IMDELLTRA POWDER FOR SOLUTION FOR INFUSION 1 MG/VIAL [SIN17299P]
Active ingredients: IMDELLTRA POWDER FOR SOLUTION FOR INFUSION 1 MG/VIAL
Product Info
IMDELLTRA POWDER FOR SOLUTION FOR INFUSION 1 MG/VIAL
[SIN17299P]
Product information
Active Ingredient and Strength | TARLATAMAB - 1 MG/VIAL |
Dosage Form | INJECTION, POWDER, LYOPHILIZED, FOR SOLUTION |
Manufacturer and Country | AMGEN INC. (AMGEN THOUSAND OAKS OR ATO) - UNITED STATES |
Registration Number | SIN17299P |
Licence Holder | AMGEN BIOTECHNOLOGY SINGAPORE PTE LTD |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | L01FX33 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
1 INDICATIONS AND USAGE
IMDELLTRA is indicated for the treatment of adult patients with extensive stage small cell lung cancer (ES-SCLC) with disease progression on or after platinum-based chemotherapy.
Dosing
2 DOSAGE AND ADMINISTRATION
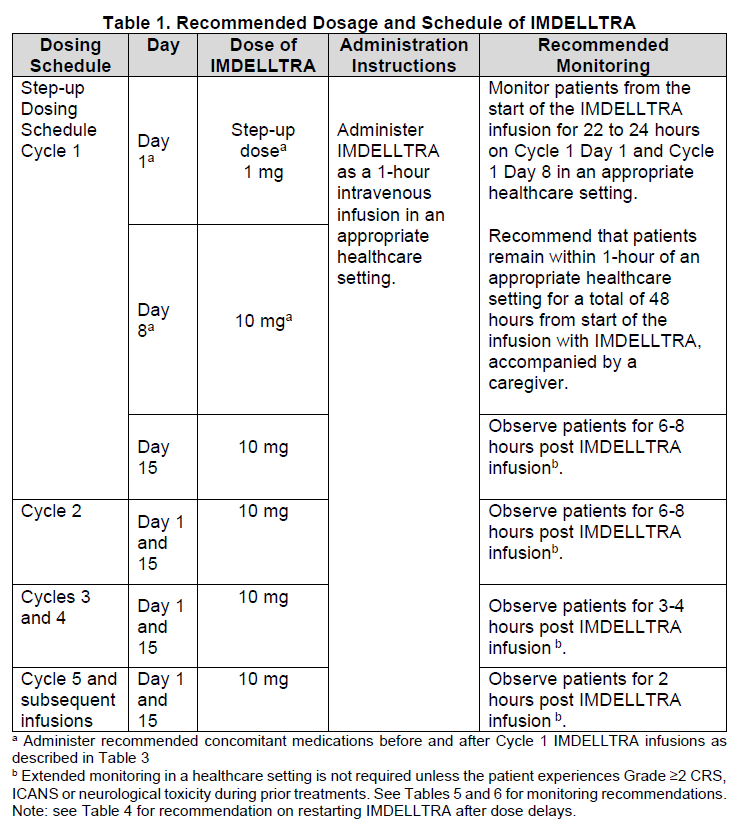
2.1 Important Dosing Information
Administer IMDELLTRA according to the step-up dosing schedule in Table 1 to reduce the incidence and severity of cytokine release syndrome (CRS) [see Dosage and Administration (2.2)].
For Cycle 1, administer recommended concomitant medications in Table 3 before and after Cycle 1 IMDELLTRA infusions to reduce the risk of CRS reactions [see Dosage and Administration (2.3)].
IMDELLTRA should only be administered by a qualified healthcare professional with appropriate medical support to manage severe reactions such as CRS and neurologic toxicity including immune effector cell-associated neurotoxicity syndrome (ICANS) [see Warnings and Precautions (5.1, 5.2) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
Due to the risk of CRS and neurologic toxicity, including ICANS, monitor patients from the start of the IMDELLTRA infusion for 22 to 24 hours on Cycle 1 Day 1 and Cycle 1 Day 8 in an appropriate healthcare setting [see Dosage and Administration (2.5) and Warnings and Precautions (5.1, 5.2) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
Recommend that patients remain within 1-hour of an appropriate healthcare setting for a total of 48 hours from start of the infusion with IMDELLTRA following Cycle 1 Day 1 and Cycle 1 Day 8 doses, accompanied by a caregiver.
