FINJUVE FOR MEN CUTANEOUS SPRAY, SOLUTION 2.275 MG/ML [SIN17303P]
Active ingredients: FINJUVE FOR MEN CUTANEOUS SPRAY, SOLUTION 2.275 MG/ML
Product Info
FINJUVE FOR MEN CUTANEOUS SPRAY, SOLUTION 2.275 MG/ML
[SIN17303P]
Product information
Active Ingredient and Strength | FINASTERIDE - 2.275 MG/ML |
Dosage Form | SPRAY |
Manufacturer and Country | ALMIRALL HERMAL GMBH - GERMANY |
Registration Number | SIN17303P |
Licence Holder | A. MENARINI SINGAPORE PTE. LTD. |
Forensic Classification | PRESCRIPTION ONLY MEDICINES |
Anatomical Therapeutic Chemical (ATC) code | D11AX10 |
Prescription-only Medicines with Exemptions for Supply without Prescription | NA |
Indication
4.1 Therapeutic indications
Finjuve is indicated for the topical treatment of adult men from 18 to 41 years of age with mild to moderate male pattern hair loss (androgenetic alopecia) to increase hair growth and prevent further hair loss.
Dosing
4.2 Posology and method of administration
Posology
Finjuve should be applied once daily to bald areas of the scalp. Depending on the size of the baldness, 1 to 4 non-overlapping spray actuations (50 to 200 microlitres of solution) can be used.
The dose selected for the size of baldness should not be increased beyond the maximum of 4 actuations. Efficacy and duration of treatment should continuously be assessed by the treating physician. Generally, 3 to 6 months of once daily treatment are required before evidence of hair growth can be expected. Continuous use is recommended to sustain benefit. There is no clinical experience with Finjuve beyond 6 months.
The bottle contains up to 180 actuations (delivering 50 microlitres each), which is sufficient for 45 days of treatment when the maximum dose of 4 actuations once daily is administered, 60 days of treatment for 3 actuations once daily, 90 days of treatment for 2 actuations once daily, and 180 days of treatment for 1 actuation once daily. The bottle should not be used beyond 180 actuations as it could result in the delivery of an insufficient dose. Patients should be advised accordingly.
Patients with renal or hepatic impairment
No dose adjustment is required in patients with renal or hepatic impairment (see section 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Paediatric population
The safety and efficacy of Finjuve in children and adolescents under 18 years of age have not been established (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Method of administration
Finjuve is for cutaneous use. It is only to be used on the scalp.
Assembly of the spray applicator
The presentation of Finjuve contains 2 separate components: a bottle with an attached metering pump, and a cone. These components require assembly prior to first use.
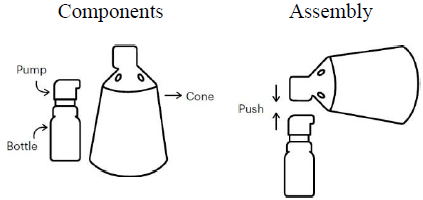
Before using Finjuve for the first time, the pump must be primed by means of 4 full actuations, directing the sprayed solution toward the bathroom sink (the sink must be rinsed afterwards). When Finjuve has not been used for at least 2 weeks the pump must be reprimed by means of 1 full actuation. Other than this, there is no need to shake or to prime the pump at each use.
Handling of the spray applicator
Finjuve should be administered by the patient himself. Hair and scalp should be fully dry prior to application of the solution. The solution should not be sprayed towards the face and should not come into contact with the hands or any part of the body other than the area to be treated on the scalp. In case of unintended contact with the solution, the affected body part should be washed thoroughly.
When spraying the scalp, the cone must be in contact with the scalp to avoid finasteride dispersion in the air. The bald scalp area covered by the cone limits the maximum treatment area for 1 actuation. To cover an area larger than the cone diameter 2, 3, or 4 actuations may be prescribed. In these cases, before applying the second, third, or fourth actuation, the cone should be moved to an area of the scalp next to, but not touching, the area of any previous actuations to avoid spray overlap.
Immediately after application the patient should avoid contact between the treated scalp and surfaces (e.g. pillows, helmets, hats etc.) until the solution has dried. Once applied, Finjuve should be left in place for at least 6 hours.
See section 4.4 for advice if the patient may be in contact with a pregnant woman or a woman who may become pregnant, or children and adolescents – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Contraindications
4.3 Contraindications
Finjuve is not intended for use by women or children and adolescents under 18 years of age.
Women who are pregnant or may become pregnant (see sections 4.4, 4.6 and 5.3 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
Hypersensitivity to the active substance or to any of the excipients listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
