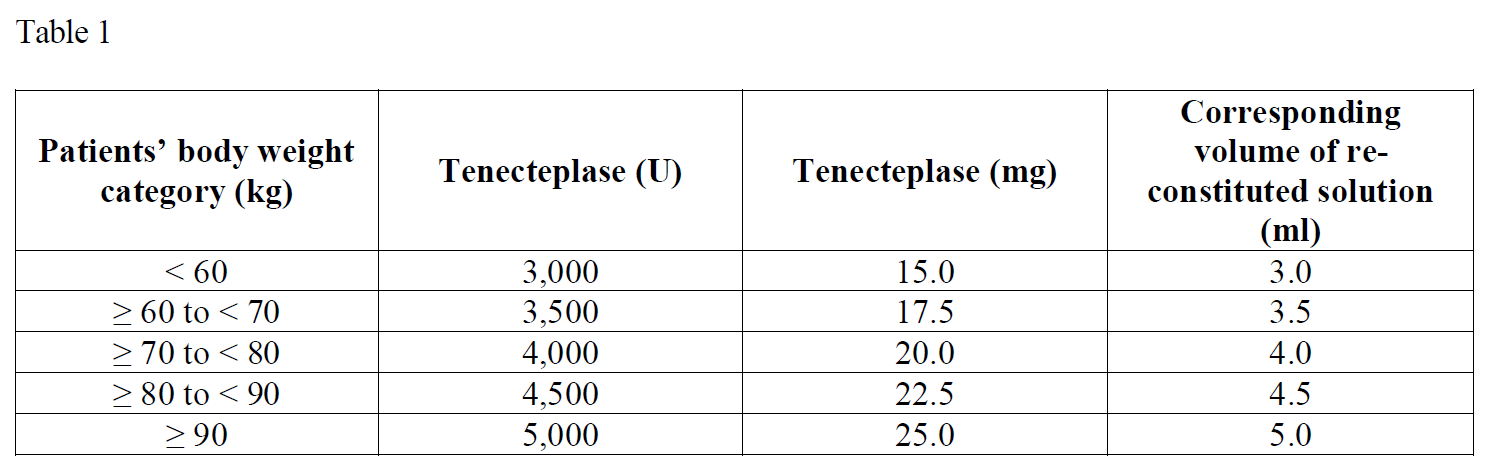
METALYSE should be administered on the basis of body weight, with a maximum single dose of 5,000 units (25 mg) tenecteplase.
Benefit-risk of tenecteplase treatment should be carefully evaluated in patients weighing 50 kg or less due to limited availability of data.
The volume required to administer the correct total dose can be calculated from the following scheme (table 1):
Adjunctive therapy:
Drugs affecting coagulation/platelet function
The safety and efficacy of this regimen with concomitant treatment with heparin or platelet aggregation inhibitors such as acetylsalicylic acid during the first 24 hours after treatment with METALYSE have not been investigated sufficiently. Therefore, administration of intravenous heparin or platelet aggregation inhibitors such as acetylsalicylic acid should be avoided in the first 24 hours after treatment with METALYSE due to an increased haemorrhagic risk.
If heparin is required for other indications the dose should not exceed 10,000 international units per day, administered subcutaneously.
Method of administration
The reconstituted solution should be administered intravenously and is for immediate use.
The required dose should be administered as a single intravenous bolus over 5 to 10 seconds.
HANDLING INSTRUCTIONS
METALYSE should be reconstituted by adding the appropriate volume of sterile water for injection to the vial containing the powder for solution for injection using a needle and a syringe (not provided in the package).
Remove the crimp cap from the vial.
Fill a syringe with 5 ml of sterile water for injection and penetrate the vial stopper in the middle with the needle.
Add all the sterile water for injection into the vial by pushing the syringe plunger down slowly to avoid foaming.
Keep the syringe attached to the vial and reconstitute by swirling gently.
The reconstituted preparation is a colourless to pale yellow, clear solution. Only clear solution without particles should be used.
Directly before the solution is administered, invert the vial with the syringe still attached, so that the syringe is below the vial.
Transfer the appropriate volume of reconstituted solution of METALYSE into the syringe, based on the patient’s weight (see table 1).
A pre-existing intravenous line, which has been used for administration of 0.9% sodium chloride solution only, may be used for administration of METALYSE. METALYSE should not be mixed with other drugs, neither in the same vial nor the same venous line (not even with heparin).
METALYSE should be administered as a single dose to the patient, intravenously over 5 to 10 seconds. It should not be administered into a line containing dextrose as METALYSE is incompatible with dextrose solution.
The line should be flushed after METALYSE injection for proper delivery.
Any unused solution should be discarded.
Contraindications
4.3 Contraindications
METALYSE is contraindicated in
patients with known hypersensitivity to the active substance tenecteplase, gentamicin (a trace residue from the manufacturing process) or to any of the excipients
situations associated with a risk of bleeding such as:
severe stroke as assessed clinically (e.g. NIHSS > 25) and/or by appropriate imaging techniques
major surgery, biopsy of a parenchymal organ, or significant trauma within the past 2 months
recent trauma to the head or cranium
symptoms of ischaemic attack beginning more than 4.5 hours prior to injection or symptoms for which the onset time is unknown and could potentially be more than 4.5 hours ago
seizure at onset of stroke
administration of heparin within the previous 48 hours and a thromboplastin time exceeding the upper limit of normal for laboratory
patients with any history of prior stroke and concomitant diabetes
prior stroke within the last 3 months
platelet count of below 100,000/mm3
systolic blood pressure (BP) > 185 mmHg or diastolic BP > 110 mmHg, or aggressive management (intravenous pharmacotherapy) necessary to reduce BP to these limits
blood glucose < 50 mg/dL or > 400 mg/dL (< 2.8 mM or > 22.2 mM)
significant bleeding disorder at present or within the past 6 months, known haemorrhagic diathesis
any history of central nervous system damage (i.e. neoplasm, aneurysm, intracranial or spinal surgery)
severe uncontrolled arterial hypertension (see section 4.4 SPECIAL WARNINGS AND PRECAUTIONS FOR USE – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information)
prolonged or traumatic cardiopulmonary resuscitation (> 2 minutes) within the past 2 weeks
severe hepatic dysfunction, including hepatic failure, cirrhosis, portal hypertension (oesophageal varices) and active hepatitis
active ulcerative gastro-intestinal disease
known arterial aneurysm and/or arterial/venous malformation
neoplasm with increased bleeding risk
bacterial endocarditis, pericarditis
acute pancreatitis
acute ischaemic stroke without disabling neurological deficit
history or evidence or suspicion of intracranial haemorrhage including subarachnoid haemorrhage
patients receiving effective anticoagulation (e.g. vitamin K antagonists with INR > 1.3) (please see section 4.4 SPECIAL WARNINGS AND PRECAUTIONS FOR USE, subsection “Bleeding” – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information)