Prior to administration of IMDELLTRA evaluate complete blood count, liver enzymes and bilirubin before each dose, and as clinically indicated [see Warnings and Precautions (5.3, 5.5) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
Ensure patients are well hydrated prior to administration of IMDELLTRA [see Warnings and Precautions (5.1) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
2.2 Recommended Dosage and Administration
Administer IMDELLTRA as an intravenous infusion over one hour.
The recommended step-up dosage schedule for IMDELLTRA is provided in Table 1. Administer following step-up dosing to reduce the incidence and severity of CRS.
After step-up dosing schedule, administer IMDELLTRA biweekly (every 2 weeks) until disease progression or unacceptable toxicity.
Administration
The intravenous (IV) catheter for concomitant medications administration can be used to administer the IMDELLTRA infusion.
To ensure patency, flush the IV catheter over 3–5 mins using 0.9% Sodium Chloride for Injection.
Administer the reconstituted and diluted IMDELLTRA as an intravenous infusion at a constant flow rate using an infusion pump. The pump should be programmable, lockable, non-elastomeric, and have an alarm.
Table 2 provides the infusion duration and rate.

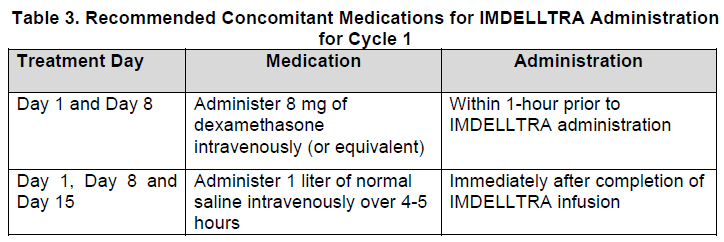
2.3 Recommended Concomitant Medications for IMDELLTRA Administration for Cycle 1
Administer recommended concomitant medications for IMDELLTRA administration during Cycle 1 as presented in Table 3 to reduce the risk of cytokine release syndrome [see Warnings and Precautions (5.1) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information].
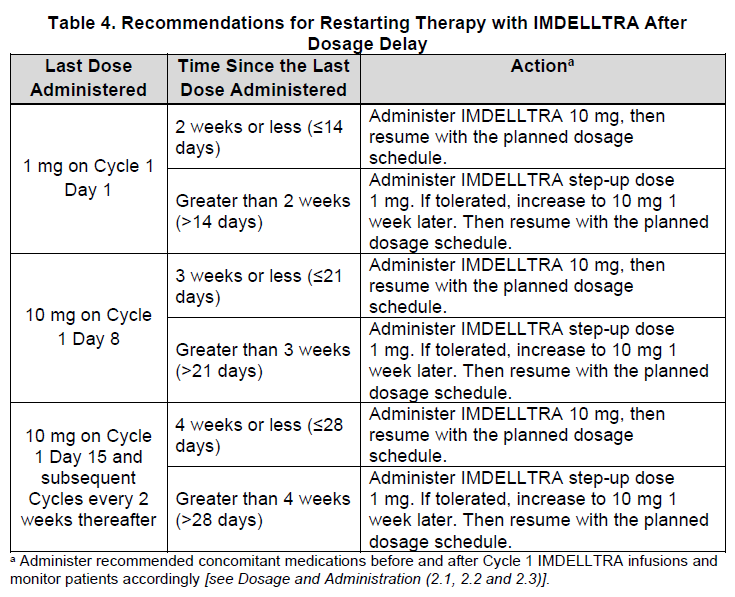
2.4 Restarting IMDELLTRA After Dosage Delay
If a dose of IMDELLTRA is delayed, restart therapy based on the recommendation as listed in Table 4 and resume the dosing schedule accordingly [see Dosage and Administration (2.2)]. Administer recommended concomitant medications as indicated in section 2.3.
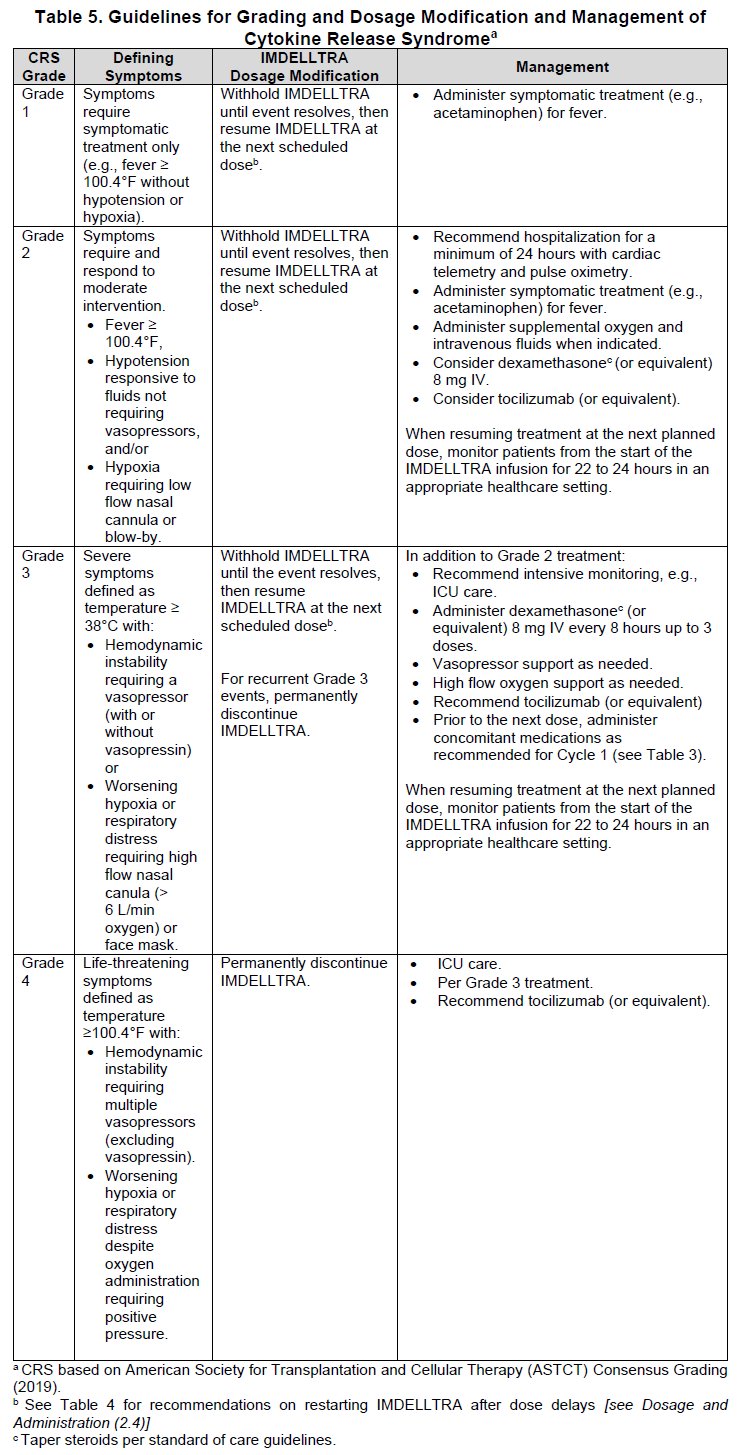
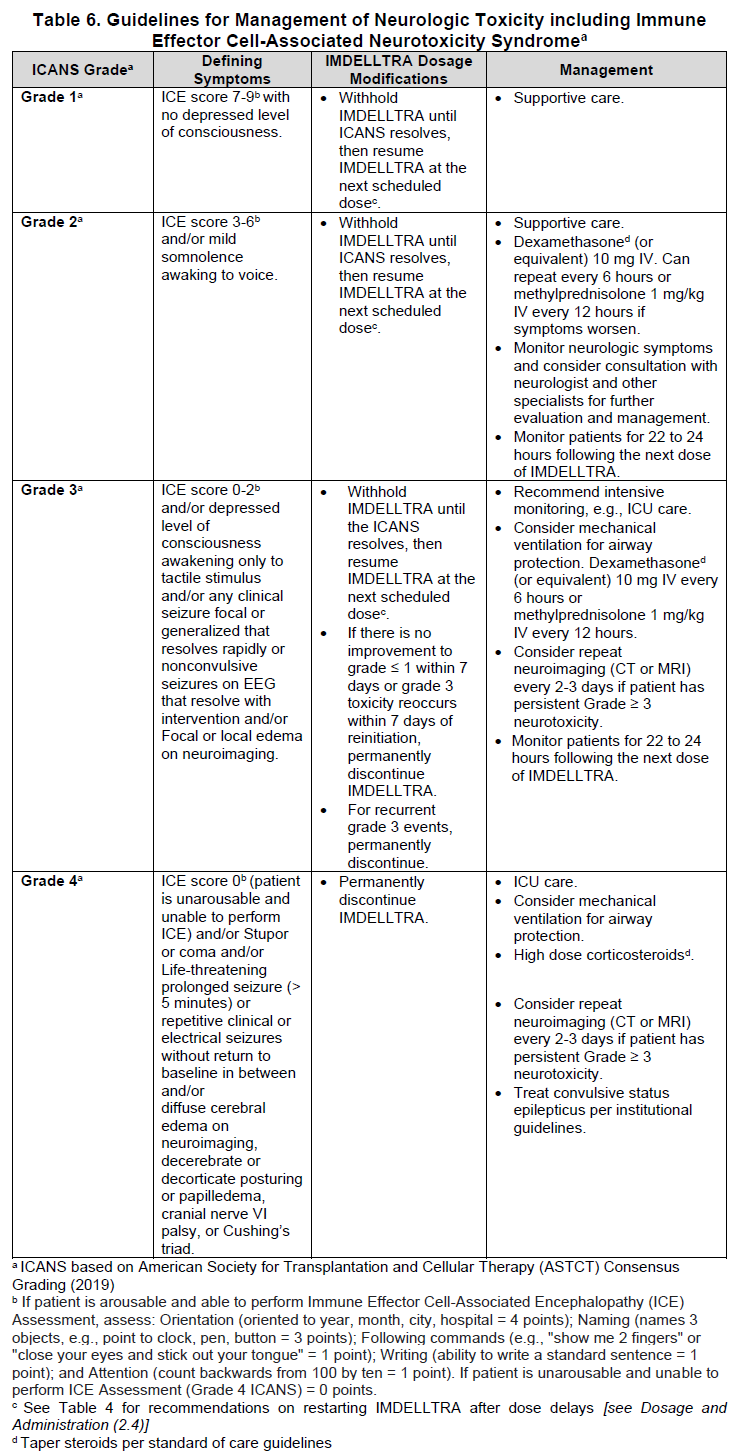
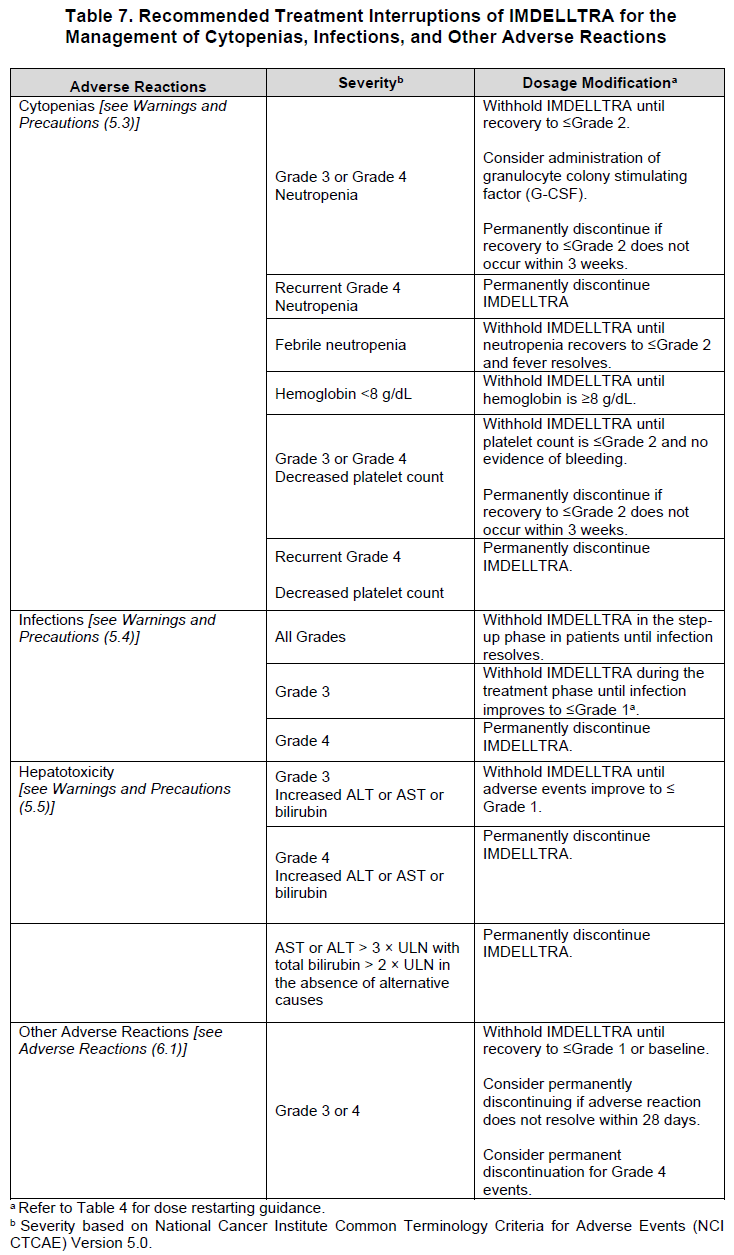
2.5 IMDELLTRA Dosage Modifications and Adverse Reaction Management
No dose reduction for IMDELLTRA is recommended. See Table 5 and Table 6 for recommended actions for the management of CRS, neurologic toxicity including ICANS respectively and Table 7 for cytopenias, infections and other adverse reactions.
Cytokine Release Syndrome (CRS)
Diagnose CRS based on clinical presentation [see Warnings and Precautions (5.1) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information]. Evaluate for and treat other causes of fever, hypoxia, and hypotension.
If CRS is suspected, manage according to the recommendations in Table 5. Monitor patients who experience Grade 2 or higher CRS (e.g., hypotension not responsive to fluids, or hypoxia requiring supplemental oxygen) with continuous cardiac telemetry and pulse oximetry.
For severe or life-threatening CRS, recommend administering tocilizumab or equivalent therapy and intensive monitoring (e.g., ICU) for supportive therapy. Perform laboratory testing to monitor for disseminated intravascular coagulation (DIC), hematology parameters, as well as pulmonary, cardiac, renal, and hepatic function.
Table 5 provides the guidelines for grading and dosage modification and management of cytokine release syndrome.
Neurologic Toxicity including ICANS
At the first sign of neurologic toxicity, including ICANS, withhold IMDELLTRA and consider neurology evaluation. Rule out other causes of neurologic symptoms. Provide supportive therapy, which may include intensive care, for severe or life-threatening neurologic toxicities, including ICANS [see Warnings and Precautions (5.2) – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information]. Manage ICANS and neurologic toxicity according to the recommendations in Table 6 and consider further management per current practice guidelines.
2.6 Preparation
Material Compatibility Information
IV bags composed of ethyl vinyl acetate (EVA), polyolefin, and polyvinyl chloride, (PVC) have been shown to be compatible with IMDELLTRA at the specified administration conditions.
IV line and catheter materials composed of polyolefin, PVC, and polyurethane have been shown to be compatible with IMDELLTRA at the specified administration conditions.
The use of Closed System Transfer Device (CSTD) is not recommended due to potential wrong dose medication error risk. Amgen has not performed compatibility testing of vial adaptor CSTDs with IMDELLTRA.
Step 1: Reconstitute IMDELLTRA with Sterile Water for Injection
Table 8 provides the required amount of sterile water for injection required to reconstitute IMDELLTRA 1 mg and 10 mg vials.
Do not use IV Solution Stabilizer (IVSS) to reconstitute IMDELLTRA.
The IV Solution Stabilizer (IVSS) is used to coat the intravenous bag prior to addition of reconstituted IMDELLTRA to prevent adsorption of IMDELLTRA to IV bags and IV tubing.

Using a needle and syringe filled with the required amount of sterile water, inject the sterile water against the glass vial. Avoid injecting the water directly onto the powder to prevent foaming.
Gently swirl the contents to mix. Do not shake.
Inspect parenteral drug products for particulate matter and discoloration prior to administration. Inspect that the solution is clear to opalescent, colorless to slightly yellow. Do not use if the solution is cloudy or has particulates.
Further dilute reconstituted IMDELLTRA.
The reconstituted IMDELLTRA must be further diluted within 4 hours of reconstitution or discarded.
Prepare the infusion bag: Steps 2 to 5
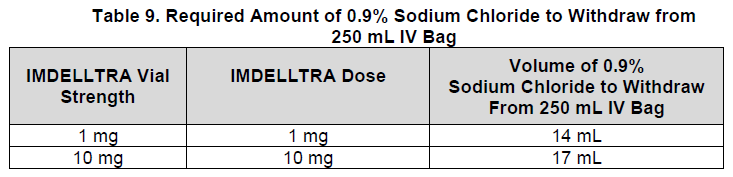
Step 2: Withdraw 0.9% Sodium Chloride for Injection
Using a 250 mL prefilled bag of 0.9% Sodium Chloride for Injection, withdraw the amount of sodium chloride specified in Table 9 and discard.
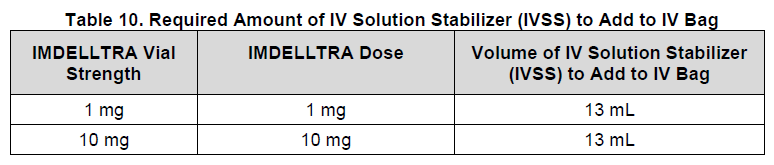
Step 3: Add IV Solution Stabilizer to the infusion bag
Inject 13 mL of IV Solution Stabilizer (IVSS) into the 250 mL 0.9% Sodium Chloride infusion bag, see Table 10.
Gently mix the contents of the infusion bag to avoid foaming. Do not shake.
Step 4: Dilute the reconstituted IMDELLTRA into the infusion bag
Transfer the required volume of reconstituted IMDELLTRA listed in Table 11 to the infusion bag (containing IV Solution Stabilizer).
NOTE: the final concentrations for the different strength vials are NOT the same following reconstitution and further dilution.

Gently mix the contents of the bag. Do not shake.
Step 5: Remove air from IV bag
Remove air from the prepared IV bag using an empty syringe to avoid foaming.
Step 6: Prime IV tubing
Prime intravenous tubing with either 0.9% Sodium Chloride for Injection or with the final prepared product.
See Table 12 for maximum storage time of prepared IMDELLTRA infusion.
Prepared IMDELLTRA Infusion Bag Storage Requirements
Administer reconstituted and diluted IMDELLTRA immediately.
Table 12 displays the maximum storage time for the prepared IMDELLTRA infusion bag.
Maximum storage time includes total duration from the time of reconstitution of the vial of IMDELLTRA to the end of the infusion.

Contraindications
4 CONTRAINDICATIONS
None.
